Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Biosynthesis, Characterisation and Thermal study of Graphine Oxide-TiO2 nanocomposite using Ocimum tenuflorum leaf extract
Authors: Manjunath Patil, Arunkumar Lagashetty
DOI Link: https://doi.org/10.22214/ijraset.2022.43814
Certificate: View Certificate
Abstract
Blend of nanomaterials and its composite utilizing plant based item incorporates the manufactured innovation and subsequently different properties and applications. The principal a piece of the work is to combine naomaterials utilizing ocimum tenuflorum plant extricate for organic decrease response in ecofriendly framework. At first graphine oxide (GO) is arranged chemicaly and TiO2 adjusted GO nanocomposite (TiO2-GO NC) was ready by bioreduction utilizing ocimum tenuflorum plant remove. The construction of as pre-arranged examples very much was portrayed utilizing X-beam diffraction (XRD) apparatus. Examining Electron Micrograph (SEM) apparatus is utilized to concentrate on the morphology of the example. Metallic perception in the example was researched by EDAX design. Shifted holding nature of the bioderived nanocomposite is concentrated on by Fourier change infrared (FT-IR) instrumentation. Warm and electrical investigations of the examples are likewise under taken to know the warm electrical way of behaving. UV absorbance investigation of the said test is additionally attempted.
Introduction
I. INTRODUCTION
Science and innovation of nanomaterials prompts the advancement of new properties and applications incorporates the materials innovation [1-2]. The future innovation nanomaterials will be worked by fitting the blocks of materials which will compel for some applications. Metal nanoparticles are bound together and have a high unambiguous surface region likewise high part of surface molecules. Physicochemical qualities of nanoparticles courses for the significance for their original techniques for amalgamation [3-4]. Plant based items remove are utilized for the arrangement nanomaterials leads exceptional properties and applications in different fields. [5-7].
Metals/metal oxides nanoparticles are ready by natural course utilizing plant extricate is creating because of its basic trial and error. Hydrocarbon energizes are staying away from in organic strategy for the materials union at nanoscale and subsequently are eco-accommodating framework. One can expect that, developing need to foster ecologically regarding blend processes without utilizing poisonous synthetic compounds [8-10]. There is a rising interest for green nanotechnology for its straightforwardness and energy base items [11-12]. Organically decrease strategy for the union of nanoparticles utilizing plant based item removes is entirely fitting technique for creation of nanoparticles [13-14].
The nanocomposites of RGO and metal oxide nanoparticle arranged by natural strategy has made a lot of interest because of its materialness in different fields [15-17]. Thsi composite innovation connected with nano measured materials improves the few applications as energy materials. Colossal engineered methods of green determined nanoparticles, for example, substance decrease of metal particles in fluid arrangements regardless of balancing out specialists are accounted for in the writing [18-19]. Likewise, composite based materials are additionally orchestrated in colloidal gel structure and are tracks down numerous applications. In some cases the amalgamation of nanoparticles utilizing plantsor parts of plants can demonstrate beneficial over other natural cycles by wiping out the intricate cycles of keeping up with microbial societies [20-21].
In continuation of the abovementioned, It is accounted for in the current review that, combination of TiO2-GO NC s by organic technique utilizing ocimum tenuflorum plant remove. Blended examples are described by different characterisation procedures, for example, XRD, SEM, IR, EDAX and so on. Warm review is additionally attempted to know the warm way of behaving of the example
II. EXPERIMENTAL
A. Materials and Methods
The required chemicals used in the present experimentation are of AR grade. Double distilled waters are used as a solvent in the preparation of ocimum tenuflorum plant leaf extract. Glass wares used in the experimentation are rinsed with chromic acid. Biological reduction of salts using ocimum tenuflorum plant extract is adopted in the synthesis of TiO2-GO NC at room temperature. .
B. Preparation of ocimum tenuflorum plant leaf Extract
Ocimum tenuflorum plant extricate leaves were gathered early morning from its tree (Figure: 1) then, at that point, washed over and again with water. Known amount (say two grams) of the stripped leaves were collapsed into little pieces and are set into a tapered carafe loaded up with 150ml refined water. Heat up the substance of the conelike jar for around 10 minutes to get the watery leaf remove and is separated through whatmann no. 40 channel paper. Gather the fluid eluent separate arrangement in dried funnel shaped flagon and cover it with aluminum foil. Store the concentrate arrangement in dim and low temperature climate.
