Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
A Comparative Study of Early Detection of Parkinsons Disease Using Machine Learning Techniques
Authors: Nandhiniumesh K
DOI Link: https://doi.org/10.22214/ijraset.2023.48979
Certificate: View Certificate
Abstract
For many years, Parkinson\'s disease (PD) has been viewed as a tragedy for humanity. A lot of research is being done on how to detect it using an automated method. This necessitates the use of a machine learning model for PD early diagnosis. Studying the currently employed computational intelligence strategies in the field of research used for PD detection is a crucial requirement for developing a full proof model. Numerous models now in use either concentrate on a single modality or only briefly consider several modalities. This prompted us to conduct a comparative literature review of four primary PD early detection techniques, specifically tremor at rest, bradykinesia, stiffness, and voice impairment. Modern Machine learning methods including K-nearest neighbours, Decision Tree, Support Vector Machine, and Logistic Regression (KNN), Stochastic Gradient Descent (SGD), and Gaussian Naive Bayes (GNB) are applied in these modalities with the corresponding datasets. Additionally, ensemble methods like Hard Voting (HV), Adaptive Boosting (AB), and Random Forest Classifier (RF) are used.
Introduction
I. INTRODUCTION
Parkinson's disease (PD) is a long-term neurological condition that impairs a person's ability to speak as well as other body functions. After Alzheimer's disease, it is the second most common neurodegenerative condition. Dr. James Parkinson was the first to describe this condition called "paraesthesia agitation" or named "the shaking palsy". In the 21st century, PD is a ubiquitous issue. In 2015, PD affected 6.2 million people and resulted in about 1, 17,400 deaths globally. This accounts for various research to be undertaken to study and eventually cure the disease. The loss of nerve cells in the part of the brain called the substantial Ingra causes PD. These nerve cells or neurons create an organic chemical named dopamine which acts as a neurotransmitter between the parts of the brain and central nervous system that helps to control and co-ordinate body movements. Although this disease can be diagnosed at an early stage its long term treatment is not yet discovered. The clinical diagnosis of the patient by the doctor was focused on his/her sense and experience, based on his/her knowledge and studying previous cases of PD from large databases in the hospitals. But with the advent of strong tools like Artificial Intelligence and Machine Learning, this took a subtle turn, various state-of-the-art machine learning tools and techniques analysed the high dimensions of data in the datasets which made the work of prediction simple.
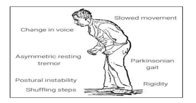
The primary symptoms of PD were the motor dysfunctions, which involved tremors of limbs, hand, legs, and jaws, bradykinesia or slowness of movement, rigidity in limbs which is observable in the PD affected patient’s gait and postural instability . Furthermore, there are several other symptoms like loss of memory and depression which are termed as non-motor symptoms. PD can be diagnosed, but its effective treatment is a challenging task. There is no definitive cure discovered for PD or either to show its progression, but there are various rating methods like Unified Parkinson’s Disease Rating Scale (UPDRS) and MDS-UPDRS, which helps to estimate the severity of the disease. Sometimes there is a possibility that patients do not cooperate with the doctors while examination which causes imprecise and inaccurate results. So, the usage of automated tools like machine learning would ease the task of clinicians and would greatly improve the quantitative measurement of bradykinesia.
II. RELATED WORKS
Parkinson's disease (PD) is a neurodegenerative condition brought on by the brain's dopamine levels declining. The four main symptoms are tremor, bradykinesia, stiffness, and postural stability. The abnormal stillness or slowness in motion brought on by bradykinesia impacts patients' daily lives much like other symptoms do. [1] An evaluation technique based on the Unified Parkinson's Disease Rating Scale is used to clinically evaluate the severity of these symptoms (UPDRS). UPDRS, however, does not use a test based on quantitative measures, but rather depends on the visual observations of medical professionals. This makes it tough to reproduce and makes it arbitrary. Due to these two significant drawbacks, scientists create unique instruments for their research. But this results in non-standard measurements and dependability problems. Therefore, the motor component of the UPDRS III was assessed bilaterally on 24 PD patients.
