Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Artificial Intelligence in 3D Bio Printing
Authors: Pramod K, Anjima AP
DOI Link: https://doi.org/10.22214/ijraset.2022.44161
Certificate: View Certificate
Abstract
3D printing in full three-dimensional printing also known as additive manufacturing is the process of making three-dimensional solid objects from a digital file. The creation of a 3D printed object is achieved using additive processes. In an additive process, an object is created by laying down successive layers of material until the object is created. Each of these layers can be seen as a thinly sliced cross-section of the object.3D printing is the opposite of subtractive manufacturing which is cutting out / hollowing out a piece of metal or plastic with for instance a milling machine.3D printing enables you to produce complex shapes using less material than traditional manufacturing methods. As a result, 3D printing creates less material wastage. “3D Bio printing” or “bio printing” is a form of additive manufacturing that uses cells and biomaterials instead of traditional metals and plastics to create 3D constructs that are functional 3D tissues. But unlike 3D printing, bio printers print with cells and biomaterials, creating organ like structures that let living cells multiply. These biomaterials are called bio-inks, and they mimic the composition of our tissues. Bio printing can be applied to a variety of areas including but not limited to regenerative medicine, drug discovery and development, and 3D cell culture.
Introduction
I. INTRODUCTION
Bio 3D printing or simply bioprinting technology is the name for Additive Manufacturing used in the medical area. It is made of natural materials rather than plastics or metal, thus the "bio" portion of the name. Bioprinting is 3D printing with cells rather than plastics or metals.
3D printed organs, for example, can be made with precision tailored tissue structures.
Layers of biomaterials or engineering tissue are built up to form a three-dimensional structure model of three dimensions The layers can be created in a variety of ways, some of which are described below .Others use liquid and solidify it, similar to the most prevalent 3D printing process, FDM just as resin 3D printing does, UV light is used. Three-dimensional (3D) printing technologies have recently become commonplace in the industry and our everyday lives .To define 3D printing at the biomedical level, the term 3D bioprinting was coined.
Machine learning is being utilised to improve 3D printing processes such as process optimization, dimensional accuracy analysis, manufacturing fault detection, and material property prediction. However, machine learning has been used in 3D bioprinting techniques in only a few research .The demand for organ transplants has increased, while the amount of available organs for transplantation has remained constant around the world. 3D bioprinting of tissue and organs is a novel topic in tissue engineering and regenerative medicine that aims to alleviate the global lack of organ donors that patients in desperate need of a transplant face. A bioprinter uses biomaterials and embedded cells to create complex and functioning three-dimensional (3D) organ or tissue constructs for regenerative medicine. Furthermore, traditional surgical 3D models are built of stiff plastic or rubber, making it impossible for surgeons to interact with real organ or tissue-like replicas.
Finding appropriate biomaterials and printing technologies can thus speed up the printing of complex organ structures as well as the construction of realistic models to fine-tune surgical techniques and tools before surgery .Furthermore, printing parameters in the bioprinting process (such as printing speed, dispensing pressure, and nozzle diameter) should be tuned.
As a result, machine learning (ML) technology can be an effective tool for optimising the many bioprinting parameters. It is, in essence, a biomedical application of rapid prototyping or additive manufacturing technology .Additive manufacturing allows a digital model of an object to be transformed into physical reality. As a result, information technology plays an important role in organ printing.3D printing combined with artificial intelligence is allowing for new and intriguing additive manufacturing applications.
A. Artificial Intelligence
Machine intelligence, often known as artificial intelligence, is intelligence demonstrated by machines. Machines have the ability to learn and acquire information. Advanced tasks can be performed on these devices as a result of this. Artificial intelligence-based machines can imitate intelligent human behaviour. AI has the potential to greatly improve 3D printing.
B. Using AI in 3D Printing
Machine learning and neural networks are frequently used to describe artificial intelligence.
The notion is that, using data and prior experiences, a machine can solve a problem without the need for human participation. When integrated with 3d technology, it has the potential to improve the performance of a 3d printer by lowering the chance of error.