C. Preparation of Graphene Oxide (GO)
GO was incorporated from Graphite utilizing the changed Hummer's strategy [1]. In an ordinary examination, 2 g of graphite powder and 2 g of sodium nitrate were taken in 75 mL of concentrated sulphuric corrosive and 25 mL of concentrated nitric corrosive blend.
This blended arrangement was mixed in an attractive stirrer for 2 hours in an ice shower. To this arrangement gradually added 2 g of potassium permanganate more than 15 minutes with incredible mixing under an ice shower. Following 2 hours of response, the ice shower was eliminated.
The temperature of the response combination arrangement was raised to 35 ?. What's more, 100 mL of twofold refined water was added to the blend. At last, 100 mL of warm water was added and 10 mL of hydrogen peroxide was added to the earthy colored shaded arrangement.
The earthy colored arrangement went to light yellow and permitted the answer for settle for the time being. The subsequent item was separated, washed with 10% hydrochloric corrosive answer for eliminate metal particles and afterward washed more than once with refined water until the pH of the arrangement became impartial. The separated GO was at long last scattered in 200 mL of deionized water and saved for 3 hours in a ultrasonic shower. A scattered stable GO arrangement was gotten
D. Preparation of TiO2-GO NC
The reduced graphene oxide, decorated with TiO2 (TiO2-GO NC), was synthesized using TTIP, GO solution by simultaneous reduction and synthesis reaction using a plant extract as a reducing agent. In a typical experiment 0.05 moles of TTIP was added in to 100 ml of distilled water which was slowly added to 100 ml GO solution with constant stirring. 100 ml of ocimum tenuflorum leaves extract was added to the solution and stirred for 3 hours, maintaining temperature at 85 ?, and then the color of the solution was changed from reddish brown to black, later washed with distilled water and centrifuged at 6000 rpm. The TiO2-GO NC hybrid nanomaterial obtained for 20 minutes was dried and stored
E. Characterization
The structures of as prepared samples were studied by X – ray diffraction using X’ Pert Pro X–ray diffractometer with CuKα as source. Phillips XL 30 FESEM and Perkin–Elmer 1600 spectrophotometer in KBr medium tools are used to study morphology and bonding of the prepared sample respectively. LEO 1530 field emission scanning electron microscope with EDX system from OXFORD is used for the scan of sample. Metllor Toledo star instrument and Elico spectrophotometer are used to study the thermal behaviour and UV visible measurements of the sample.
III. RESULTS AND DISCUSSION
Visual Observation of Colour Change
Physical confirmation of the synthesized sample TiO2-GO NC was done by visual observation of the colour change in the solution. ocimum tenuflorum leaves extract is in green colour where as salt solution is in colourless. The formation of brownish colour solution appeared when plant extract is mixed with salt solutions indicates the reduction reaction leading to the formation of TiO2-GO NC.
A. X-ray Diffraction Analysis
Figure 1 (a) shows XRD pattern of TiO2, TiO2-RGO and TiO2-RGO NC samples respectively and its indexing is given table-1. The pattern shows the presence of intensed Bragg’s reflections for TiO2 is due to crystalline nature of the sample and is indexed in the pattern in accordance with JCPDS of TiO2. The d-spacing estimations of the sample is coordinated well with above JCPDS record confirmed the formation of the sample. The prepared TiO2 sample modified with RGO prepared manually reduced the crystallinity by masking. Hence, reduced intensity of the available reflections and also some reflections are masked. It also observed from pattern that, no reflection are found in the TiO2-RGO NC sample indicates the complete masking of the sample.
