Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Camparison of Extraction Techniques for Isolation Fungal DNA
Authors: Brijesh M. Chauhan, Keval M. Kunjadiya, Sanjay N. Parekh
DOI Link: https://doi.org/10.22214/ijraset.2022.41542
Certificate: View Certificate
Abstract
For effective molecular screening of fungi using polymerase chain reaction, a simple, quick, reliable, and inexpensive DNA extraction technique is always obliged. Because of the low sensitivity of culture-based detection methods, PCR-based techniques have been developed to amplify DNA from fungal infections. In PCR-based assay, pure DNA must be required, as well as an easy-to-follow DNA extraction approach. Due to amount of fungus in a tissue biopsy or clinical sample may be relatively small, extraction efficiency is extremely crucial in PCR-based medical diagnostic applications. In this study, we are comparing four different methods of DNA extraction and compared their efficacy in the extraction of DNA from filamentous fungi commonly isolated in our laboratory. It was found that the effectiveness of cell wall disruption decreasing in the following order: GeNei™ Fungal Genomic DNA Extraction Teaching Kit > SDS containing extraction buffer > CTAB containing extraction buffer and vortex with glass beads > CTAB and SDS containing extraction buffer.
Introduction
I. INTRODUCTION
The rapid and convenient detection of microbial pathogens is a fundamental requirement for the diagnosis of food-borne illnesses and diseases. Polymerase chain reaction (PCR)-based approaches have been established to amplify DNA of fungal infections because culture based detection approaches have a low accuracy. The proven DNA extraction procedures for bacteria and viruses result in poor fungal DNA release. This is due to the fact that fungi have a rigid and extremely complicated cell wall that protects and provides firmness to their cell structures. A variety of extraction protocol is available for isolation of DNA. No single extraction method seems to be optimal for all organisms [1]. The fungal cell wall structure is extremely complex. Fungal cell wall made up of Thick layers of chitin, (1–3)-B-d-glucan, (1,6) B-glucans, lipids, and peptides. There may also be a tough surface layer of melanin that is strongly resistant to UV light, enzymatic digestion, and chemical breakdown. Because of these physical characteristics of fungi, the development of a single universal fungal DNA extraction method has been hampered [8]. Many fungal DNA isolation methods use a variety of disruption methods to lyse fungal cell wall. All the disruption methods are classified into 3 groups (A) Physical Method (B) Chemical method (C) Enzymatic method. The physical methods including Glass beads method [3], Sonification method [4], Microwave method [5], and chemical methods includes Standard CTAB method [6,8], sodium dodecyl sulfate (SDS) method [9], Urea/Chelex/SDS method [10], and enzymatic methods includes Enzymatic lysis method [11].
We are studied four alternative methods to isolate fungal DNA. The first method is using SDS containing extraction buffer [3], the second method uses CTAB containing extraction buffer and vortex with glass beads [4], the third method is GeNei™ Fungal Genomic DNA Extraction Teaching Kit, and the fourth method uses CTAB and SDS containing extraction buffer [12] First, third and fourth method used chemicals for fungal cell wall breakage whereas second method use combination of physical and chemical method for the same purpose.
