Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Early Detection of Stages of Alzheimer\\'s Disease from MRI Scans using Analysis of Hippocampus Region
Authors: Kavitha Vasanth, Darshana Thilakan, Ashwini N, Anagha J A
DOI Link: https://doi.org/10.22214/ijraset.2022.44241
Certificate: View Certificate
Abstract
Alzheimer\'s disease is a progressive neurological brain disorder that gradually destroys memory cells and causes the brain to shrink. It is the most common cause of Dementia, which results in a steady decline in cognitive skills such as thinking, social, and behavioral skills, affecting the person\'s ability to work independently. This disease is usually found in the age group of 65 years and above. Alzheimer\'s disease must be detected early in order to prevent the disease from progressing irreversibly. In this work, an approach for detecting Alzheimer\'s disease in its early stages is proposed. The proposed method employs a Machine Learning algorithm to diagnose Alzheimer\'s disease from an MRI scan of the Hippocampus. The Hippocampus region\'s area, shape, and texture features are retrieved, and the percentage and stages of Alzheimer\'s Disease are detected using these features. For feature extraction and feature classification, Machine Learning algorithms such as Convolutional Neural Network and K-Nearest Neighbor are utilised. In comparison to other conventional methodologies, the proposed approach is predicted to provide improved accuracy.
Introduction
I. INTRODUCTION
Alzheimer's disease (AD) is a degenerative neurological illness that is the leading cause of dementia among people aged 65 and higher. The disease causes the brain cells to shrink and to die. Continuous shrinkage pattern of brain cell is observed in this disease. At first, Alzheimer's disease damages neurons and their connections in the hippocampus region of the brain, which is crucial in memory. Later on, it affects the parts of the cerebral cortex that control language, logic, and social interaction. Various other areas and parts of the brain are also eliminated throughout time. Alzheimer's disease gradually deteriorates a person's ability to live and function freely. In the end, this illness is fatal. The brain typically shrinks to some degree which results in the formation of plagues and tangles in the hippocampal region. The aberrant buildup of proteins in the brain causes Alzheimer's Disease.
These proteins trigger cell death and are known as amyloid and tau. Amyloid proteins clump together in brain cells, forming plaques, or larger aggregates. Tau protein tangles, on the other hand, are formed by twisted fibers. These plaques and tangles hinder nerve cell communication, making it impossible for them to perform their jobs. Patients' symptoms are the result of a slow and steady loss of nerve cells that starts in one part of the brain and progresses to other parts. The brain has been severely damaged, with many neurons losing their ability to function, losing connections with other neurons, and eventually dying. The hippocampus is a complex brain structure located in the temporal lobe of the brain. It is mostly involved in memory and learning. The loss of cells and damage to connections are the main causes of hippocampal atrophy. In the early, middle, and late stages of Alzheimer's disease, the disease usually progresses slowly. Because this disease affects people differently, each person may experience symptoms in different ways as the disease progresses. In the early stages of Alzheimer's disease, a person with the disease may be able to operate independently. He or she may be able to work and socialise at the same time. Despite this, the individual may experience memory lapses, such as forgetting familiar words or the location of common place objects.
The intermediate stage of Alzheimer's disease is the most severe and can linger for years. The following are some of the symptoms that may be experienced during this stage: the person may misunderstand words, become upset or furious, and act in unexpected ways. In the medium stage of Alzheimer's disease, the individual suffering from the disease can still engage in daily activities with the help of others. In the last stages of dementia, the symptoms are severe. Individuals lose their ability to react to their environment, communicate, and control their motions. Communication gets harder during this stage as memory and cognitive abilities deteriorate. Non-automated methods such as Cognitive Impairment Testing, Mini-Mental State Examination (MMSE), and Clinical Dementia Rating (CDR), as well as imaging techniques such as Magnetic Resonance Imaging (MRI), Positron Emission Tomography (PET), and Single-Photon Emission Computed Tomography (SPECT), make early diagnosis of Alzheimer's Disease difficult at the present time.