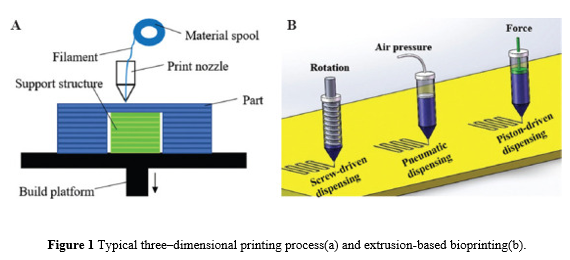
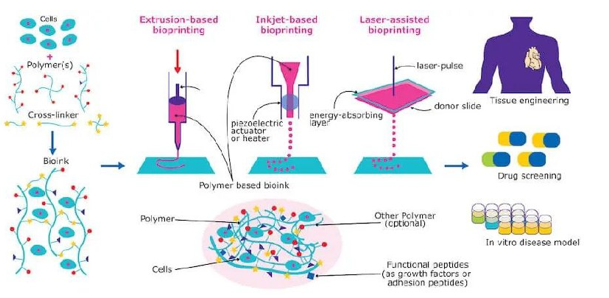
The 3D printing method works from the bottom up, layer by layer, from the bottom to the top of a product. It's an additive method that gradually adds materials until the entire component is built.A typical extrusion-based 3D printing technique for manufacturing a component is shown in Figure a. 3D bioprinting is a method of fabricating biomedical parts made up of biomaterials, growth factors, and cells that closely resemble real tissue features using 3D printing-like technologies.The fabrication process for 3D bioprinting is similar to that of 3D printing, which deposits materials layer by layer. Bio-inks are utilised in 3D bioprinting rather than polymer, metal, or ceramic, which are used in typical 3D printing techniques.
There are five major bioprinting processes now available: stereolithography, inkjet, extrusion, and laser-assisted bioprinting.
Extrusion-based bioprinting is the most popular method among them. The standard three extrusion-based bioprinting techniques are shown in Figure b. The type of force used can be air pressure (pneumatic dispensing), direct force (piston-driven dispensing), or rotation (screw-driven dispensing).
Machine learning is a new technology that can help systems run more efficiently by making better use of products, materials, and services .Machine learning has the potential to reduce fabrication time, lower costs, and improve quality in 3D printing processes. Machine learning has already been used to optimise processes, analyse dimensional accuracy, detect manufacturing defects, and predict material properties in the literature. In order to demonstrate its influence on 3D bioprinting, related machine learning employed in 3D printing will be briefly discussed.
C. Machine Learning
One of today's fastest-growing technology sectors is machine learning. It's a subset of AI that focuses on system design. Machine learning enables these programmed systems to learn and make predictions based on prior experience, or data in machine words.
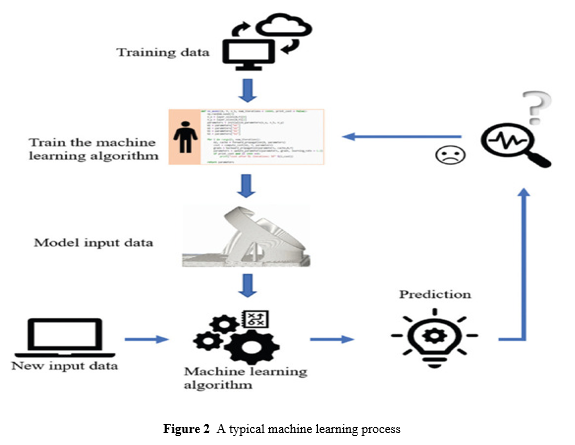
The machine learning process is depicted in Figure 2. The algorithm must first train on the data in the training set.
The algorithm's parameters will be improved during the training phase, and a machine learning model will be generated.It may then forecast the results with new input data using the modified machine learning settings.
The training data in supervised learning are a collection of x and y form pairs, and the goal is to get the expected result y in response to a query x. In machine learning, x, y can be more than one element that will be written as a vector.Supervised learning is being utilised in email spam classification, medical diagnosis systems for patients, and image face recognition.Unsupervised learning differs from supervised learning in that the input data is unlabeled. Algorithms will learn and extract the features of the incoming data automatically, and then divide them into separate clusters. Unsupervised learning seeks to model the input data's underlying distribution or structure in order to learn more about it.Unsupervised learning is being used in market segmentation to target the right customers, document clustering based on content, picture division, and anomaly or fraud detection in banking.
The information from the training data supplied into algorithms is midway between unsupervised and supervised learning in reinforcement learning. In supervised learning, the training data are supposed to provide simply an indicator of whether an action is proper or not, rather than specifying the correct output for a given input.
Reinforcement learning is currently being applied in aeroplane control, robot motion control, traffic light control, online system configuration, and video games (e.g., AlphaGo).
In addition to these three learning paradigms, other machine learning approaches such as semi-supervised learning have been created.
Each machine learning approach has a number of algorithms accessible.