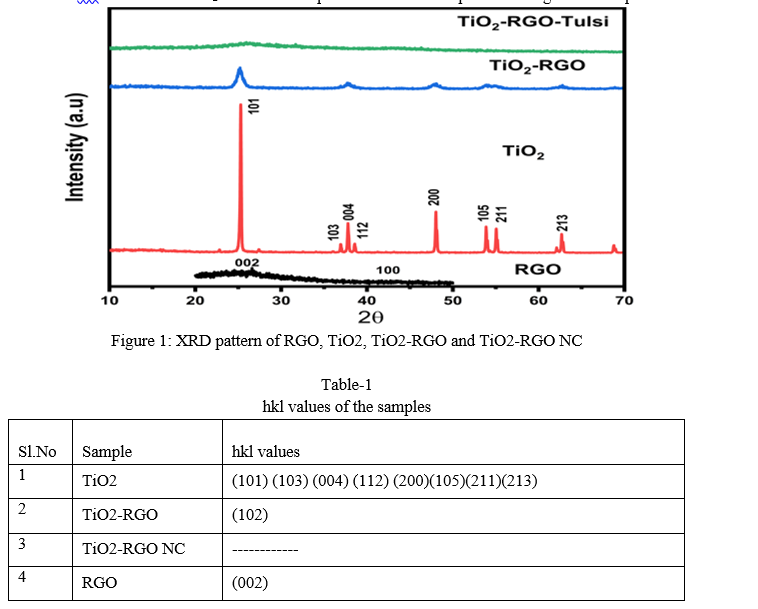
B. Scanning Electron Microscopy Analysis (SEM)
Field emanation filtering electron magnifying instrument (FESEM) has been utilized to look at the morphology of the arranged nanoparticle tests.
Figure 2 shows the SEM picture of arranged TiO2-RGO and TiO2-RGO NC, RGO, and TiO2 tests. The picture 2(a) shows fine development of framework morphology bound with exceptionally low crystallinity. Unpredictable particles with sheet structure explored the nanocomposite materials. Though figure 2(b) shows SEM pictures of TiO2-RGO NC nanoparticles test. One can find the fluctuated morphology in correlation with figure 2(a). Here likewise tracks down composite morphology with changed crystallinity.
Figure 2(c) shows the SEM picture of plane RGO test. It shows the somewhat glasslike nature. Fine consolidated thin state of the particles are seen in the morphological appraisal. The figure 2(d) shows plane TiO2 known for its glasslike nature. The picture shows translucent morphology with sporadic formed particles are seen in the picture. The majority of the circular formed particles characterize the translucent idea of the pre-arranged example. Improved molecule conservativeness is additionally be seen plainly in the picture.

C. Energy Dispersive X-Rays (EDX) Analysis
EDX report confirms the chemical composition or contaminants for the synthesized TiO2-RGO NC sample. Figure 3 shows EDAX pattern of as prepared TiO2-RGO NC and the data is given intable-2. The pattern shows Ag highest percentage signals at respective position confirms the formation the above NC sample. highest percentage signals in comparison with standard confirms the formation sample respective metals.
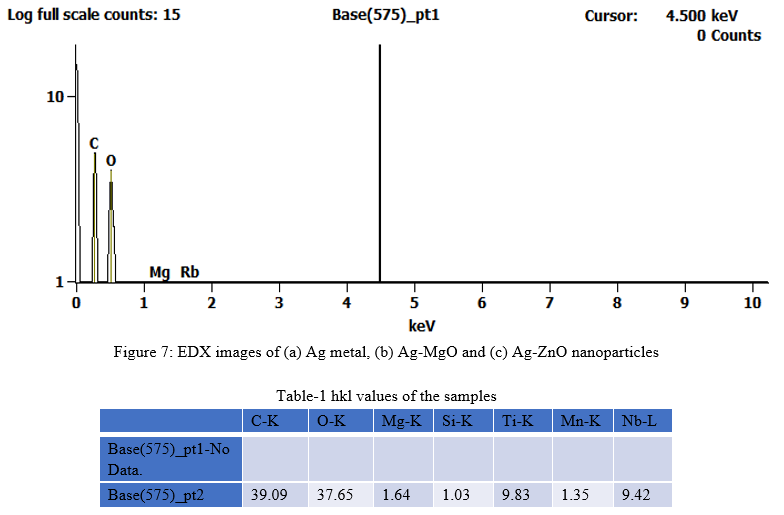
D. Infrared Study (FT-IR) analysis
Figure 3 shows FT-IR spectra of prepared (a) TiO2-RGO (b) TiO2-RGO NC (c) RGO (d) TiO2 samples respectively. The metal-oxygen bonding and nature of the synthesized oxide sample was carried out by infrared study. Metal oxides generally give absorption bands below 1000 cm-1 arising from inter-atomic vibrations. Peaks below 1000 cm-1 corresponds to Metal-oxygen (M-O) vibrational modes of the samples conform the formation of the above samples.