Parkinson’s disease (PD) is a neurodegenerative movement disease affecting over 6 million people worldwide. Current diagnosis is based on clinical and observational criteria only, resulting in a high misdiagnosis rate. Approximately 75% of people with PD have hand tremor, which can precede clinical diagnosis by up to 6 years. Previous studies have shown that early PD can be accurately detected from keystroke features while typing, and this study investigated whether tremor can be detected as well. [2] Typing data from 76 subjects, with and without PD, including 27 with PD and 15 with tremor, was analysed and showed that hand tremor in PD can be detected from keystroke features. This novel technique has not been used before and was able to achieve an overall sensitivity of 67% and a specificity of 80% and was also able to differentiate PD tremor from essential tremor. This means that the diagnosis of early PD through typing can achieve the clinical requirement of at least two cardinal features being present (bradykinesia and tremor). The method does not need any specialised tools, and it only requires a little more than half a page of typing. This could occur while the patient is at home using a computer to type normally.
Automatic PD symptom detection and disease progression tracking could be accomplished by machine learning algorithms using data streams obtained from soft wearable sensors. However, gathering such data is expensive, and these algorithms must be trained on annotated data from clinical specialists who can identify symptoms. To successfully deploy these models outside of the clinic, it is essential to comprehend how many sensors and how much labelled data are needed. Here, during a series of clinical evaluations that were completed over the course of 1 day and again 2 weeks later, we recorded movement data from 20 people with Parkinson's disease using 6 flexible wearable sensors. 13 common actions, including walking or typing, were completed by participants, and a clinician graded the severity of the symptoms (bradykinesia and tremor). Then, [3] using data from tasks completed by other people, we trained convolutional neural networks and statistical ensembles to determine whether a segment of movement displayed symptoms of bradykinesia or tremor. According to our findings, utilizing wearable sensors on both sides of the body does not increase performance when trying to identify bradykinesia and tremor in the upper extremities. Performance can be enhanced by adding more test subjects, but repeated assessments with the same subjects—even at various medication states—does not significantly enhance detection over days. According to our findings, PD symptoms can be recognised throughout a variety of activities and are best modelled by a dataset that includes numerous individuals.
Adaptive deep brain stimulation (aDBS) is an emerging method to alleviate the side effects and improve the efficacy of conventional open-loop stimulation for movement disorders. [4] However, current adaptive DBS techniques are primarily based on single-feature thresholding, precluding an optimized delivery of stimulation for precise control of motor symptoms. Here, we propose to use a machine learning approach for resting-state tremor detection from local field potentials (LFPs) recorded from sub thalamic nucleus (STN) in 12 Parkinson’s patients. We compare the performance of state-of-the-art classifiers and LFP-based biomarkers for tremor detection, showing that the high-frequency oscillations and Horthy parameters achieve a high discriminative performance. In addition, using Kaman filtering in the feature space, we show that the tremor detection performance significantly improves (F(1,15)=32.16, p<0.0001). The proposed method holds great promise for efficient on-demand delivery of stimulation in Parkinson’s disease.
Background Wearable sensors have been shown in multiple studies to be a quantitative and trustworthy method for tracking patients' motor performance. These sensors are used to measure Parkinson's disease (PD) motor symptoms and record their fluctuations. Objective The purpose of this work is to advance our understanding of how to quantitatively describe PD motor symptoms. [5] The particular goals are to position sensors in the most practical location to monitor bradykinesia and rigidity associated with Parkinson's disease (PD) and to find objective indexes that can distinguish between patients with PD who are in OFF or ON motor status and healthy individuals (HSs). Methods Thirteen age-matched HSs and fourteen PD patients (H&Y stages 1-2.5) were included. As motion tracking systems, five magneto-inertial wearable sensors that were attached to the index finger, thumb, metacarpus, wrist, and arm were used. Both the dominant hand of HS patients and the arm of PD patients with the most damage received sensors.
The stiffness (task 22), finger tapping (task 23), and prone-supination motions of the hands (task 24) of the three UPDRS part III tasks were assessed. The three tasks were evaluated by a movement disorders specialist using the UPDRS part III scoring scheme. Different kinematic indexes from sensors were extracted in order to define each activity. Results The following four kinematic indexes were taken: smoothness, total power, total time, and fatigability. The previous three well-described PD OFF/ON motor statuses were recorded using an index finger sensor during finger-tapping tasks. Wrist sensor was able to distinguish PD OFF/ON motor status during prone-supination task
III. METHODOLOGY
A. Proposed System
In proposed system, we use implement machine learning algorithms to detect Parkinson’s such as K-nearest neighbours, Support Vector Machines, Decision Trees, and Logistic Regression (KNN), Stochastic Gradient Descent (SGD) and Gaussian Naive Bayes (GNB) are executed in these modalities with their respective datasets. Furthermore, ensemble approaches such as Random Forest Classifier (RF), Adaptive Boosting (AB) and Hard Voting (HV) are implemented.
