Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Economical Wastewater Treatment for Removal of Copper (II) Ions From Wastewater
Authors: Deokar G. N., Desale L. R, Harkal S. P
DOI Link: https://doi.org/10.22214/ijraset.2023.51068
Certificate: View Certificate
Abstract
Water pollution is one of the major problems faced by the entire world. The industrial and urban development leads to the water pollution due to accumulation of heavy metals in the wastewater. Several techniques have been used such as chemical precipitation, electrolysis, ion exchange etc. to treat such water contaminated with heavy metals. One of the best option is adsorption, which is easier environment friendly method to treat wastewater. By using various natural and agro-based wastes as an adsorbent is useful in reducing cost in adsorption process. An attempt to use natural material like soil with activation and banana peel has made to treat water contaminated with heavy metals. In this study, the copper ions are removed with adsorption by using banana peels and activated soil. The performance of adsorbent is observed by varying different parameters such as pH, adsorbent dose, initial concentration and contact time. It is found that the sufficient removal of Cu ions at optimum parameters like pH, adsorbent dose, initial concentration and contact time is at 5, 2 gm, 5 mg/l and 150 minute respectively for banana peels and 6, 2gm, 5mg/l and 150 minute respectively for activated soil. For all optimum parameters the maximum reduction in metal ions is observed in the range of 70% to 80% for banana peels whereas for activated soil it is 65 % to 75%. The R2 value in the present analysis is less than one, representing that the adsorption of copper ions onto both the adsorbents is favorable and the equilibrium data also fit well with Langmuir and Freundlich isotherm.
Introduction
I. INTRODUCTION
During the past decades, heavy metals have emerged as a major inorganic pollutant and threat to natural environment and human health. The only solution to protect the environment and human health is their removal from metal laden wastewater before discharging them into aqueous streams.
These metal ions, primarily originate from electroplating, mining & ore processing, paint, chemicals and fertilizer industries and metal surface treatment processes. Among the heavy metals, copper is the major available type of heavy metal in the aquatic environment. The excess copper compound in the body has gastrointestinal effects. During the last few decades, the sustainable removal of heavy metals from water and wastewater has become a prime concern and a major challenge for scientists. Owing to the toxic and adverse effects of heavy metals, most industries are advised to treat wastewaters systematically so that the metal contents can be minimized in their wastes.
Several conventional technologies are in use for heavy metal removal from wastewater. These technologies include chemical precipitation, redox approach, ion exchange, electrolysis, membrane separation and adsorption. Generally, all these treatments lead to certain disadvantages such as incomplete removal of heavy metals, high-energy requirements and production of toxic sludge. .Extensive studies have been undertaken for the development of more effective methods in removing metal pollution. Amongst these, adsorption is more reliable and promising owing to its efficiency, local availability of adsorbents, operational simplicity, cost effectiveness and regeneration potential of the adsorbents. Numerous approaches have been studied in recent years in order to find an economic adsorbents for water treatment and heavy metals removing.
II. METHODOLOGY
A. Material Preparation
- Preparation of Adsorbent
Banana peels from Banana fruits are collected. Banana peels were dried in sunlight for 3 days and cuts into small pieces then washed with distilled water and dried it in oven at 105°C for 5 hours. Oven dried pieces were grounded and sieved using IS sieve.
Soil required for brick manufacturing is collected. The soil sample is oven dried for 24 hrs and then washed with distilled water and diluted hydrochloric acid. Further it is dried in oven at 105°C for 5 to 6 hours.
2. Preparation of Stock Solution
The stock solution 1000 mg/L of Cu(II) ions is prepared by dissolving 3.51g of pure salt CuSO4.5H2O in 1L of distilled water and practiced for all experiments with required dilution.
B. Test on adsorbents
- pH
Take 10 gm of sample add 100 ml distilled water in it. The sample is transferred to bottle and kept for shaking at 200 rpm for 15 minutes.
The sample is kept still for 15 minutes, then the solution is passed through filter paper.The filtrate so obtained is tested for pH by using digital pH meter.
pH of Banana peel – 5.11
pH of activated soil – 7.65
2. Electrical conductivity
Take 10 gm of sample add 100 ml distilled water in it.The sample is transferred to bottle and kept for shaking at 200 rpm for 15 minutes. The sample is kept still for 15 minutes, then the solution is passed through filter paper.The filtrate so obtained is tested for electrical conductivity by using electrical conductivity meter.