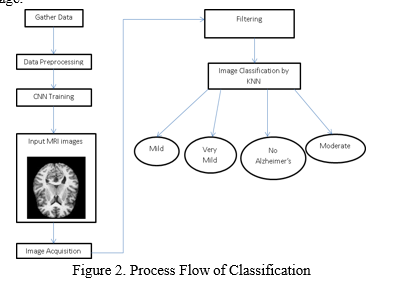
MRI scans are used in the proposed method for early diagnosis of Alzheimer's disease. The texture, area, and shape properties are derived from the Hippocampus, which has been designated as the Region Of Interest (ROI). Convolutional Neural Networks (CNN) are used to extract these traits. As a result, the percentage of the AD is calculated followed by classification of the various stages of the disease is done using K- Nearest Neighbor (KNN) Algorithm.
II. RELATED WORK
Priyanka Lodha, Ajay Talele, Kishori Degaonkar, proposed Support Vector Machine (SVM) and Gradient Boosting Algorithms for classification problems. Neural Network and Random Forest Algorithms were used for detecting AD. Data sets (CT, MRI, PET, EEG) were obtained from Alzheimer’s Disease Neuroimaging Initiative (ADNI). The remarks obtained from this paper is to evaluate the approach more extensively using a larger data set.
Salim Lahmiri, Amir Shmuel proposed a system that focuses Linear Discriminant Analysis (LDA), K- Nearest Neighbor (KNN) Algorithm, Nave Bayes (NB), and Support Vector Machine (SVM) are four extensively used machine learning classifiers to discriminate between photos of Alzheimer's patients and normal person. Data sets were taken from ADNI. This approach focuses on evaluating the effectiveness of the system, incorporating the SVM classifier trained with cortical matrix for accurate distinction between AD and healthy control subjects.
Gokce UYSAL, Mahmut OZTURK proposed an approach using KNN, SVM, Decision Tree and Random Forest. In this study right and left Hippocampal volume, age and gender parameters are the data sets from ADNI. Only prediction of AD is done and accuracy can be improved. As only prediction is done, accuracy of the entire system can be improved by classifying the AD into different stages.
Karrar A.Kadhim, Farhan Mohamed, Zaid Nidhal Khuthair proposed approach using Deep Learning and MRI Scan techniques to detect AD. Feature extraction using SVM classifier was done. Data sets were taken from ADNI. Datasets were limited and clarity can be improved. The future work can be done by evaluating the approach more extensively using a larger data set.
Emtiaz Hussain, Mahmudul Hasan, Syed Zafrul Hassan proposed an approach using brain MRI data, CNN was used to do binary classification and detection of Alzheimer's disease. The data has been collected from Open Access Series Of Imaging Study (OASIS). Binary classification and detection of AD was performed but lacked multi-class classification. The goal of future research is to use the OASIS data set to do multi-class classification and early diagnosis of Alzheimer's disease.
J.Neelaveni, M.S. Geetha Devasena proposed approach using algorithms like SVM, Decision Tree to predict the disease using psychological parameters like age, number of visit, MMSE score and education. The data sets were taken from ADNI. The disease could be accurately predicted at an earlier stage. Future research could combine both brain MRI scans and psychosocial characteristics to predict disease with more accuracy.
Aakash Shah, Dhruvi Lalakiya, Shreya proposed an approach which was carried out between analysis of five algorithms namely SVM, Voting Classifier, Decision Tree, Random Forest and XG Boost. To have a more accurate detection system, multi-modal frameworks might be constructed. To achieve precision and accuracy, image data can be mixed with textual data.
Sivakani R, Gufran Ahmad Ansari proposed a approach in which the EM Algorithm is used for feature extraction and the Best First Algorithm is used for feature selection. The Gaussian Process algorithm is used to classify the data. Data sets are taken from OASIS. Feature extraction and selection was not done accurately. To improve categorization, more research into feature extraction and selection is required.