B. Support Vector Machine
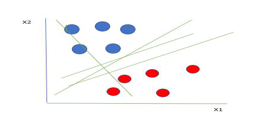
Support Vector Machine (SVM) is a supervised machine learning algorithm used for both classification and regression. Though we say regression problems as well its best suited for classification. The objective of SVM algorithm is to find a hyper plane in an N-dimensional space that distinctly classifies the data points. The dimension of the hyper plane depends upon the number of features. If the number of input features is two, then the hyper plane is just a line. If the number of input features is three, then the hyper plane becomes a 2-D plane. It becomes difficult to imagine when the number of features exceeds three.
Let’s consider two independent variables x1, x2 and one dependent variable which is either a blue circle or a red circle. Types of SVM.

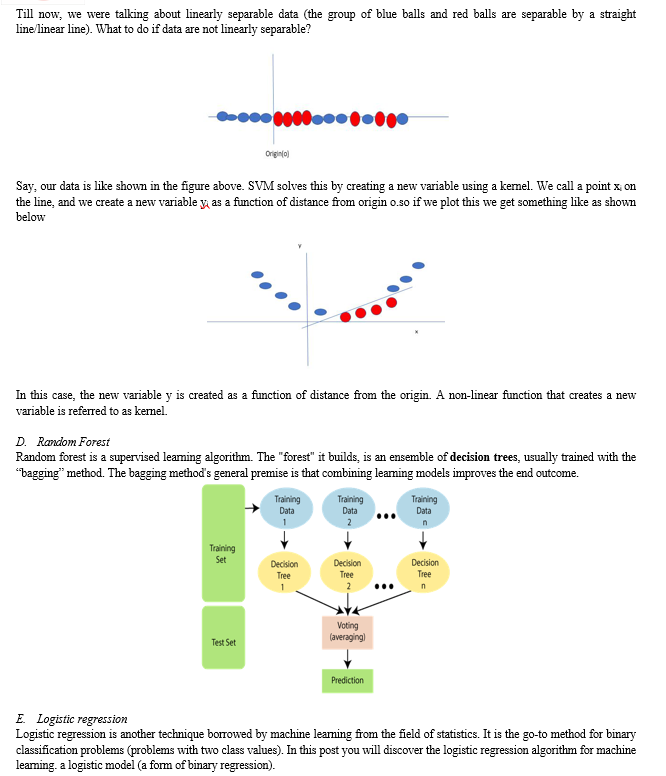
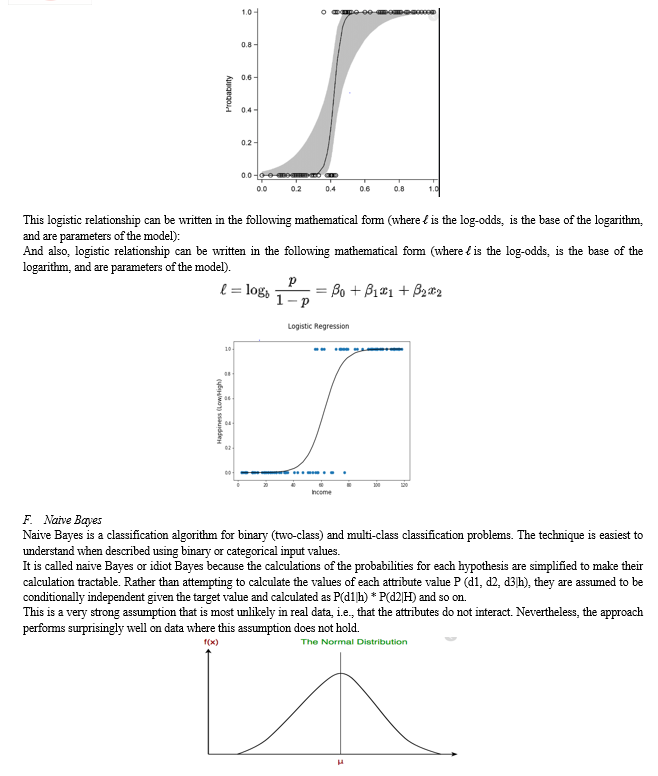

- Root Node: It represents the entire population or sample and this further gets divided into two or more homogeneous sets.
- Splitting: It is a process of dividing a node into two or more sub-nodes.
- Decision Node: When a sub-node splits into further sub-nodes, then it is called the decision node.