II. MATERIAL AND METHODS
A. Fungal Culture
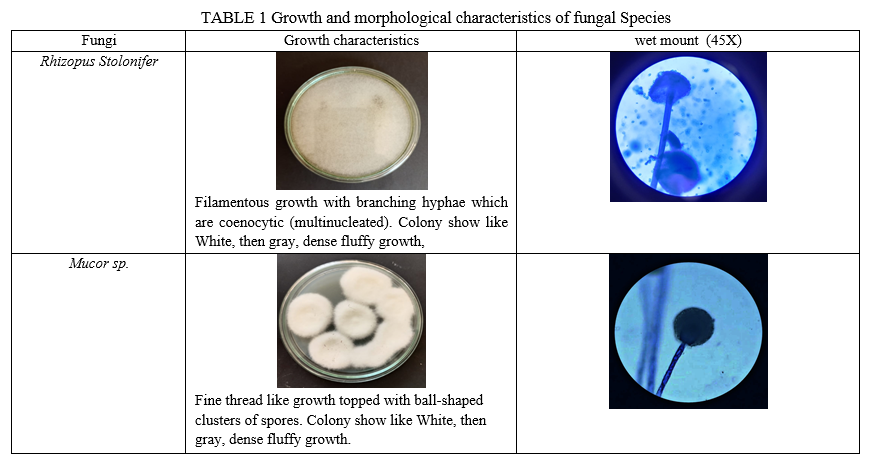
A pure culture of Rhizopus stolonifer species [NCIM 880] and mucor species [NCIM 880] were grow and maintained in our laboratory on Sabouraud Dextrose Agar (SDA) and Potato Dextrose Agar (PDA) respectively for 4-5 days at 30?.
B. DNA Extraction Methods
Following four methods were performed to extract DNA from both fungal cultures in duplicate.
- Method A: SDS containing Extraction buffer
Take fungal mass (approx. 50 mg) obtained from the culture plate or broth was placed in a microfuge tube containing 800 µl extraction buffer (1.0 M Tris-HCl pH 8, 0.5 M EDTA pH 8, 1.0 M KCL, 3.0 % SDS, 150 µL of 20 mg/mL proteinase K) and homogenized for 10 min. Put the tube in water bath maintained at 60°C for 30 min. Centrifuge at 10,000 rpm for 10 min. supernatant was transferred to a fresh microfuge tube.
Add equal volume of phenol: chloroform: Isoamyl alcohol (25:24:1) was added in supernatant and mixed well followed by centrifugation at 10,000 rpm for 10 min (Note: this step can be repeated once more to completely get rid of proteins/cell debris).Add equal volume of chloroform: Isoamyl alcohol (24:1) was added in supernatant and mixed well followed by centrifugation at 10.000 rpm for 10 min. The upper aqueous layer was taken in a fresh microcentrifuge tube and then adds equal volume of 100% ethanol. Incubate at for precipitation at -20°C for 30 min, the whole content was centrifuged at 10.000 rpm for 10 min to pellet down the DNA. The DNA pellet was washed with 800 μl 70% ethanol and centrifuged at 10,000 rpm for 5 min. Air dry DNA pellets and dissolved in 100 μl TE buffer (10 mM Tris-HCl pH 8, 1 mM EDTA) [3].
2. Method B: CTAB containing Extraction buffer and vortex with glass beads
Fresh fungal mycelium (approx.50 mg) was transferred to a sterilized 1.5 mL microcentrifuge tube with the help of sterilized scalpel and 800 µL of extraction buffer (0.1 M Tris-HCl pH 8, 10 mM EDTA pH 8, 2.5 M NaCl, 3.5% CTAB, 150 µL of 20 mg/mL proteinase K) was added with mixed sterilized 0.5-1 mm glass beads. The mixture was vortexed at high speed on a homogenizer for 5 min. The samples were placed in a water bath at 65°C for 30 min. The samples were then centrifuged at 10,000 rpm for 10 min at room temperature. Supernatant was collected and equal volume of phenol-chloroform-isoamylalcohol (25:24:1) was mixed. The samples were again centrifuged at 10,000 rpm for 10 min at room temperature. Supernatant was again collected and equal volume of chloroform-iso-amyl-alcohol (24:1) was mixed. Samples were again centrifuged under the conditions mentioned above. Supernatant was collected and equal volume of ice-cold isopropanol was added. Samples were incubated at -20°C for 1-2 h. The samples were centrifuged for 15 min at 10,000 rpm to pellet the DNA. Supernatant was decanted and DNA pellet was washed with 800 µL of 70% ethanol. DNA pellet was air-dried and dissolved in 100 µL TE buffer[4].