Conductivity of banana peel solution – 3.302 mhos
Conductivity of activated soil solution – 495.3 micromhos
3. Specific gravity
Clean and dry the density bottle. Weigh the empty bottle with stopper(W1). Take 5 to 10 gm of soil sample which is cooled in dessicator. Transfer it to the bottle. Find the weight of the bottle and soil (W2). Now add distilled water to the bottle and weigh the bottle fille with soil and water (W3). Fill the empty bottle with water and weigh it as (W4)
Specific gravity of soil sample is 2.62
4. Bulk Density
Specific gravity bottle of 25 ml capacity without the stopper was weighed.Then it is filled with the sample upto the edge of the neck tapping the bottle up to 20 times and weighed. The bulk density is obtained by dividing the weight of the sample with volume of the sample.
Bulk density of soil sample is 1.39 gm/ml
5. FTIR Analysis
a. Sample- Banana peels
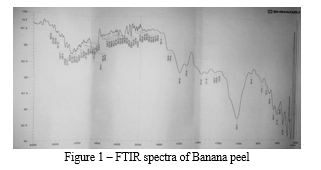
FTIR spectroscopy was performed to characterize the chemical functional groupsof banana peels. These spectra were obtained from scanning in the range of 500- 4000 cm-1. The band of banana peel shifted to 3662.94cm-1,to3018.70cm-1 indicating that primary and secondary amines and amides (stretch) groups are involved and also involve H-bounded, carboxylic acids. From the spectra, the results indicate that aldehydes, esters, hydrocarbons, nitro group alkene, aromatic and alkyne are also observed.
C. Procedure of work
- Study of pH
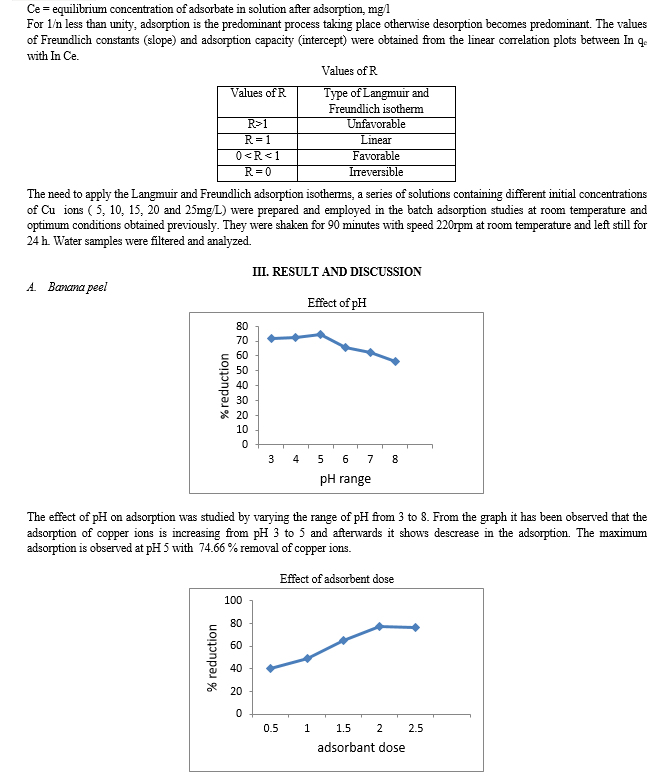
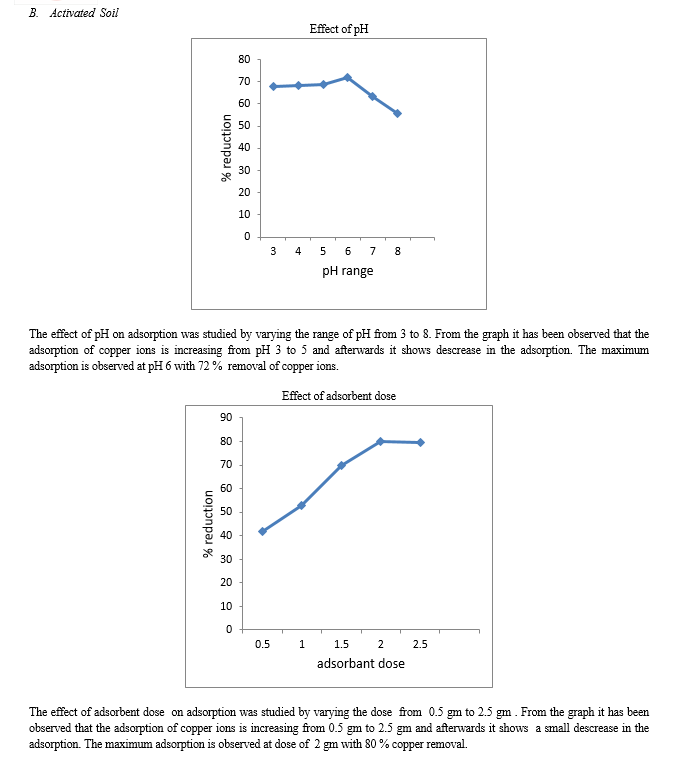
To study the effect of pH on Cu(II) adsorption onto banana peel and activated soil were kept constant at 2g/100 ml, and varying the pH of the samples using 0.1N HCl and 0.1N NaOH between (4 to 8). Each of Cu(II) concentrations were 5 mg/l for all solutions during the experiment. Adsorption studies were carried out at room temperature. The samples were shaken for 90 minute with 220 rpm (this speed was able to mix and reaches to equilibrium), at room in the 125 ml bottle volume and left still for 24 hours. Optimum pH value helps to make the study of effect of other factors, more easier.