- Leaf / Terminal Node: Nodes do not split is called Leaf or Terminal node.
- Pruning: When we remove sub-nodes of a decision node, this process is called pruning. You can say the opposite process of splitting.
- Branch / Sub-Tree: A subsection of the entire tree is called branch or sub-tree.
Conclusion
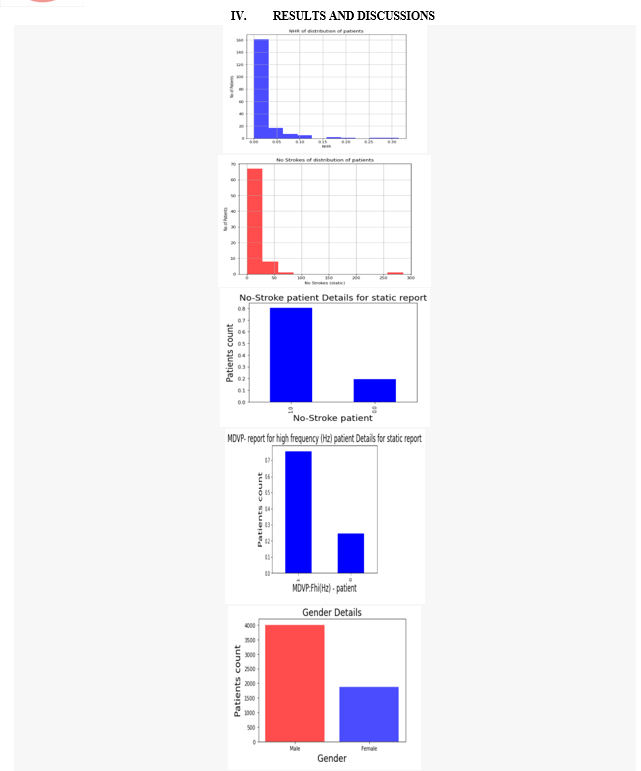
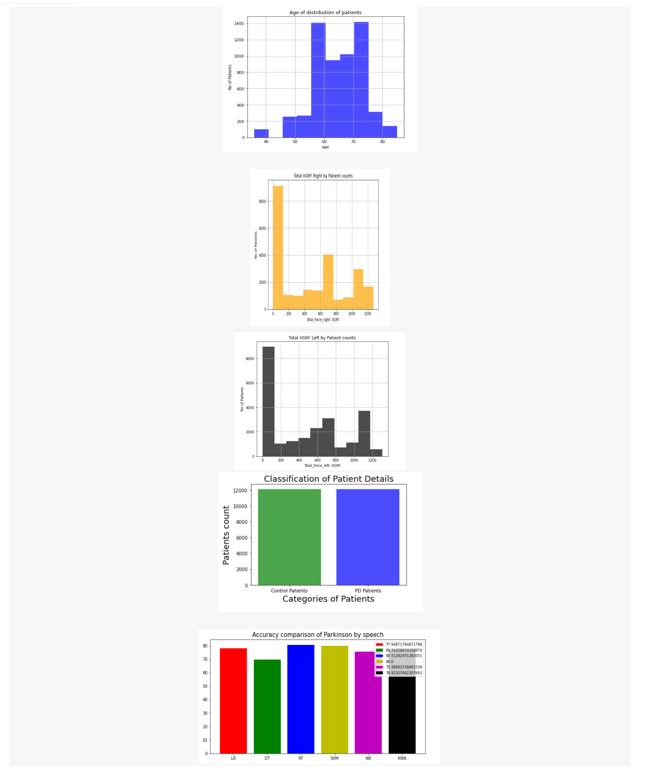
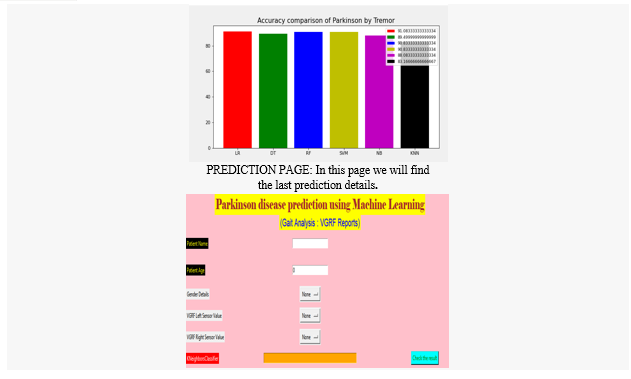
Artificial Intelligence and medical sciences have developed a relationship that helps to cure pervasive diseases like PD. various symptoms like Bradykinesia, Tremor at rest, Rigidity and Voice Impairment can be detected for early detection of PD. There is no definite medical procedure/diagnosis to cure Parkinsonism of a person, which even applies to bioinformatics. But strong tools like Machine Learning have abridged the process of detecting PD by making it economically viable and effective. Based on the research discussed in this paper, machine learning can assist doctors in detecting PD. Simple electronic devices, like a mobile phone for voice recording, using software like happy for detecting slowness in movement, and many more can be utilized for detection. According to the results shown in section V, the detection of bradykinesia and tremor leads to the concrete results for the early detection of this disease. Moreover, noticed the accuracy of detection could be increased in two ways, by implementing ensemble approaches like bagging, boosting, voting, and by increasing the size of the dataset.