Perspective on using Machine Learning in Bio Printing
D. Process Optimization
Aoyagi et al proposed utilising a support vector machine to generate a process map for 3D printing in typical 3D printing methods.This approach can forecast a process state that is suitable for producing a low-pore-density product.Menon et al. employed hierarchical machine learning to optimise material, process factors, and formulation for freeform reversible embedding 3D printing of silicone elastomer.In a vat photopolymerization process (Continuous Liquid Interface Production), He et al studied employing multiple machine learning algorithms for modelling and forecasting the correct printing speed.The Siamese network model had the highest accuracy in their research.In addition, a conceptual framework for combining mathematical modelling and machine learning to evaluate and optimize parameters in Powder Bed Fusion processes was proposed by Baturynska et al.
Machine learning can also be utilised to improve the fabrication process in 3D bioprinting, such as anticipating process conditions and optimising process parameters.Via extrusion-based bioprinting, for example, it is now possible to create stable organoids with low-concentration gelatin-methacryloyl using electrostatic attraction.
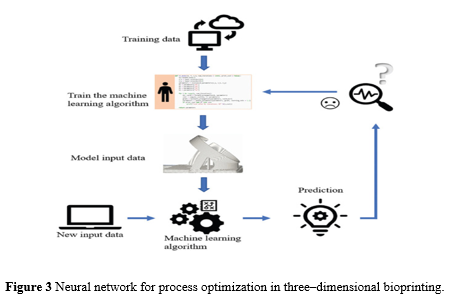
Figure 3 depicts an example of neural networks being used to improve the bioprinting process.
The variables are the factors that influence the objective outcomes (e.g., cell damage, cost, and time). Voltage, gas, nozzle size, pressure, and other variables can be supplied into the neural network for training in this situation.To tune machine learning parameters, corresponding outputs (cell damage, cost, time, etc.) must be provided.Once the method is complete, additional input data can be utilised to evaluate performance.
E. Manufacturing Defect Detection
They employed computer vision techniques and unsupervised machine learning to identify in situ melt pool characteristics indicative of fault generation in a laser powder bed fusion process in the traditional 3D printing process. Caggiano and colleagues created a machine learning method for detecting metal material flaws in Selective Laser Melting procedures in real time. For fault detection, images from the layer-by-layer manufacturing process are analysed using a bi-stream deep CNN. A CNN technique for detecting porosity in laser additive manufacturing (AM) processes was described by Zhang et al. For in-process sensing, the melt-pool data were obtained using a high-speed digital camera.The data was then processed using their newly constructed neural network.
Machine learning can also be utilised in 3D bioprinting to detect faults such as incorrectly positioned cells, curved layers, and microstructure errors in a fabrication process that can monitor the entire bioprinting process. In 3D bioprinting, Figure 4 provides an example of employing CNN to discover flaws, errors, or defects. Images captured by high-resolution cameras during the bioprinting process can be used as input data. The data is then examined by the CNN model in order to forecast a defect or other goals. In the traditional 3D printing process, Francis and Bian[22] developed a deep learning method that can
accurately predict the distortion of parts in laser-based AM. Similarly, Khanzadeh et al.[23] proposed an unsupervised machine learning approach (self-organizing map) to quantify the geometric deviations of additively manufactured parts in fused ?lament fabrication processes. In addition, Zhu et al.[24] also developed a strategy coupled with machine learning to address the modeling of shape deviations in AM. Tootooni et al.[25] compared six machine learning techniques (sparse representation, k-nearest neighbors, neural network, naïve Bayes, support vector machine, and decision tree) with regard to the accuracy of predicting dimensional variation in fused deposition modeling (FDM) printed parts. Based on their study, the sparse representation approach has the best classi?cation performance. In 3D bioprinting, similarly, machine learning can be used for analyzing the accuracy of fabricated bio-parts. For example, the tissue-engineered scaffolds are generally very complex because they supporting cell growth in an expected way to achieve corresponding functions. If the Dimensional accuracy analysisIn the traditional 3D printing process, Francis and Bian[22] developed a deep learning method that can accurately predict the distortion of parts in laser- based AM. Similarly, Khanzadeh et al.[23] proposed an unsupervised machine learning approach (self-organizing map) to quantify the geometric deviations of additively manufactured parts in fused ?lament fabrication processes. In addition, Zhu et al.[24] also developed a strategy coupled with machine learning to address the modeling of shape deviations in AM. Tootooni et al.[25] compared six machine learning techniques (sparse representation, k-nearest neighbors, neural network, naïve Bayes, support vector machine, and decision tree) with regard to the accuracy