2. Study of Adsorbent
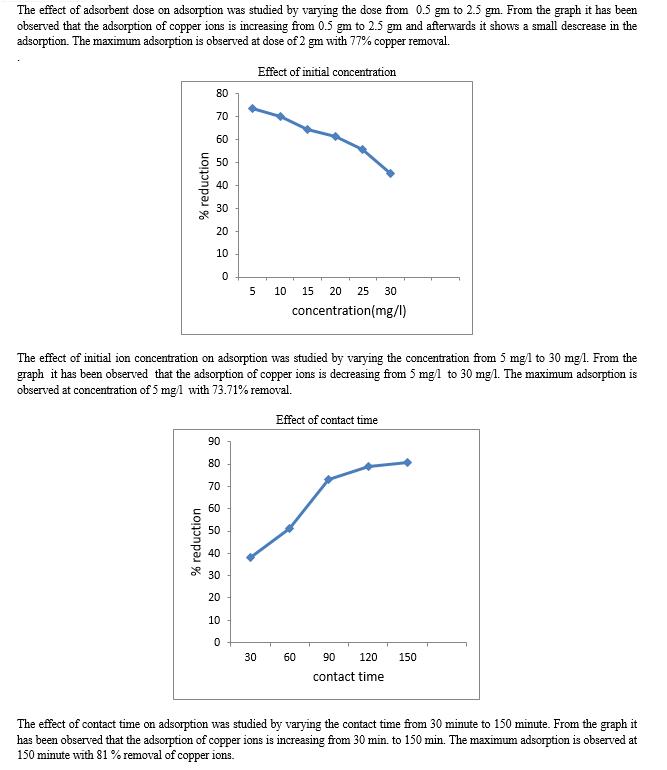
The impact of the adsorbent amount on the equilibrium adsorption for each of Cu(II) were investigated with banana peels and activated soil of 0.5,1,1.5, 2 and 2.5g in five sets of 100 ml water, which contained 5 mg/l of Cu(II) concentrations each, at pH 5.The samples were shaken at speed 220 rpm at room temperature for 90 minutes and were left for 24 hours before the water samples filtered and analyzed in terms of Cu(II) and . The same procedure was done with the activated soil as an adsorbent.
