Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Green Synthesis of Schiff Bases by Using Natural Acid Catalysts
Authors: Gangadhar B. Gundlewad
DOI Link: https://doi.org/10.22214/ijraset.2022.45263
Certificate: View Certificate
Abstract
Schiff bases have been synthesized by the condensation of primary aryl amine with acetophenones under solvent free condition. The reaction is catalyzed by natural acid found unripe mango. This reaction is very simple and has many benefits because of cheap catalyst, good yield of product, simple experimental conditions and easily available natural products. The products were identified by various spectroscopic techniques.
Introduction
I. INTRODUCTION
This document is a template. Schiff bases are condensation products of primary amines with carbonyl compounds and they were first reported by Schiff in 1864. The common structural feature of these compounds is the azomethine group with a general formula RHC=N-R1, where R and R1 are alkyl, aryl, cyclo alkyl or heterocyclic groups which may be variously substituted. These compounds are also known as anils, imines or azomethines. Several studies2 showed that the presence of a lone pair of electrons in an sp2 hybridized orbital of nitrogen atom of the azomethine group is of considerable chemical and biological importance. Because of the relative easiness of preparation, synthetic flexibility, and the special property of C=N group, Schiff bases are generally excellent chelating agents,[6-12] especially when a functional group like –OH or –SH is present close to the azomethine group so as to form a five or six membered ring with the metal ion. Versatility of Schiff base ligands and biological, analytical and industrial applications of their complexes make further investigations in this area highly desirable. Schiff bases have been known since 1864 when Hugo Schiff reported the condensation of primary amines with carbonyl compounds. Nowadays, the research field dealing with Schiff base coordination chemistry has expanded enormously. The importance of Schiff base complexes for bioinorganic chemistry, biomedical applications, supramolecular chemistry, catalysis and material science, separation and encapsulation processes, and formation of compounds with unusual properties and structures has been well recognized and reviewed2. Schiff bases resulted from aromatic aldehydes ortho-substituted with a hydroxyl group have initially arouse the researchers' interest because of their ability to act as bidentate ligands for transitional metal ions3-7. Later, in studies concerning quantitative structure-antitumor activity relationship of a series of Schiff bases derived from variously substituted aromatic amines and aldehydes, it has been shown that azomethines from salicylaldehydes gave the best correlation8,9. Schiff bases of salicylaldehydes have also been reported as plant growth regulators10 and antimicrobian11 or antimycotic12 activity. Schiff bases also show some analytical applications13. Schiff Bases are characterized by the -N=CH- (imine) group which imports in elucidating the mechanism of transamination and rasemination reaction in biological system14,15. Schiff bases are active against a wide range of organisms for example; Candida Albicans, Escherichia coli Staphylococcus aureus, Bacillus polymxa, Trychophyton gypseum, Mycobacteria, Erysiphe graminis and Plasmopora viticola. A large number of different Schiff base ligands have been used as cation carriers in potentiometric sensors as they have shown excellent selectivity, sensitivity, and stability for specific metal ions such as Ag(II), Al(III), Co(II), Cu(II), Gd(III), Hg(II), Ni(II), Pb(II), Y(III), and Zn(II) [29-34]. Schiff bases have been studied for their important properties in catalysis [35]. They show catalytic activity in hydrogenation of olefins16. They find applications in biomimetic catalytic reactions. An interesting application of Schiff bases is their use as an effective corrosion inhibitor, which is based on their ability to spontaneously form a monolayer on the surface to be protected. Many commercial inhibitors include aldehydes or amines, but presumably due to the C=N bond the Schiff bases function more efficiently in many cases17. The principal interaction between the inhibitor and the metal surface is chemisorption18. The inhibitor molecule should have centers capable of forming bonds with the metal surface by electron transfer. In such cases the metal acts as electrophiles and the inhibitor acts as a Lewis base. Nucleophilic centers, such as oxygen and nitrogen atoms, of the protective compound have free electron pairs which are readily available for sharing. Together with the atoms of the benzene rings they create multiple absorption sites for the inhibitor thus enabling stable monolayer formation. Imines also have biological importance. An imine linkage between the aldehyde derived from vitamin A and the protein opsin in the retina of the eye plays an important role in the chemistry of vision.