of predicting dimensional variation in fused deposition modeling (FDM) printed parts. Based on their study, the sparse representation approach has the best classification performance. In 3D bioprinting, similarly, machine learning can be used for analyzing the accuracy of fabricated bio-parts. For example, the tissue-engineered scaffolds are generally very complex because they supporting cell growth in an expected way to achieve corresponding functions. If the Dimensional accuracy analysis In the traditional 3D printing process, Francis and Bian[22] developed a deep learning method that can accurately predict the distortion of parts in laser-based AM. Similarly, Khanzadeh et al.[23] proposed an unsupervised machine learning approach (self-organizing map) to quantify the geometric deviations of additively manufactured parts in fused ?lament fabrication processes. In addition, Zhu et al.[24] also developed a strategy coupled with machine learning to address the modeling of shape deviations in AM. Tootooni et al.[25] compared six machine learning techniques (sparse representation, k-nearest neighbors, neural network, naïve Bayes, support vector machine, and decision tree) about the accuracy of predicting dimensional variation infused deposition modeling (FDM) printed parts. Based In their study, the sparse representation approach has the best classic?cation performance.In 3D bioprinting, similarly, machine learning can be used for analysing the accuracy of fabricated bio-parts. For example, the tissue-engineered scaffolds are generally very complex because they supporting cell growth in an expected way to achieve corresponding functions. If theDimensional accuracy analysis In the traditional 3D printing process, Francis and Bian[22] developed a deep learning method that can accurately predict the distortion of parts in laser-based AM. Similarly, Khanzadeh et al.[23] proposed an unsupervised machine learning approach
(self-organizing map) to quantify the geometric deviations of additively manufactured parts in fused ?lament fabrication processes. In addition, Zhu et al.[24] also developed a strategy coupled with machine learning to address the modeling of shape deviations in AM. Tootooni et al.[25] compared six machine learning techniques (sparse representation, k-nearest neighbors, neural network, naïve Bayes, support vector machine, and decision tree) about the accuracy of predicting dimensional variation infused deposition modeling (FDM) printed parts. Based In their study, the sparse representation approach has the best classic?cation performance.In 3D bioprinting, similarly, machine learning can be used for analyzing the accuracy of fabricated bio-parts. For example, the tissue-engineered scaffolds are generally very complex becausethey supporting cell growth in an expected way to achieve corresponding functions.
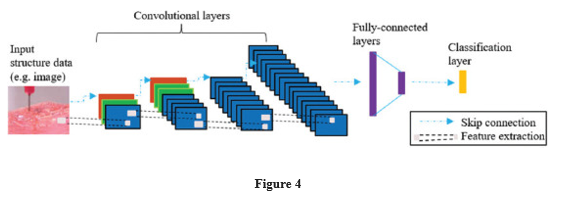
An example of using machine learning (convolutional neural network) in three-dimensional bioprinting.
F. Dimensional Accuracy Analysis
Francis and Bian devised a deep learning method in the traditional 3D printing process that can accurately anticipate part distortion in laser-based AM. Khanzadeh et al. developed a self-organizing map approach to quantifying unsupervised machine learning.With fused filament fabrication, the geometric aberrations of additively created parts processes. Furthermore, Zhu et al. devised an approach based on machine learning to Tootooni et al. compared six machine models to model form deviations in AM approaches for learning (sparse representation, k-nearest neighbours, neural network, nave Bayes, etc.)concerning the accuracy of forecasting dimensionality (support vector machine and decision tree)Fused deposition modelling (FDM) printed items vary. According to their research, the scarce the classification performance of the representation technique is the best.Machine learning can also be utilised in 3D bioprinting to assess the correctness of manufactured bio-parts. For example, the tissue-engineered scaffolds are often quite complicated because they expectedly enable cell proliferation to attain matching roles. If machine learning can analyse accuracy in advance, the final produced bio-parts can be guaranteed to be of good quality. As seen in Figure 4, the technique is similar, but the input data is different.
G. Material Property Design or Prediction
Gu et al. developed a machine learning-enabled approach for designing hierarchical composites for manufacturing, trained with a library of adequate structures from finite element analysis, as an alternative to traditional 3D printing. Hamel et al. demonstrated a machine learning method for designing active composite structures with target shapeshifting reactions that can be printed in 4D. Li et al. proposed a machine learning-based predictive modelling system that can accurately forecast the surface roughness of FDM printed objects. Jiang et al. are now analysing and predicting printed bridge length in FDM processes using a backpropagation neural network.