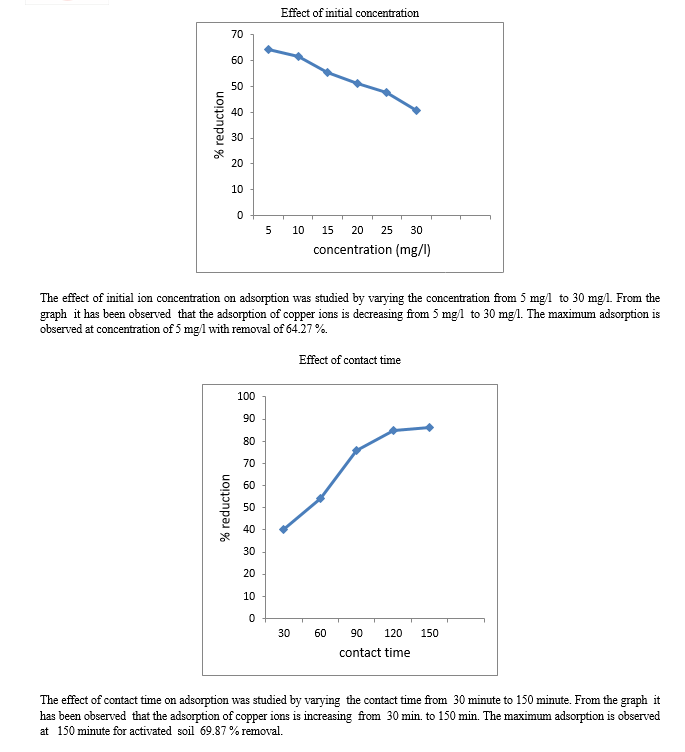
3. Study of Initial Concentration
The impact of the initial concentration on the equilibrium adsorption for each of Cu (II) were investigated with banana peels and activated soil of 5, 10, 15, 20, 25 mg/l in five sets of 100 ml water at pH 5. The samples were shaken at 220 rpm at room temperature for 90 minutes and were left for 24 hours before the water samples filtered and analysed in terms of Cu(II) and the same procedure was done with the activated soil as an adsorbent.
4. Study Of Contact Time
The impact of contact time on the equilibrium adsorbtion for Cu (II) was investigated with banana peels and activated soil for 30, 60, 90, 120 and 150 minute contact time in five sets of 100 ml , which contained 5 mg/l of Cu (II) concentration at pH 5. The samples were shaken at 220 rpm at room temperature for 90 minutes and were left for 24 hours before the water sample filtered and analysed in terms of Cu(II) and the same procedure was done with the activated soil.
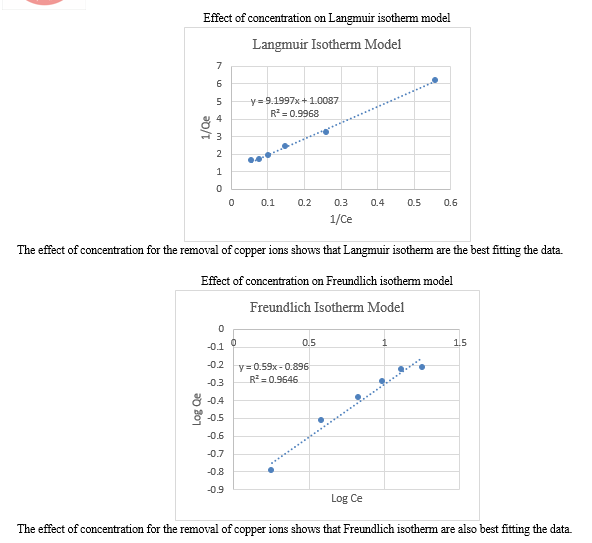
5. Study of Equilibrium Isotherm Models
The two models were used to fit the experimental data namely Langmuir model and Freundlich model. Langmuir and Freundlich model were used to describe the adsorption equilibrium.
a. Langmuir Isotherm
Langmuir proposed theory to describe the adsorption of gas molecules onto metal surfaces. The Langmuir adsorption isotherm has found successful application to many other real adsorption processes. The Langmuir isotherm is based on assuming monolayer sorption onto surfaces onto surface with fixed number of well defined sites. The form of Langmuir isotherm is expressed as below

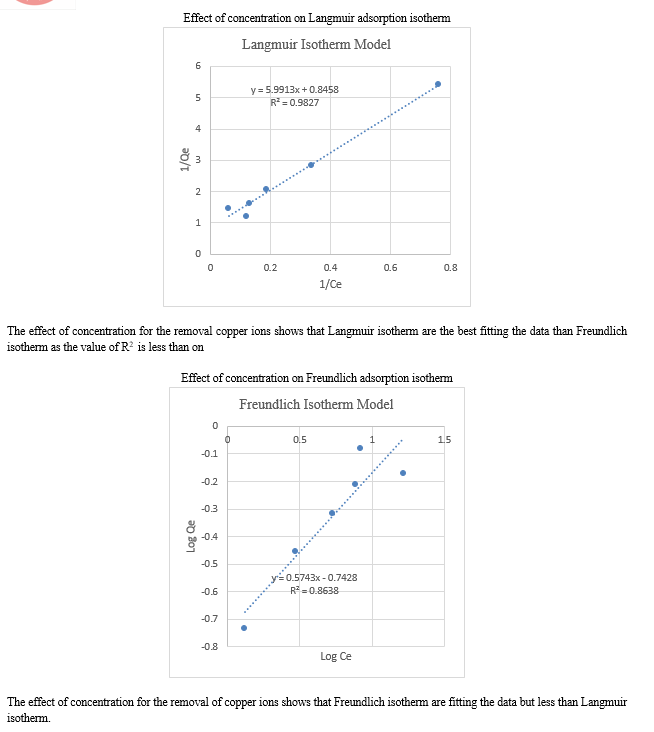
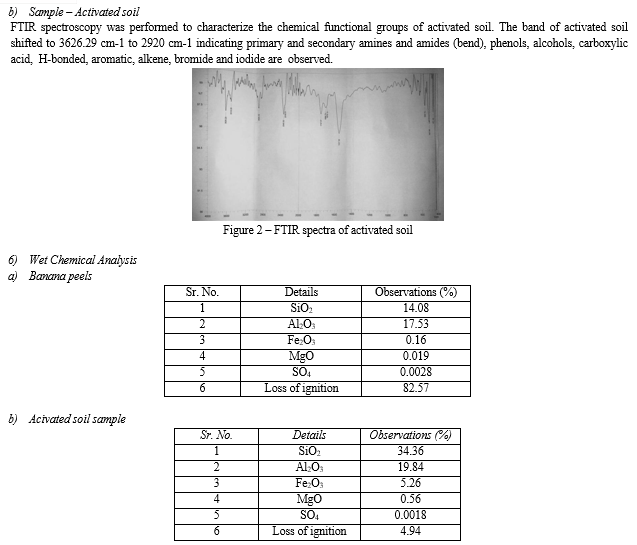
Conclusion
1) Banana peels and activated soil are easily available and natural materials for preparing adsorbent. This study has explored the economically viable adsorbents for copper removal from water. 2) The pH is an important parameter, which affects the adsorption process. Maximum percentage removal of metal ion is observed at a pH of 5 for banana peels and 6 for activated soil. 3) The increase of adsorbent dosage had resulted in the increase of percentage removal, the maximum percentage removal was at 2 g for both banana peels and activated soil. 4) The percentage removal increases with increase in contact time which is maximum at 150 minute for both adsorbents. 5) The adsorption was decreased with increasing initial metal ions concentration from 5 mg/l to 30 mg/l.
References