References
[1] D. B. Calne “Is idiopathic parkinsonism the consequence of an event or a process” Neurology, Vol. 44, no. 15, pp. 5–5, 1994. [2] J. Parkinson “An Essay on Shaking Palsy. London: Whittingham and Rowland Printing 1817. [3] Surathi, P., Jhunjhunwala, K., Yadav, R., Pal, P. KAnnals of Indian Academy of Neurology, 19(1), 9–20, \"Research in Parkinson\'s disease: A review,\" 2016. doi:10.4103/0972-2327.167713. [4] Muthane UB, Chickabasaviah YT, Henderson J, Kingsbury AE, Kilford L, Shankar SK, et al. Numbers of melanoid nigral neurons in Nigerian and British people” Mov Disord. 2006;21:123941. [5] Baldereschi M, Di Carlo A, Rocca WA, Vanni P, Maggi S, Perissinotto E, et al. “Parkinson’s disease and parkinsonism in a longitudinal study: Two-fold higher incidence in men. ILSA Working Group. Italian Lon- gitudinal Study on Aging.” Neurology. 2000;55:135863. [6] Steven T. DeKosky, Kenneth Marek “Looking Backward to Move For- ward: Early Detection of Neurodegenerative Disorders” The American Association for the Advancement of Science Vol. 302, Issue 5646, pp. 830-834 DOI: 10.1126/science.1090349, 2003. [7] Farhad Soleimanian Gharehchopogh, Peyman Mohammadi and Parvin Hakimi “Application of Decision Tree Algorithm for Data Mining in Healthcare Operations: A Case Study.” 2012\'s International Journal of Computer Applications, Volume 52, Number 6, Pages 21–26. [8] A. Schrag, C. D. Good, K. Miszkiel, H. R. Morris, C. J. Mathias, A. J. Lees, and N. P. Quinn “Differentiation of atypical parkinsonian syndromes with routine MRI.” Neurology, Vol. 54, pp. 697–702, 2000. [9] R. Angel, W. Alston, and J. R. Higgins. “Control of movement in Parkinson’s disease.” Brain, Vol. 93, no. 1, pp. 1–14, 1970. [10] S. L. Wu, R. M. Liscic, S. Kim, S. Sorbi, and Y. H. Yang “Nonmotor symptoms of Parkinson’s disease.” Parkinson’s Dis., 2017. DOI:10.1155/2017/4382518, 2017. [11] T. Yousaf, H. Wilson, and M. Politis “Imaging the nonmotor symptoms in Parkinson’s disease.” Int. Rev. Neurobiol., Vol. 133, pp. 179–257, 2017. [12] C. G. Goetz, et al. “Movement disorder society-sponsored revision of the unified Parkinson’s disease rating scale (MDSUPDRS): Scale presentation and clinimetric testing results.” Mov. Disord., Vol. 23, no. 15, pp. 2129–70, 2008. [13] B. Post, M. P. Merkus, R. M. de Bie, R. J. de Haan, and J. D. Speelman “Unified Parkinson’s disease rating scale motor examination: Are ratings of nurses, residents in neurology, and movement disorders specialists interchangeable?” Movement Dis., Vol. 20, pp. 1577–84, 2005. [14] R.L. Albin, A.B. Young and J.B. Penney, “The functional anatomy of basal ganglia disorders.” Trends Neurosci 1989; 12: 366–75, 1989. [15] M.R. DeLong “Primate models of movement disorders of basal ganglia origin.” Trends Neurosci. 1990 Jul;13(7):281-5, 1990.
Copyright
Copyright © 2023 Nandhiniumesh K. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET48979
Publish Date : 2023-02-02
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