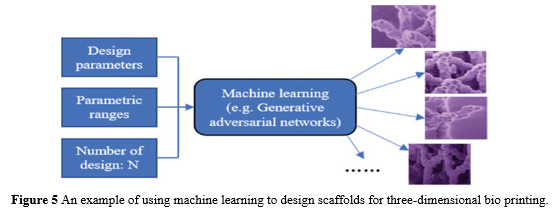
Machine learning can also be utilised in the 3D bioprinting process to develop or assess material qualities. Tissue-engineered scaffolds, for example, are critical in 3D bioprinting, and their shapes must be carefully tailored for effective cell growth and function. For example, if we wish to print an organ, the scaffolds should have a specific structure for the successful growth of cells to produce a functional organ. If machine learning can be utilised to build these qualified scaffolds with various functions, the time it takes to develop biomedical or tissue engineering combined with bioprinting will be significantly shortened in the future. Most importantly, machine learning might be able to develop some unanticipated novel structures that assist cell growth and functionalization better than ever before.
Figure 5 illustrates how machine learning (e.g., the generative design method) can be used to produce a large number of qualified scaffolds for selection or testing in 3D bioprinting. The number of predicted generated scaffolds can then be set when the design variables and their respective range values have been provided. The scaffolds can then be tested for various objectives using the many possibilities given by machine learning
II. LITERATURE SURVEY
A. Deep Learning for Fabrication and Maturation of 3D Bio-Printed Tissues and Organs
Bioprinting is a relatively new and promising tissue engineering technology for addressing the problem of organ transplant donor shortages. It's a cutting-edge bio manufacturing technique that allows for the layer-by-layer printing of biomaterials, living cells, and growth factors to create 3D tissue-engineered constructions. From medical image processing through printing parameter optimization and modifications during post-printing tissue maturation, the current procedure entails a plethora of manufacturing difficulties.Deep learning is a sophisticated machine learning technology that has accelerated the development of picture and language applications during the last decade. We highlight the integration of deep learning into 3D bioprinting technology and the execution of practical guidelines in this viewpoint paper.We look at how deep learning could be used in several 3D bioprinting processes such image processing and segmentation, printing parameter optimization and in-situ correction, and tissue maturation refinement.Finally, we examine the implications of deep learning for 3D bioprinting uptake and regulation. The synergistic interplay of biology, materials, and deep learning-enabled computational design will eventually enable the manufacture of biomimetic patient-specific tissues/organs, paving the way for 3D bioprinting of tissues/organs.
B. Artificial Intelligence in 3D Printing: A Revolution in Health Care
Artificial Intelligence (AI) is being catapulted out of research labs and into our everyday lives in the digital age, thanks to continually evolving medical technology. Emerging AI developments benefit the healthcare system by reducing the amount of manual labour performed by humans. As a result, AI is a game-changer in health care since it can improve performance by minimising the chance of error and facilitating automated production when integrated with three-dimensional (3D) printing technology. 3D printing has quickly become a necessary and potentially transformative tool to swiftly alter health care. One of the chapters' goals is to provide an overview of core AI ideas such as machine learning, the internet of things, cloud computing, and deep learning.The additional dimensions will also describe 3D printing types. A picture of stereolithography (SLA), which is the oldest and most dependable technology in 3D printing is also described. The current chapter will also discuss key healthcare trends that are affecting the 3D printing industry, such as using 3D printing to combat the COVID-19 pandemic, regenerative medicine, eliminating the need for animal testing to confirm efficacy and safety before human testing, and personalised medicines. The final portion covers significant uses, problems, and the future of 3D printing, allowing readers to see how this developing technology is quickly becoming society's most useful tool and how they can apply the information to their own studies.
C Coupling Machine Learning with 3D Bioprinting to Fast-Track Optimisation of Extrusion Printing
Three-dimensional bioprinting, a paradigm change in tissue engineering, has promise for regenerative medicine and disease modelling. To create cell-laden 3D constructs, 3D scaffolds are fabricated for subsequent cell seeding or incorporated directly into the bio-ink. The optimization of the printing process is influenced by a variety of elements such as bio-ink characteristics, printing parameters, and post-print curing. Although there have been many studies on qualitative evaluations of printability, there are few studies on quantitative ways to assess printability. As a result, this research investigates machine learning as a unique technique for quantitatively evaluating printability and expediting extrusion printing optimization in order to achieve a repeatable 3D scaffold.A score system was developed to measure the printability of gelatine methacryloyl (GelMA) and hyaluronic acid methacrylate (HAMA) bioinks, and Bayesian Optimisation, a machine learning method, was used to optimise 3D plotting. The performance of two basic criteria encountered in the printing process have been added into the scoring metric: filament production of the bio-ink and layer stacking of the 3D scaffold. In 19, 4, and 47 experiments, the optimal print parameters for GelMA containing inks with varying concentrations (10 percent, 7.5 percent, and 5 percent (w/v)) were obtained, respectively. 10:2 percent, 7.5:2 percent, and 5:2 percent (w/v) required 32, 25, and 32 tests, respectively. In comparison to the 6000 to 10,000 possible possibilities in the Bayesian approach, this number of experiments is substantially reduced.Hence, this study will be a stepping-stone to unraveling the benefits of machine learning in this rapidly developing area of 3D bioprinting.