Arpita Raut, Vipul Dalal proposed an ANN-based method for detecting Alzheimer's disease from MRI scans. It extracts characteristics from MRI scans' hippocampus area using Gray Level Co-occurrence Matrix and Moment Invariants. A MRI based data set is considered from OASIS. This approach is not able to detect AD at early stage. The proposed approach is now being implemented and is expected to improve accuracy.
Ms. Kirthiga Devi T, Dr. Maragatham G, Ms.Janani S, Mr. Dhanush Kumar K proposed an approach using brain MRI image to detect AD. CNN is used for image recognition, Rectified Linear Unit for feature extraction. The data sets are from ADNI. This system does not include a user interface that can be used by non-professionals to upload photos and detect the onset of disease. As a part of future work user interface can be provided.
III. PROPOSED APPROACH
The proposed approach is split into two parts:
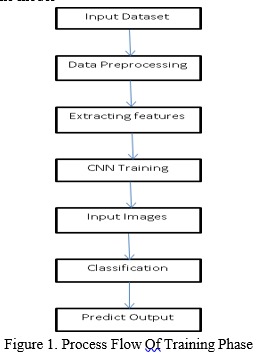
- Training Phase: A set of labeled data sets i.e. MRI Scans are imported and are used as an input for further processing. The various steps involved are as follows:
- Preprocessing Stage: It is the first and most important step in the process of developing a machine learning model. Data preprocessing is a process of preparing the raw data and making it suitable and understandable for Machine Learning Model. The quality of the data should be checked before applying Machine Learning or Data Mining Algorithms. Hence enhaces the clarity of the data sets.
The various data preprocessing techniques include:
a. Data Integration: Data Integration is a preprocessing technique that involves combining data from multiple heterogeneous data sources into a coherent data store and provide a unified view of the data.
b. Data Cleaning: Data Cleaning is the process of fixing or removing incorrect, corrupted, incorrectly formatted, duplicate or incomplete data within a data set. When combining multiple data sources, there are many opportunities for data to be duplicated or mislabeled.
c. Filtering: Filter methods are generally used as a preprocessing step. The selection of the features is independent of any machine learning algorithms. Instead features are selected on the basis of their scores in various statistical steps for their co-relation with the outcome variable. Data filtering is the process of choosing a smaller part of the data set and using that subset for viewing or analysis. Filtering is generally (but not always) temporary-the complete data set is kept, but only a part of it is used for the calculation.
3. Feature Extraction: The texture, area, and shape features of the Hippocampus region of the MRI scan are retrieved in this step. Theses subsets are used for viewing or analysis.
4. Training the Data Sets: The Convolutional Neural Network (CNN) Algorithm is used to train the data sets. CNN is a deep learning algorithm. It is used to differentiate the MRI Scans based on the shrinkage of the Hippocampus region. Because of its great accuracy, it is utilized for image categorization and recognition.
The Training in CNN is done using three layers namely:
a. Convolutional Layer: Here, the feature extraction will take place where only the useful features which are needed to the machine will be collected and unwanted features will be removed so that training period will be finished soon.
b. Pooling Layer: In this, the size of the data or image will be reduced and a compressed document with the important features which is needed for the machine is obtained.
c. Fully Connected Layer: Here, the above data which we get from the previous layer will be fed to fully connected layer in a vector form. Then the compressed features will be split and get trained using CNN resulting in final output.
The data will be split into training and testing. Then using Conv2D the feature extraction from the MRI Scans are done. Using Max Pooling the pixel size of the image are reduced in order to finish the training process soon. Finally, the compressed features are put into vector form and trained each feature to the model
- Classification Phase: The Classification of MRI Scans into different stages and percentage of AD is done using K- Nearest Neighbor (KNN) Algorithm and this is given as an output. It stores all the available data and classify them based on similar features. It is a versatile algorithm because it can address both classification and regression problems. It is a simple algorithm to interpret having high accuracy as comparison with better supervised learning model is not needed. This algorithm has quick calculation time. KNN is a Supervised Machine Learning Algorithm. It's used for classification in this case. The KNN classifier identifies the AD by looking at the components that determine the data vectors while taking the classes into account. This strategy improves data mining efficiency and performance. The categorization time was shortened. The KNN Algorithm maintains the data set throughout the training phase, and when it receives new data, it classifies it into a category that is quite similar to fresh data. When an image is taken as input the KNN classifier checks as to which stage the MRI Scan belongs to and predicts that stage as an output. The data sets are classified into different stages namely: No Alzheimer’s, Very Mild , Mild and Moderate Alzheimer’s Stage.