Vitamins are also called coenzymes, meaning that they are to the functioning of many enzymes, which are large proteins that catalyze chemical changes in cell. An example of a biologically important aldehyde is pyridoxal phosphate, which is the active form of the vitamin B6. Vitamin B6 serves as a coenzyme by forming an imine with an amino acid grouping an enzyme. The coenzyme, bound to the enzyme, is involved in transamination reaction, the transfer of the amino group from one amino acid to another, which is important in the metabolism and the biosynthesis of amino acids. In the last step, enzyme-catalyzed hydrolysis cleaves the imine to pyridoxal and the modified amino acid. Schiff bases have been reported in their biological properties, such as, antibacterial, antifungal activities. Their metal complexes have been widely studied because they have anticancer and herbicidal applications. They serve as models for biologically important species. O-phenylenediamine Schiff bases show clinical properties. Isatin Schiff bases were reported to possess antiviral, anti-HIV, antiprotozoal and anthelmintic activities. They also exhibit significant anticonvulsant activity, apart from other pharmacological properties. Certain cobalt Schiff base complexes are potent antiviral agents. Schiff bases derived from 4-dimethylamine benzaldehyde shows antibacterial activity. In medicine these are used as antibodies and anti-inflammatory agents. This paper presents a series of new Schiff bases resulted from the natural acid (Unripen mango extract) catalyzed condensation of aryl ketones with aromatic amines.
II. MATERIALS AND METHODS
All the chemicals were of analytical grade and used as received from the supplier. All the melting points were determined by open capillary methods and are uncorrected. All the synthesized compounds were purified by simple crystallization. The purity of compounds was checked by TLC.
The EIMS (Electron impact mass spectrometry) were scanned on a Jeol-JMS HX-110 mass spectrometer. The 1H-NMR (Nuclear magnetic resonance) spectra and 13C NMR Spectra were recorded on a Bruker spectrometer operating at 500 MHz. The chemical shift values are reported in δ (ppm) relative to TMS (Tetra methyl silane) as internal standard.
General procedure for the synthesis of Schiff base: The synthesis of Schiff base is described as a representative example: A mixture of Acetophenone (10 m.mol) and Aniline (10 m.mol) and 0.5 mL mango extract was stirred at room temperature for 1 -2 hour with monitoring by TLC. After completion of reaction the reaction mixture was poured on ice crystals the solid product was collected by filtration and purified by recrystallization with ethanol.
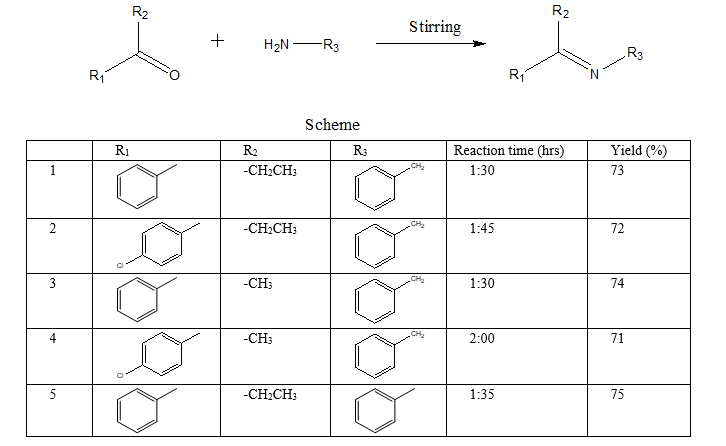
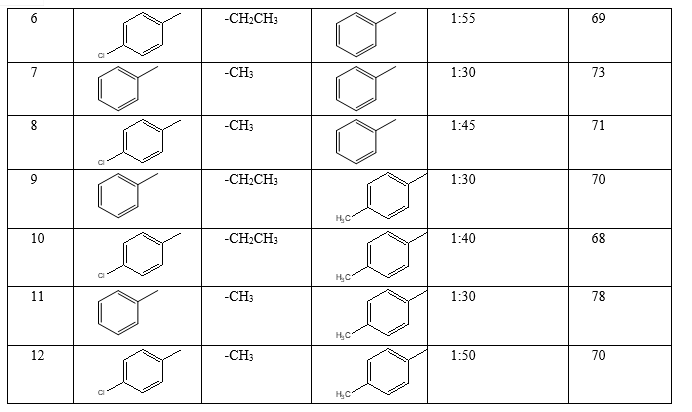
III. RESULTS AND DISCUSSION
It was observed that Schiff bases have been synthesized by the condensation of aromatic ketones with arylamines. In the condensation reaction first step is the protonation of carbonyl oxygen. In these method natural acids found in unripe mango extract was used as a catalyst to protonate carbonyl oxygen and under solvent free condition good yield was obtained.