III. DATASET
In the instance of bioprinting, machine learning has previously been applied to optimise the printability of bioinks, reducing the number of experiments from thousands to hundreds[10]. Moreover, as recently described in Zhang et al. and Schwab et al., many mathematical models on bioink printability have been established. These mathematical models can be used to create virtual bioprinting. In vitro studies have also employed ML models to identify cell signature genes from complex gene expression profiles among various cell types. Virtual histological staining is another in vitro example, which avoids the time-consuming and tedious procedure of tissue preparation. Deep learning was utilised to transform autofluorescence photos of tissue into images that were equivalent to histologically stained tissue, and the researchers were able to mix several stains by assigning each stain at the pixel level. Furthermore, mathematical and machine learning models, which aid in the understanding of biological systems and the extraction of novel biological knowledge from large datasets, are predicted to bring tissue engineering much closer to clinical reality.
Indeed, numerous experimental data and materials directly connected to bioprinting have been generated over the years, making bioprinting a potentially data-driven research, but there is now only a small database dedicated to bioprinting. We expect to see additional improvements in this area in the future, as databases for 3D printing develop. It's feasible that using only the present literature and ignoring expert comments, it'll be possible to forecast fresh bioprinting discoveries.The use of machine learning in bioprinting and biofabrication will result in significant changes, and bioprinting will become a part of the digital economy and information technology.
In recent decades, additive manufacturing technology have advanced, particularly when utilised to print biofunctional structures like scaffolds and vessels with living cells for tissue engineering applications. Maintaining the biocompatibility and structural integrity required for synthetic tissue constructions requires part quality and reliability. As a result, it's vital to look for abnormalities in the 3D-bioprinting process that could result in a mismatch between the anticipated designs and the produced shapes. However, precisely and efficiently detecting flaws inside sometimes transparent bioprinted and complicated printing features is difficult. In this article, an anomaly detection method for transparent hydrogel-based bioprinted materials is developed using layer-by-layer sensor pictures and machine learning algorithms to recognise and classify flaws.On extracted small image patches, high anomaly detection accuracy is achieved using convolutional neural network approaches as well as advanced image processing and augmentation techniques. The category of infill pattern and position information on the picture patches may be precisely determined, in addition to the prediction of various abnormalities. Real-time autonomous correction of process parameters can be performed utilising our detection system to categorise and identify printing anomalies, resulting in high-quality tissue constructs in 3D-bioprinting processes.
Bioink material content, solvent used, polymer crosslinking information, printing parameters, cell viability, and printability results were gathered from 75 EBB articles during the previous 13 years and entered into the database. Data utilised for bioprinting ML model training and testing has only been acquired from and applied within group, as shown in prior works. To our knowledge, this is the first compilation of experimental data and parameters provided from several bioprinting laboratories for ML applications. There are 617 distinct cases of cell viability and 339 distinct instances of printability in the database. Based on specific combinations of material, biological, and printer factors, we investigated the capacity of machine learning (ML) regression and classification approaches to accurately and precisely predict cell viability and printability outcomes.
IV. METHODOLOGY
Despite the similarities between a 3D bioprinter and a 3D printer, a bioprinter must meet certain requirements ensure that live cells survive This implies that it must possess specific distinguishing qualities such as the capacity to print in moderate temperatures.The majority of bioprinters work by printing a bio-ink, which is a type of 3D gel.gelatine, cells, and a printed biomaterial Hydrogel biomaterials are similar to bio inkswhich scientists utilise to make non-printed tissue structures, usually in microplates but not always.One significant difference is that bio inks can be extruded or deposited from a printer.
A. Bio ink
Bio-ink is a printable material made up of living cells from the patient's body embedded in a bio-compatible polymer that serves as the 3D molecular scaffold in a 3D bio-printer. After that, the bio-ink is 3D printed into a functional tissue construct that can be used for transplantation, drug testing, or disease modelling. High-resolution 3D printing is simple using Bio-ink. During the printing process, the cell-friendly bio-ink incorporates living cells into microstructures. This helps the printed tissues' bio functioning.