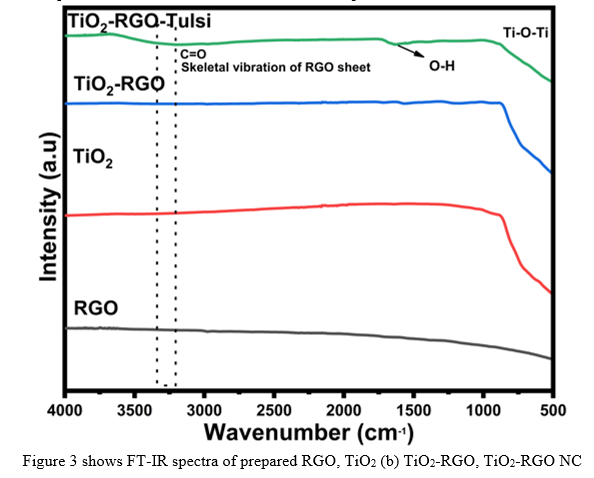
E. TGA
Figure 4 shows the thermal analysis (TGA) of Figure 3 shows FT-IR spectra of prepared RGO, TiO2 (b) TiO2-RGO, TiO2-RGO NC samples, which gives the information about the percentage weight loss of samples upon increasing the temperature, which in turn indicates thermal stability, material purity and moisture content of the samples. The thermograms of the samples indicates mass loss with prevalent weight loss. Initially, it starts decomposition gradually with rise in temperature. The major decomposition of nanocomposite occurs at intermediate temperature, indicating the thermal stability of composite. The binding force in the composite is due to the interaction between two component nanomaterials. The primary mass loss demonstrates loss of moisture and organic element from sample materials and finally weight loss of the sample.
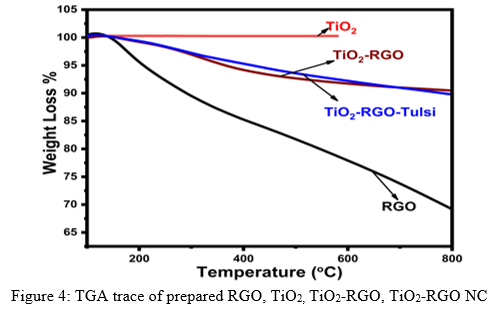
F. UV-Vis study
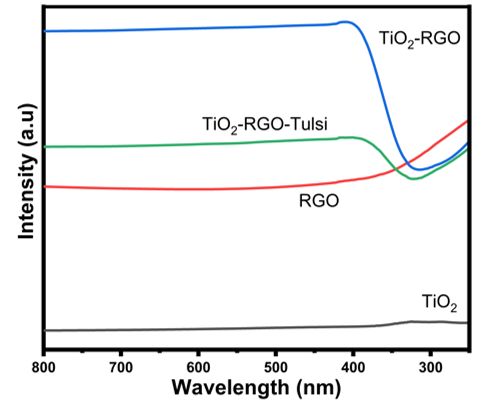
UV-Visible spectroscopy gave a solitary ingestion band around 320 nm for all the nanoparticle tests, which is areas of strength for an of the fruitful combination process. Figure 5 shows the UV spectra of as blended RGO, TiO2, TiO2-RGO, TiO2-RGO NC. Single plasmon band of tests at 430 nm that is at the center of the most extreme retention spectra. It is likewise affirmed that the nanoparticle parts are a not genuinely installed monometallic nanoparticles of various examples. The arrangement of the colloidal gel affirmation is made by noticing the moving the top towards red side.
Conclusion
Plant based nanocomposite materials are successfully prepared with biological reduction, which is towards environmental eco-friendly system. Ocimum tenuflorum plant leaf extract is used as a reducing agent and hence it can be used for the preparation other nanocomposite base materials. A. Funding This work is not financially supported by any funding agency Conflicts of Interest The authors declare no conflict of interest. B. Acknowledgement Authors offer sincere thanks to Prof. Dinesh Rangappa, Professor, Department of Nanotechnology, VTU regional centre Muddenhalli, Karnataka, India for providing instrumentation facility..