3. Method C: GeNei™ Fungal Genomic DNA Extraction Teaching Kit
Fresh fungal mycelium (approx.50 mg) was transferred to a sterilized 1.5 mL of microcentrifuge tube and Add 450 μl of Lysis Buffer A to each tube. Crush each of the fungal mycelium with the help of a tissue grinder to homogeneity. Add 50 µl of Solution B to each tube and crush again. Incubate at 65° C for 30 minutes with intermittent crushing. Add 500 µl Chlorofom: Iso-amylachol mixture directly to each of the crushed fungal pellet and mix well by inverting the vial. Centrifuge the vial at 8000 rpm for 10 minutes at room temperature. Remove the supernatant to a fresh vial and add equal volume of alcohol to the supernatant. Keep at room temperature for 15 minutes. Centrifuge at 14000 rpm for 30 minutes at room temperature. Discard the supernatant and air dry the DNA pellets. Note: Do not overdry. Add 50 μl of Solution D to each of the DNA Pellet. Note: To ensure complete recovery/ rehydration of the DNA, leave the sample in Solution D for 20-30 minutes at Room Temperature.
4. Method D: CTAB and SDS containing Extraction buffer
50 mg of lyophilized mycelial powder added with 500μl extraction buffer (250mM Tris-HCl pH 8.0, 20mM EDTA pH 8.0, 2.5 M NaCl, 10% CTAB, 0.15% SDS) was vortexed and then boiled for 10min at 50°C followed by centrifugation at 10,000rpm for 10min. The upper supernatant was pipetted out and mixed with one volume of chloroform: isoamyl alcohol (23:2) and centrifuged at 10,000rpm for 5min. The aqueous phase was recovered and mixed with one volume of ice cold isopropanol to precipitate DNA. Add 100 µL TE buffer [12].
C. Detection of DNA purity and Concentration
The concentration and purity of total DNA extract in the samples were measured spectrophotometrically at wavelengths
of A260 and A280 using Epoch microplate spectrophotometer (BioTek Inc. USA) using following equation.
Concentration (µg/ml) = (A260 reading – A320 reading) × dilution factor × 50µg/ml
12 microliters of purified DNA from each method was run on a 1% W/V agarose gel for electrophoretic separation, Gel visualized using the Biorad Gel doc XR system (BIO-RAD Laboratories Inc. USA)
D. PCR Amplification
PCR amplification of fungal DNA was performed in Biorad MyCycler (BIO-RAD Laboratories Inc. USA) using universal primers targeting a highly conserved region of the fungal 18S small-subunit rRNA. PCR was carried out using primer set ITs1[TCCGTAGGTGAACCTGCGG]/ITs4[TCCTCCGCTTATTGATATGC] with 2X PCR TaqMixture (HiMedia Laboratories Pvt. Ltd., India). It is a pre-mixed, ready-to-use solution containing optimal concentrations of Taq DNA polymerase, dNTPs, MgCl2, and reaction buffers for efficient PCR amplification of DNA templates.
Because there are fewer pipetting steps required for PCR setup, this pre-mixed formulation saves time and reduces contamination. The mixture has been optimised for efficient and repeatable PCR. The reaction conditions were as follows: initial denaturation step at 94°C for 4 min. 40 amplification cycle of denaturation at 94°C for 1 min. annealing at 50°C for 1 min and primer extension at 72°C for 3 min: followed by a final extension at 72°C for 10 min. Aliquots of the PCR products (12µL) were analyzed in 1.5% (w/v) agarose gels electrophoresis.
III. RESULTS AND DISCUSSION
The primary criteria for evaluating DNA extraction methods is purity and amount of DNA obtained. The extraction methods must provide the enough amount of pure DNA that can be amplified further by PCR while not containing inhibitors that would block PCR [18]. The amount of DNA obtained was always proportional to the amount of mycelium used for DNA isolation. Each method of DNA isolation was performed for Rhizopus stolonifer [NCIM 880] and mucor species [NCIM 880] in duplicate. Growth and morphological characteristics of both this fungi are shown in Table I. Guo J. pointed out the differences in DNA extraction results between individual fungal strains and species [19].