(E)-N-(1-phenylpropylidene)benzenamine (1) Yellow solid M. P. 121oC 1HNMR (CDCl3): δ 0.9 (t 3H); 1.4 (q 2H); 7.29 (t 2H); 7.3 (m 5H); 7.31 (t 1H); 7.62 (d 2H); 13CNMR (CDCl3): δ 8.1; 21.4; 122.3; 127.3; 128.6; 129.2; 130.1; 131.1; 134.0; 153.2; 164.6; Mass (m/z) M+ 209 Anal. Calc.(Found) for C15H15N, C- 86.08% (86.08%); H-7.22% (7.29%); N-6.64% (6.67%).
(E)-N-(1-(4-chlorophenyl)propylidene)benzenamine (2) Yellow solid M. P. 131oC 1HNMR (CDCl3): δ 0.92 (t 3H); 1.5 (q 2H); 7.30 (d 2H); 7.39 (m 5H); 7.56 (d 2H); 13CNMR (CDCl3): δ 8.1; 21.4; 122.6; 127.6; 129.0; 130.3; 131.1; 132.1; 136.6; 153.2; 164.6; Mass (m/z) M+ 243 Anal. Calc.(Found) for C15H14ClN, C- 73.92% (73.95%); H-5.79% (5.78%); N-5.75% (5.72%).
(E)-N-(1-phenylethylidene)benzenamine (3) Yellow solid M. P. 127oC 1HNMR (CDCl3): δ 0.92 (s 3H); 7.3 (m 5H); 7.29 (m 3H); 7.62 (d 2H); 13CNMR (CDCl3): δ 16.0; 122.3; 127.3; 128.9; 129.3; 130.1; 131.2; 134.2; 153.2; 165.2; Mass (m/z) M+ 195 Anal. Calc.(Found) for C14H13N, C- 86.12% (86.08%); H-6.71% (6.69%); N-7.17% (7.17%).
(E)-N-(1-p-tolylethylidene)benzenamine (4) Yellow solid M. P. 111oC 1HNMR (CDCl3): δ 0.92 (s 3H); 2.35 (s 3H); 7.09 (d 2H); 7.3 (m 5H); 7.50 (d 2H); 13CNMR (CDCl3): δ 16.2; 24.3; 122.3; 127.3; 129.2; 129.9; 130.1; 131.0; 140.7; 153.2; 165.2; Mass (m/z) M+ 209 Anal. Calc.(Found) for C15H15N, C- 86.08% (86.09%); H-7.22% (7.23%); N-6.69% (6.7%).
(E)-phenyl-N-(1-phenylpropylidene)methanamine (5) Yellow solid M. P. 141oC 1HNMR (CDCl3): δ 0.9 (t 3H); 1.4 (q 2H); 4.81 (s 2H); 7.06 (d 2H); 7.09 (t 1H); 7.14 (t 2H); 7.19 (d 1H); 7.29 (t 2H); 7.62 (d 2H); 13CNMR (CDCl3): δ 8.1; 21.8; 58.2; 125.8; 128.7; 128.9; 129.1; 129.3; 131.2; 138.9; 140.1; 171.1; Mass (m/z) M+ 223 Anal. Calc.(Found) for C16H17N, C- 86.05% (86.08%); H-7.76% (6.75%); N-6.27% (6.25%).
(E)-N-(1-(4-chlorophenyl)propylidene)(phenyl)methanamine (6) Yellow solid M. P. 124oC 1HNMR (CDCl3): δ 0.92 (s 3H); 1.4 (q 2H); 4.81 (s 2H); 7.06 (d 2H); 7.07 (t 1H); 7.14 (d 2H) 7.3 (d 2H); 7.56 (d 2H); 13CNMR (CDCl3): δ 8.1; 21.8; 58.2; 126.8; 128.7; 129.0; 129.1; 130.6; 136.6; 138.2; 171.1; Mass (m/z) M+ 257 Anal. Calc.(Found) for C16H16ClN, C- 74.55% (74.58%); H-6.26% (6.29%); N-5.43% (5.43%).