References
[1] R. Vaidyanathan, S. Gopalram, K. Kalishwaralal, V. Deepak, S. R Pandian, S. Gurunathan. (2010) Enhanced silver nanoparticle synthesis by optimization of nitrate reductase activity”, Colloids Surf. B Biointerfaces, 75(1): 335-341. [2] Kaviya S, S.J., Viswanathan B., \"Green Synthesis of silver nanoparticles using Polyalthialongifolia Leaf extract along with D-Sorbitol.\". Journal of nanotechnology, 2011: p. 1-5. [3] R. Vaidyanathan, S. Gopalram,: K. Kalishwaralal, V.Deepak, S. R Pandian, S. Gurunathan, Biosynthesis,purification and characterization of silver nanoparticles using Escherichiacoli,Colloids,Surf.BBiointerfaces, 2010,74,328-335. [4] M. Sastry, N. I. Islam, R. Kumar. Biosynthesis of metal nanoparticles using fungi and actinomycete, Current Sc., 2003, 85(2), 162-170. [5] Kasthuri J, K.K., Rajendran N., \"Phyllanthin assisted biosynthesis of silver and gold nanoarticles a novel biological approach.\". J Nanopart Res, 2009. 11: p. 1075-1085. [6] S. Basavaraja, S. D. Balaji, A.Lagashetty, S. Rajasab, A. Venkataraman, Preparation, characterization, and thermal studies of ?-Fe2O3 and CuO dispersed polycarbonate nanocomposites, Mater. Res. Bull. 2008, 41, 1162-1169. [7] Simin Wei, Yinghui Wang, Zhishu Tang, Jinhang Hu, Rui Su, Jingjie Lin, Tuan Zhou, Hui Guo, Nan Wang and Rongrong Xu, (2020) A size-controlled green synthesis of silver nanoparticles by using the berry extract of Sea Buckthorn and their biological activities, New J. Chem., 44: 9304-9312. [8] Guidelli EJ, R.M., Zaniquelli D, Baffa O., \"Green synthesis of colloidal silver nanoparticles using natural rubber latex extracted from Heveabrasiliensis.\". MolBiomolSpectrosc, 2011. 82(1): p. 140-145 [9] A.Lagashetty, Green Synthesis and Characterization of Silver Nanoparticles using Piper Betle [10] Leaf Extract, Bull.Adv.Sci.Res. 2015, 1 (5) 136-138. [11] Amarendra D.D, Biosynthesis of silver and gold nanoparticles using Chenopodium album leaf [12] Extracts, Colloid Surface A, 2010, 369 (3), 27-33. [13] S. Iravani, Green synthesis of metal nanoparticles using plants Green Chem, 2011, 13, 2638. [14] R. Upendra Kumar Parashar,: Preeti S.: Saxena,: Anchal Bioinspired Synthesis of Silver Nanoparticles Digest Journal of Nanomaterials and Biostructures, 2009, 4, 159-166. [15] E.K.P. Elumalai, T.N.V.K.V. Hemachandran, S. Viviyan, T.Thirumalai, E. David, [16] Extracellular synthesis of silver nanoparticles using leaves of Euphorbia hirta and their [17] Antibacterial activities Pharm Sci& Res, 2010, 2(9), 549-564. [18] Synthesis, Characterization and Antibacterial study of Ag-Au Bi-metallic nanocomposite by bio reduction using piper Betal leaf extract, Arunkumar Lagashetty and Sangappa K Ganiger, Shashidhar Reddy, Heliyon.5 (2019)e02794 [19] Lagashetty, A., Patil, M.K. Ganiger Sangappa K, Green Synthesis,Characterization, and Thermal Study of Silver Nanoparticles by Achras sapota, Psidium guajava, and Azadirachta indica Plant Extracts Plasmonics, 2019, 15, 1-8. [20] Sharma V K 1, Yngard R A, Lin Y, Silver nanoparticles green synthesis and their antimicrobial activities .Advances in Colloid and Interface Science, 2009,145(1),83-96. [21] P. Mohanpuria, K.N. Rana, S.K. Yadav, Bio-synthesis of nanoparticles: technological concepts and future applications. J. Nanopart. Res. 2008, 10, 507-517. [22] T. Thungunta, A.C. Reddy, D.C. N Lakshmana Reddy, Green synthesis of [23] Nanoparticles current prospectus, Nanotechnol. Rev. 2015, 4(4), 303–323, [24] K. Murthuramalingan, S.P.B. Sakthi, V.S. Benitha. Ind. J.Sci. 2015, 16(50), 30-39. [25] R. A. Sperling, P. R. Gil, F. Zhang, M. Zanella, W. J. Parak, Biological applications of gold nanoparticles, Chem. Soc. Rev. 2008, 37(9), 1896-1902.
Copyright
Copyright © 2022 Manjunath Patil, Arunkumar Lagashetty. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET43814
Publish Date : 2022-06-04
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