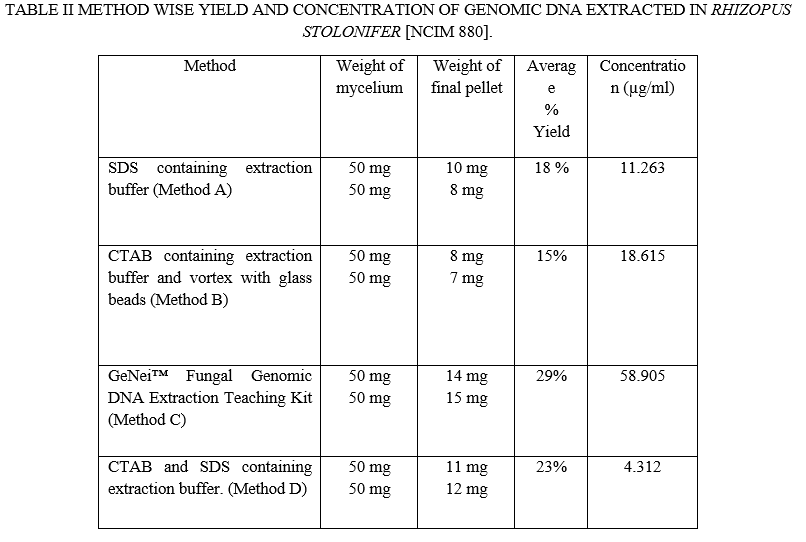
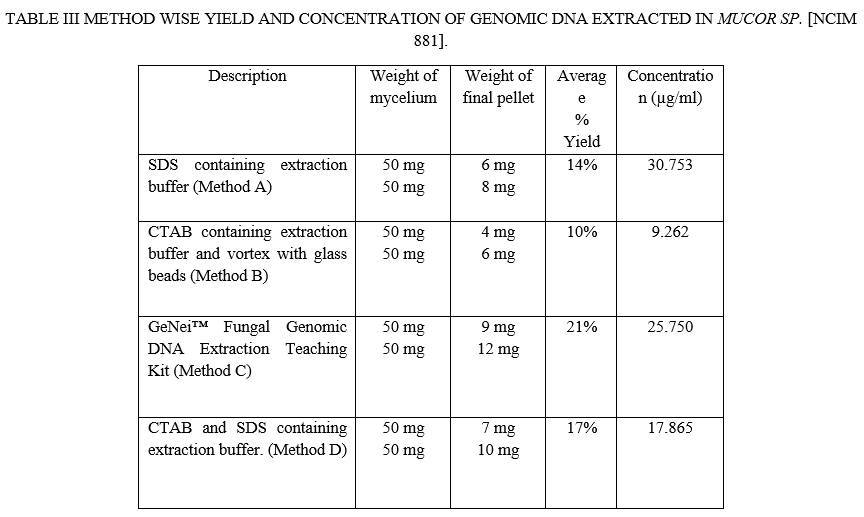
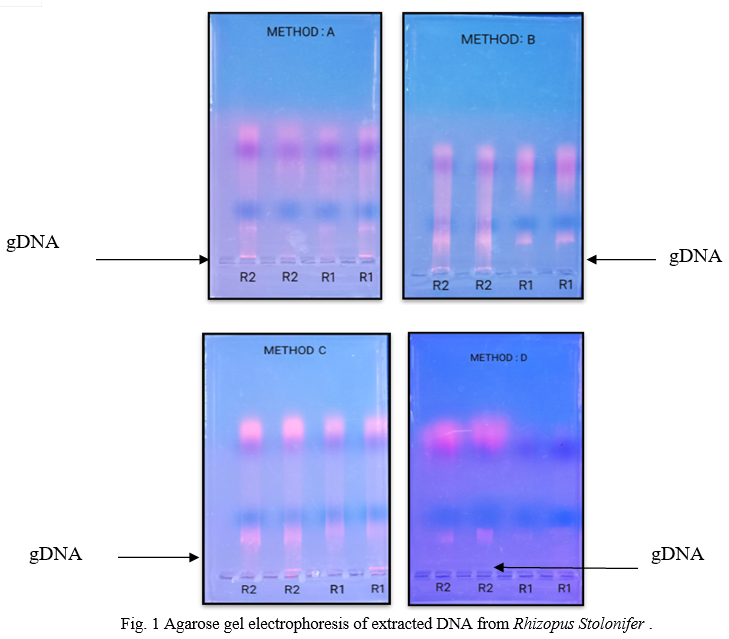
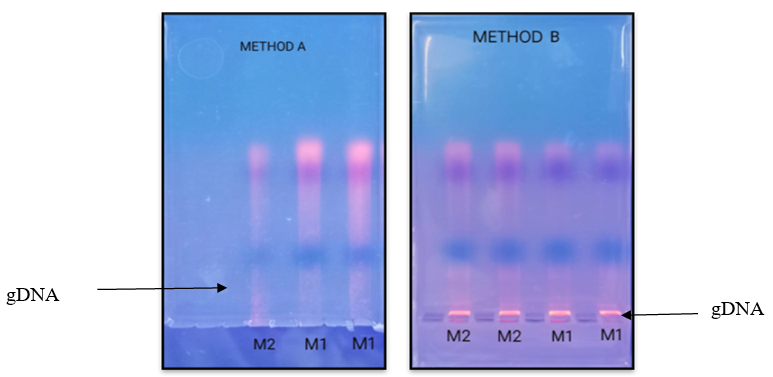
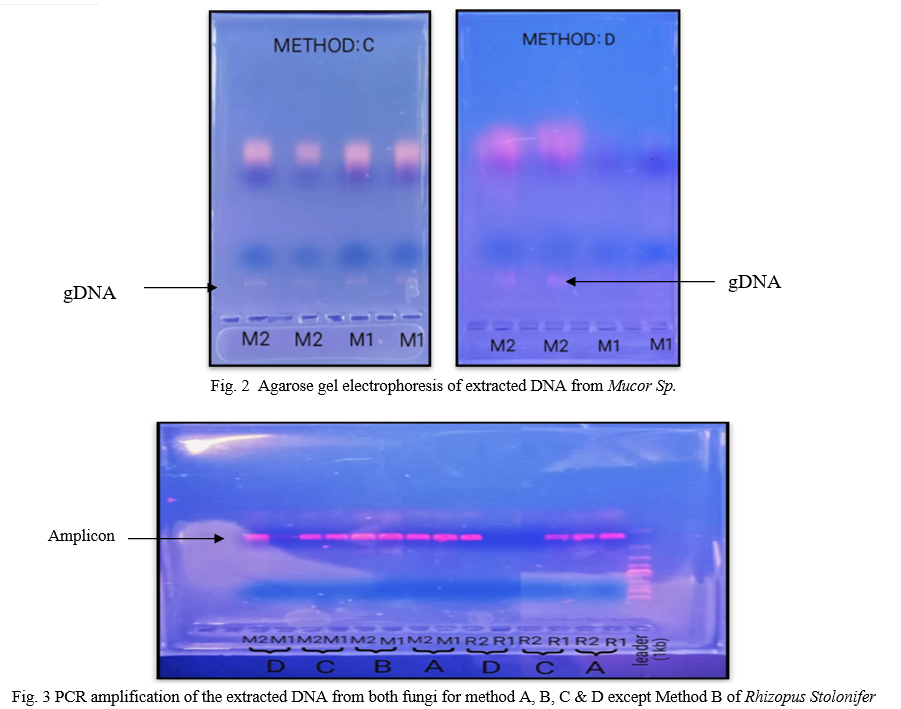
Electrophoretic separation of fungal genomic DNA extraction was confirmed on agarose gel for both fungi by loading duplicate sample (like R1, R2, M1, M2) as shown in figure 1& 2. Spectroscopic analysis to determine yield and concentration of extracted DNA is shown for all methods in Table II & III for Rhizopus Sp. And Mucor Sp. respectively. The GeNei™ Fungal Genomic DNA Extraction Teaching Kit (Method C) gave highest yield whereas CTAB containing extraction buffer and vortex with glass beads (Method B) gave lowest yield with both fungi. The amount of DNA obtained by the combined CTAB and SDS method (Method D) for the Rhizopus Stolonifer was found to be low. One possibility is that there is no DNA in the reaction mixture due to insufficient cell wall disruption or DNA loss during the purification steps. According to the results obtained, the method with the highest to lowest yield of DNA can be summarize as C > D > A > B for both fungal species. Results of Rhizopus Stolonifer and Mucor Sp. shows that the amount of DNA obtained from highest to lowest concentration is C > B > A > D and A > C > D > B respectively.