B. The Bioprinter
The 3D bioprinter works similarly to a 3D printer, but with a slight difference. The bioprinter works with cells and biomaterials as starting materials, using 3D digital files as a template. The tissues are also adequately vascularized for the exchange of blood, oxygen, and other nutrients thanks to 3D bioprinting.
The 3D bio-printing process involves three stages.
C. The Preparatory Stage
To print a tissue, you must first create a digital design in a 3D modelling programme. The 3D design-construct is derived directly from the patient using biomedical diagnostic imaging techniques such as computed tomography (CT), 3D laser scanning, or magnetic resonance imaging (MRI) scans.Then, using computer-aided design and computer-aided manufacturing (CAD/CAM) graphic software, anatomically correct 3D models are created. A slicer is used to load the 3D model. The slicer is a type of computer programme that has been carefully optimised. It analyses the model geometry to generate a succession of 3D stacks that will form the printed tissue's shape. After the model has been sliced, the Gcodes are saved and transmitted to a 3D bioprinter for printing. It's difficult to segment tissue images at a single cell level and reconstruct them into a 3D tissue model with cross-scale cellular resolution and tissue attributes in pre-processing. Because of the multiscale difficulties in representing biological tissue models, machine learning is required. In addition, ML can also help predict the compatibility of dissimilar materials used in bioprinting
D. The Bioprinting Stage
G-codes are a set of instructions that tell the bio-printer how to build the 3D tissue layer by layer. The research scientist chooses the bio-ink, mixes it with the tissue-specific cell, packs the cell-laden bio-ink into a cartridge, and inserts it into the printer, depending on the tissue to be produced. The printer determines the pace for the bioprinting step once it gets data on control settings from the Gcodes. Once all of the G-code commands
E. The Post Bioprinting Stage
Cross-linking stabilises the manufactured constructions after bioprinting. Depending on the make-up, the constructions can be treated with an ionic solution or UV light during the crosslinking process. The goal of 3D bioprinting will be thwarted if there are heterogeneous tissues with well-defined sections that lack biological functions. The structure of the bio-print is thus produced in specific culture mediums under controlled conditions for several days to reproduce the biological processes found in living cells. The printed cells develop interconnected networks during this maturation phase, resulting in tissues with physiological potential. This step is crucial because it aids the tissue construct in achieving the proper bio-physiological function. The tissue is ready to utilise once it has been created.When the number of variable parameters exceeds a specific amount, such as 10, it is nearly difficult to run wet experiments in processing and post-processing.Because of the multiparameter complexity of discovering the best bioprinting procedure, machine learning is required. Here, it is hoped that machine learning combined with big data can tackle multiscale and multiparameter problems, transforming current 3D bioprinting into future 3D bioprinting that is "heavily virtual" in nature.
F. 3D Bio printing Technologies
- Extrusion bio printing:Extrusion-based bioprinting is the most similar type of 3D bioprinting to FDM3D printing. A computer-controlled print head is used to extrude a highly viscous bio-ink onto a surface like a petri dish, layer by layer. Air pressure, pistons, or a reciprocating screw can all be used to extrude material. Because of its versatility and cost, extrusion-based bioprinting (also known as direct ink writing), which is derived from inkjet printing, is the most extensively utilised approach to 3D bioprinting. Extrusion-based bioprinting produces continuous filaments instead of a single droplet due to the continuous extrusion force. This method can be used to print a variety of biomaterial viscosities as well as varied cell concentrations. As a result, extrusion-based bioprinting is preferred by researchers for creating tissue architectures with sufficient mechanical properties.In addition, coaxial and multi material bioprinting can also be perfectly compatible with extrusion-based bioprinting for various kinds of applications.
- Inkjet bio printing: Inkjet 3D bioprinting, also known as drop-on-demand bioprinting, is a quick and precise bioprinting process that uses droplets of bio-ink to print on a surface. While inkjet bioprinting enables for precise cell control, it is less effective for 3D printing scaffolds due to the bio-inks' low viscosity requirements.
- Laser-assisted bio printing: Laser-assisted 3D bioprinters, unlike extrusion and inkjet 3D bioprinters, do not use a nozzle to dispense bio-ink. Instead, UV light is focused on a vat of photosensitive bioink, which hardens when exposed to light. The bioprinter, like other SLA printers, includes a moving platform that allows for the printing of additional layers.