Conclusion
The goal of this project is to create a reliable system that uses Machine Learning Algorithms to diagnose Alzheimer\'s Disease. The Hippocampus region is considered the Region Of Interest (ROI) using MRI scans as data sets. Using the Convolutional Neural Network (CNN) Algorithm, the texture, shape, and area properties of the Hippocampus are retrieved. The percentage and the stages of the AD are predicted using KNN Algorithm. This project is very helpful in the medical fields. It can be used by Doctors or other professional staffs to detect the stages of AD accurately. If it is in the final stage then immediate actions can be taken. It reduces the time and efforts of the doctor as large number of MRI Scans can be predicted in less amount of time.
References
[1] McKee, A.C., Daneshvar, D.H., 2015, The neuropathology of traumatic brain injury, Handbook of Clinical Neurology 147(3), In: Grafman, I.J., Salazar A.M. (ed), Chapter 4, Elsevier B.V., Boston, 45-66. [2] Woo, C.W., Chang, L.J., Lindquist, M.A., Wager, T.D., 2017, Building better biomarkers: brain models in translational neuroimaging, Nat Neurosci, 20,365-377. [3] Petrella, J. R., 2013, Neuroimaging and the Search for a Cure for Alzheimer Disease, Radiology, 269 (3), 671–691. [4] Weiner, M. W., Veitch, D. P., Aisen, P. S., Beckett, L. A., Cairns, N. J., Green, R. C., Harvey, D., Jack, C.R., Jagust, W., Liu, E., Morris, J.C., Petersen, R.C, Saykin, A.J., Schmidt, M.E., Shaw, L., Siuciak, J.A., Soares, H., Toga, A.W., Trojanowski,J.Q., 2012, The Alzheimer’s Disease Neuroimaging Initiative: A review of papers published since its inception, Alzheimer’s & Dementia, 8(1), 1–68. [5] Padurariu, M., Ciobica, A., Mavroudis, I., Fotiou, D., Baloyannis, S., 2012, Hippocampal neuronal loss in the CA1 and CA3 areas of Alzheimer’s disease patients, Psychiatr Danub, 24 (2), 152-158. [6] Pokorski, R.J., 2002, Differentiating Age-Related Memory Loss From Early Dementia, Journal of Insurance Medicine, 34, 100- 113. [7] Richter-Levin, G., 2004, The Amygdala, the Hippocampus, and Emotional Modulation of Memory, The Neuroscientist, 10 (1), 31–39. [8] Eagleman D. The Brain: The Story of You. Istanbul. Domingo 2018; 11. [9] Risacher, S.L., Saykin, A.J., West, J.D., Shen, L., Firpi, H.A., McDonald, B.C., ADNI, 2009, Baseline MRI Predictors of Conversion from MCI to Probable AD in the ADNI Cohort, Current Alzheimer Research, 6, 347- 361. [10] Rowland, D.C., Moser, M.B., 2013, Time finds its place in the hippocampus, Neuron, 78 (6), 953-954. [10] Sontheimer, H., 2015, Aging, Dementia, and Alzheimer Disease, Diseases of the Nervous System, 4, 99–131. [11] Treves, A., Rolls, E.T., 1994, Computational analysis of the role of the hippocampus in memory, Hippocampus, 4 (3), 374–391. [12] Zandifar, A., Fonov, V., Coupé, P., Pruessner, J., Collins, D.L., 2017, A comparison of accurate automatic hippocampal segmentation methods, NeuroImage, 155, 383–393.
Copyright
Copyright © 2022 Kavitha Vasanth, Darshana Thilakan, Ashwini N, Anagha J A. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET44241
Publish Date : 2022-06-14
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