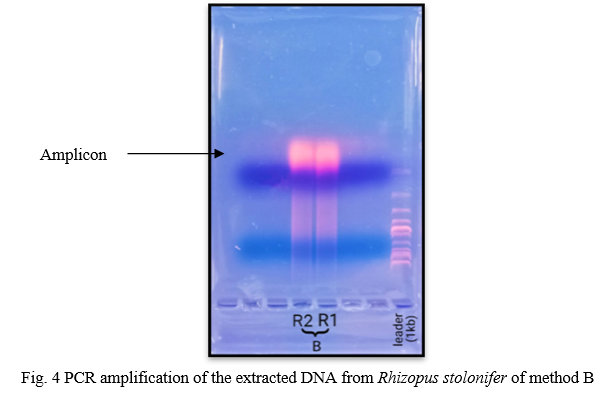
For confirmation of genomic DNA, PCR reaction was carried out using primers (ITS1-TCCGTAGGTGAACCTGCGG / ITS4-TCCTCCGCTTATTGATATGC) complementary to the 18S small-subunit rRNA. All methods have produced enough DNA for PCR assays. According to Olexov et. al, if DNA is isolated for the following use in PCR, the concentration of the amplifiable amount of DNA best characterises the quality of the DNA used [18]. Again results of PCR reaction suggest that GeNei™ Fungal Genomic DNA Extraction Teaching Kit (Method C) produce good amount of amplified DNA band. According to the PCR results, four methods can be arrange in order as follow C > B > A > D with respect to band obtained.
IV. ACKNOWLEDGEMENT
We sincerely acknowledge and thank all researchers for their significant contributions to this endeavour.
Conclusion
The methods for isolating fungal genomic DNA were compared in this study based on the following criteria: the purity of the DNA obtained, the amount of DNA, the speed of the isolation method, the equipment used, and the cost demands. Four different methods for extraction DNA were introduced and optimised. These included not only commercially available kits, but also the traditional method of DNA isolation using various types of cell wall disruption. For cell wall disruption, various extraction buffers were used. It was shown that the method of cell wall disruption has a significant impact on the effectiveness of DNA isolation. Effectiveness of cell wall disruption decreasing in the following order: GeNei™ Fungal Genomic DNA Extraction Teaching Kit > SDS containing extraction buffer > CTAB containing extraction buffer and vortex with glass beads > CTAB and SDS containing extraction buffer.