- Misc. bio printing technologies: Other types of 3D bioprinting are more difficult to categorise. For example, instead of employing a biomaterial scaffold, several 3D bioprinting businesses have developed methods that 3D print cells onto a needle array. Clusters of pure cells, not combined with other biomaterials, are skewered onto upright needles to produce 3D tissue structures using this technique.
G. Where does AI come In For 3D Bioprinting?
It is undeniable that combining two disruptive technologies will result in an entirely new world of possibilities. 3D has made significant progress in making functioning bioproducts available, as well as in developing novel assay models for predicting medication effects in humans. The challenges in the various stages of the 3D bioprinting workflow, on the other hand, present exciting AI prospects.To begin with, the biodegradable material employed to keep printed tissue in shape can trigger an immunological response and cause cell toxicity. Fortunately, artificial immune systems that imitate human immune responses exist. When implanted in the human body, artificial immune systems can detect any aberration or intrusive immunological response the tissue may provoke.AI can also forecast the outcomes of many 3D print applications before they are used, which will cut clinical trial failure rates.Still, by analysing data and finding trends at every stage of the 3D bioprinting process, AI can make recommendations on the best possible printing parameters for producing tissue that is biocompatible with a patient's physiological composition
Conclusion
Three-dimensional bioprinting has made remarkable progress in proving the feasibility of organ transplants and improving patients’ treatment methods. Personal genetic information varies; hence, understanding personalized medicine is essential. Personalized medicine is a promising and emerging medical practice that actively utilizes an individual’s genetic profile to guide disease prevention, diagnosis, and treatment decisions. Here, 3D bioprinting can create complete replacements of the tissue/organs for transplant surgery. Furthermore, 3D printed organs/tissues using individual decellularized extracellular matrix and stem cells could be used for patient-specific transplantation. However, fabricating fully functional tissues/organs still has many challenges due to limitations such as incarnating vasculatures for the solid organs (e.g., liver, pancreas, spleen, and adrenal glands). Moreover, novel techniques are demanded to process and understand these complex data collected from personalized medicine diagnostic approaches. Fortunately, computer science has developed swiftly to progress techniques that enable the storage, processing, and analysis of these complicated datasets, a feat that traditional statistics and early computing technologies could not accomplish. In this regard, this review paper explored the contents of 3D bioprinting and ML, respectively, and examined the recent studies that ML has been applied for the development of 3D bioprinting. ML can optimize the bioprinting process and parameters to increase printing fidelity. Hence, combining 3D bioprinting with ML techniques would be a great research area to accelerate future development.
References
[1] Yu C, Jiang J. A Perspective on Using Machine Learning in 3D Bioprinting. Int J Bioprinting. 2020;6:95. https://doi.org/10.18063/ijb.v6i1.253. [PMC free article] [PubMed] [Google Scholar]. [2] Ruberu K, Senadeera M, Rana S, et al. Coupling Machine Learning with 3D Bioprinting to Fast Track Optimisation of Extrusion Printing. Appl Mater Today. 22:100914. https://doi.org/10.1016/j.apmt.2020.100914. [Google Scholar]. [3] Ruberu K, Senadeera M, Rana S, et al. Coupling Machine Learning with 3D Bioprinting to Fast Track Optimisation of Extrusion Printing. Appl Mater Today. 22:100914. https://doi.org/10.1016/j.apmt.2020.100914. [Google Scholar]. [4] Dernowsek JD, Rezende RA, Lopes da Silva JV. 2-17, The Role of Information Technology in the Future of 3D Biofabrication. J 3D Print Med. 1:63–74. [Google Scholar]. [5] Yu, Chunling & Jiang, Jingchao. (2020). A Perspective on Using Machine Learning in 3D Bioprinting. 6. 1-8. 10.18063/ijb.v6i1.253. [6] An J, Chua CK, Mironov V. Application of Machine Learning in 3D Bioprinting: Focus on Development of Big Data and Digital Twin. Int J Bioprint. 2021;7(1):342. Published 2021 Jan 29. doi:10.18063/ijb.v7i1.342. [7] Santoni, S., Gugliandolo, S.G., Sponchioni, M. et al. 3D bioprinting: current status and trends—a guide to the literature and industrial practice. Bio-des. Manuf. 5, 14–42 (2022). https://doi.org/10.1007/s42242-021-00165-0. [8] Shin J, Lee Y, Li Z, Hu J, Park SS, Kim K. Optimized 3D Bioprinting Technology Based on Machine Learning: A Review of Recent Trends and Advances. Micromachines. 2022; 13(3):363. https://doi.org/10.3390/mi13030363
Copyright
Copyright © 2022 Pramod K, Anjima AP. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET44161
Publish Date : 2022-06-12
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