[1] ‘Christian G. Daughton’ (2017), “Monitoring wastewater for assessing community health: Sewage Chemical Information Mining (SCIM)” (Science of the Total Environment), pp. 748-764. [2] ‘Nidaa Adel Jasem’ (2015), “Removal of Copper (II) and Cadmium (II) ions from aqueous solutions using Banana Peels and Bentonite Clay as Adsorbents” (Journal of Engineering and Development,) , ISSN 1813- 7822, Vol.19, No.4, pp.49-68. [3] ‘P. Rajasulochana and V. Preethy’ (2016), “Comparison on efficiency of various techniques in treatment of waste and sewage water – A comprehensive review” pp.175-184. [4] ‘Dr. Nidhi Jain’ (2015), “Removal of heavy metal by using different fruit peels, vegetable peels and organic waste - A review” (International journal of Advanced Research) ISSN 2320-5407, Volume 3, Issue 11, pp. 916-920. [5] ‘Sabrina Wilhelm et.al’ (2017), “Does wastewater treatment plant upgrading with activated carbon result in an improvement of fish health ?” (Aquatic Toxicology) pp.184-197 [6] ‘M. A. Hossain, H. Hao Ngo, W. S. Guo and T. V. Nguyen’ (2012), “ Removal of Copper from Water by Adsorption onto Banana Peel as Bioadsorbent” (International Journal of Geomate ), ISSN:2186-2982(P), 2186-2990(O), Vol. 2, No. 2, pp. 227-234. [7] ‘Sana Chaoua, Samia Boussaa, Abdelhay El Gharmali and Ali Boumezzough’ (2018), “Impact of irrigation with wastewater on accumulation of heavy metals in soil and crops in the region of Marrakech in Morocco” (Journal of the Saudi Society of Agricultural Sciences), pp. 1-8 [8] ‘Jimoh Abdulfatai , Abdulkareem Ambali Saka , Ayo Samuel Afolabi , and Onazi Micheal’(2016)“Development of Adsorbent from Banana Peel for Wastewater Treatment” (Applied Mechanics and Materials) Vol. 248, pp 310-315 [9] ‘Gunatilake S.K.’ (2015) “Methods of Removing Heavy Metals from Industrial Wastewater”(Journal of Multidisciplinary Engineering Science Studies) ISSN: 2912-1309 Vol. 1 Issue 1, pp 12-18 [10] ‘Khairia M. Al-Qahtani’ (2015) “Water purification using different waste fruit cortexes for the removal of heavy metals” (Journal of Taibah University for Science) , pp 700-708 [11] ‘Sabino De Gisi, Giusy Lofrano, Mariangela Grassi,Michele Notarnicola’(2016) “Characteristics and adsorption capacities of low-cost sorbents for wastewater treatment: A review” (Sustainable materials and technologies ) pp 10-40 [12] ‘Suresh Sundaramurthy, Suantak Kamsonlian, C.B. Majumder and S.Chand (2011) “Characterization of banana and orange peels: Biosorption mechanism” IISN: 2229-6646 vol. 4, issue 2, pp 1-7 [13] ‘Bonnie Ransom Stern, Marc Solioz, Daniel Krewski’ (2007) “Copper and Human health: Biochemistry, Genetics, andStrategies for modeling dose-response relationships” (Journal of Toxicology and Environmental Health) pp157-222. [14] ‘Magdalena Araya, Manuel Olivares and Fernando Pizarro’ (2007) “Copper in Human heath” (International Journal on Environment and Health) Vol. 1, No. 4, pp 608-220. [15] ‘Sana Chaoua , Samia Boussaa , Abdelhay El Gharmali , Ali Boumezzough’ (2018) “Impact of irrigation with wastewater on accumulation of heavy metals in soil and crops in the region of Marrakech in Morocco” (Journal of the Saudi Society of Agricultural Sciences) pp 1-8 [16] ‘Renata S. D. Castro,La_ercio Caetano,Guilherme Ferreira’ (2011) “Banana Peel Applied to the Solid Phase Extraction of Copper and Lead from River Water: Preconcentration of Metal Ions with a Fruit Waste” pp 3446-3451 [17] ‘Lutfullah, Saurabh Sharma,Nafisur Rahman, Syed Najmul Hejaz Azmi’(2010) “UV Spectrophotometric Determination of Cu(II) in Synthetic Mixture and Water Samples” ( Journal of the Chinese Chemical Society) pp 622-631. [18] ‘S.Krupanidhi, Arun Sreekumarand C.B. Sanjeevi’ (2008) “Copper & biological health” pp 448-461 [19] ‘Saurabh Sharma, Nasifur Rehman , Syed Najmul Hejaz Azmi and Bashir Iqbal’ (2018) “UV Spectrophotometric Determination of Cu(II) in Synthetic Mixture and Water Samples” (Journal of Chinese Chemical Society) Vol. 57 No.4A, pp 622-631 [20] ‘Sebahat Seker and Bnu Kutlu’ (2014) “Determination of Copper (Cu) Levels for Rivers in Tunceli, Turkey” (World Environment) pp 168-171 [21] ‘U.Rafique and S.Nazir’(2013) “Heavy Metal Removal from Wastewater using Environment Friendly Bio-materials: A Cost Effective and Eco-friendly Technique” (International Journal of Chemical and Environmental Engineering ) Vol. 2 no. 4, pp 90-99. [22] ‘P.Bavani and B.Sujata’(2014)“Impact Of Toxic Metals Leading To Environmental Pollution” (Journal of Chemical and Pharmaceutical Sciences) IISN : 0974 -2115, pp 70-72. [23] ‘Arjuman Banu and Dr. Nagrani Santhankrishnan’ (2016) “Preparation of Low Cost Activated Carbon Adsorbents from Natural Sources” ISSN 2394 – 3386 Vol. 3 Issue 4, pp 43-46. Books [1] ‘Introduction to Spectroscopy’ by Donald L. Pavia, Gary M. Lampman, George S. Kriz and James R. Vyvyan [2] ‘Standard Methods for the Examination of Water and Wastewater’ by American Public Health Association, American Water Works Association and Water Environment Federation
Copyright
Copyright © 2023 Deokar G. N., Desale L. R, Harkal S. P. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET51068
Publish Date : 2023-04-26
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