References
[1] Bolano et al., \"Rapid methods to extract DNA and RNA from Cryptococcus neoformans\", FEMS Yeast Research, vol. 1, no. 3, pp. 221-224, 2001. [2] A. A. Karakousis, L. Tan, D. Ellis, H. Alexiou and P. Wormald, \"An assessment of the efficiency of fungal DNA extraction methods for maximizing the detection of medically important fungi using PCR\", Journal of Microbiological Methods, vol. 65, no. 1, pp. 38-48, 2006. [3] I. Gontia-Mishra, N. Tripathi and S. Tiwari, \"A simple and rapid DNA extraction protocol for filamentous fungi efficient for molecular studies\", Nopr.niscair.res.in, 2022. [4] VAN BURIK, SCHRECKHISE, WHITE, BOWDEN and MYERSON, \"Comparison of six extraction techniques for isolation of DNA from filamentous fungi\", Medical Mycology, vol. 36, no. 5, pp. 299-303, 1998. [5] S. Tendulkar, A. Gupta and B. Chattoo, \"A simple protocol for isolation of fungal DNA\", Biotechnology Letters, vol. 25, no. 22, pp. 1941-1944, 2003. [6] S. Lee, M. Milgroom and J. Taylor, \"A rapid, high yield mini-prep method for isolation of total genomic DNA from fungi.\", Fungal Genetics Reports, vol. 35, no. 1, p. 23, 1988.. [7] S. Aamir, \"A rapid and efficient method of fungal genomic DNA extraction, suitable for PCR based molecular methods\", Plant Pathology & Quarantine, vol. 5, no. 2, pp. 74-81, 2015. [8] Y. Zhang, S. Zhang, X. Liu, H. Wen and M. Wang, \"A simple method of genomic DNA extraction suitable for analysis of bulk fungal strains\", Letters in Applied Microbiology, vol. 51, pp. 114-118, 2010.. [9] T. R. Prabha, K. Revathi, M. S. Vinod, S. P. Shanthakumar and Paul Bernard, \"A simple method for total genomic DNA extraction from water moulds\", Research gate, 2013. [10] F. Mseddi, M. Jarboui, A. Sellami, H. Sellami and A. Ayadi, \"A rapid and easy method for the DNA extraction from Cryptococcus neoformans\", Biological Procedures Online, vol. 13, no. 1, 2011. [11] R. Flamm, D. Hinrichs and M. Thomashow, \"Introduction of pAM beta 1 into Listeria monocytogenes by conjugation and homology between native L. monocytogenes plasmids\", Infection and Immunity, vol. 44, no. 1, pp. 157-161, 1984. [12] S. Umesha, H. Manukumar and S. Raghava, \"A rapid method for isolation of genomic DNA from food-borne fungal pathogens\", 3 Biotech, vol. 6, no. 2, 2016. [13] S. Scharf, A. Bartels, M. Kondakci, K. Pfeffer, B. Henrich and R. Haas, \"Introduction of a bead beating step improves fungal DNA extraction from selected patient specimens\", International Journal of Medical Microbiology, vol. 310, no. 6, p. 151443, 2020.. [14] P. Mo?ková and J. Vyt?asová, \"Comparison of methods for isolating fungal DNA\", Czech Journal of Food Sciences, vol. 29, no., pp. S76-S85, 2012. [15] A. Karakousis, L. Tan, D. Ellis, H. Alexiou and P. Wormald, \"An assessment of the efficiency of fungal DNA extraction methods for maximizing the detection of medically important fungi using PCR\", Journal of Microbiological Methods, vol. 65, no. 1, pp. 38-48, 2006. [16] M. Kumar and M. Mugunthan, \"Evaluation of three DNA extraction methods from fungal cultures\", Medical Journal Armed Forces India, vol. 74, no. 4, pp. 333-336, 2018. [17] Cenis, J., 1992. Rapid extraction of fungal DNA for PCR amplification. Nucleic Acids Research, 20(9), pp.2380-2380. [18] l. Olexova, T. Kuchta and L. Dovicovicova, \"Comparison of three types of methods for the isolation of DNA from flours, biscuits and instant paps\", European Food Research and Technology, vol. 218, no. 4, pp. 390-393, 2004. [19] J. Guo, F. Schnieder, K. Abd-Elsalam and J. Verreet, \"Rapid and efficient extraction of genomic DNA from differentphytopathogenic fungi using DNAzol reagent\", Biotechnology Letters, vol. 27, no. 1, pp. 3-6, 2005.
Copyright
Copyright © 2022 Brijesh M. Chauhan, Keval M. Kunjadiya, Sanjay N. Parekh. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET41542
Publish Date : 2022-04-17
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