(E)-phenyl-N-(1-phenylethylidene)methanamine (7) Yellow solid M. P. 132oC 1HNMR (CDCl3): δ 0.9 (s 3H); 4.18 (s 2H); 7.06 (d 2H); 7.1 (t 1H); 7.14 (d 2H); 7.29 (m 3H); 7.62 (d 2H); 13CNMR (CDCl3): δ 16.4; 57.9; 125.8; 128.7; 128.9; 129.1; 129.2; 131.2; 138.9; 140.1; 164.6; Mass (m/z) M+ 209 Anal. Calc.(Found) for C15H15N, C- 86.08% (86.08%); H-7.22% (7.21%); N-6.69% (6.67%).
(E)-phenyl-N-(1-p-tolylethylidene)methanamine (8) Yellow solid M. P. 133oC 1HNMR (CDCl3): δ 0.92 (s 3H); 2.35 (s 3H); 4.81 (s 2H); 7.06 (d 2H); 7.09 (t 1H); 7.19 (d 2H); 7.24 (d 2H); 7.50 (d 2H); 13CNMR (CDCl3): δ 16.4; 23.4; 57.9; 125.8; 128.7; 129.1; 129.2; 137.1; 138.9; 140.7; 164.6; Mass (m/z) M+ 223 Anal. Calc.(Found) for C16H17N, C- 86.05% (86.08%); H-7.67% (7.61%); N-6.69% (6.67%).
(E)-4-methyl-N-(1-phenylpropylidene)benzenamine (9) Yellow solid M. P. 125oC 1HNMR (CDCl3): δ 0.93 (s 3H); 1.4 (q 2H); 2.35 (s 3H); 7.1 (m 4H); 7.29 (m 3H); 7.62 (d 2H); 13CNMR (CDCl3): δ 8.1; 21.4; 24.3; 122.2; 128.9; 129.2; 130.4; 131.2; 134.0; 136.3; 150.2; 164.6; Mass (m/z) M+ 223 Anal. Calc.(Found) for C16H17N, C- 86.05% (86.08%); H-7.67% (7.66%); N-6.27% (6.27%).
(E)-N-(1-(4-chlorophenyl)propylidene)-4-methylbenzenamine (10) Yellow solid M. P. 132oC 1HNMR (CDCl3): δ 0.9 (s 3H); 1.4 (q 2H); 2.25 (s 3H); 7.1 (m 4H); 7.3 (d 2H); 7.56 (d 2H); 13CNMR (CDCl3): δ 8.1; 21.4; 24.3; 122.2; 129.0; 130.4; 130.6; 132.1; 134.6; 136.6; 136.9; 150.2; Mass (m/z) M+ 257 Anal. Calc.(Found) for C16H16ClN, C- 74.55% (74.56%); H-6.62% (6.61%); N-5.43% (5.47%).
(E)-4-methyl-N-(1-phenylethylidene)benzenamine (11) Yellow solid M. P. 133oC 1HNMR (CDCl3): δ 0.9 (s 3H); 2.35 (s 3H); 7.1 (m 4H); 7.26 (d 2H); 7.29 (m 3H); 13CNMR (CDCl3): δ 16.2; 24.9; 122.5; 128.5; 129.3; 130.8; 132.1; 134.0; 136.4; 150.2; 165.2; Mass (m/z) M+ 209 Anal. Calc.(Found) for C15H15N, C- 86.08% (86.06%); H-7.22% (7.23%); N-6.69% (6.71%).
(E)-4-methyl-N-(1-p-tolylethylidene)benzenamine (12) Yellow solid M. P. 127oC 1HNMR (CDCl3): δ 0.9 (s 3H); 1.91 (s 3H); 2.35 (s 3H); 7.09 (d 2H); 7.2 (m 4H); 7.5 (d 2H); 13CNMR (CDCl3): δ 16.0; 18.3; 24.3; 122.2; 128.8; 129.2; 130.4; 131.0; 136.9; 140.7; 150.2; 165.2; Mass (m/z) M+ 232 Anal. Calc.(Found) for C16H17N, C- 86.5% (86.06%); H-7.67% (7.66%); N-6.27% (6.24%).
Conclusion
The present study revealed that Schiff bases can simply be synthesized by natural acids found in natural products. This environmental friendly method is easily approachable and gives good yield.
References
[1] Bendale, A.R., Bhatt, R., Nagar, A., Jadhav, A.G. and Vidyasagar, G. (2011). Der Pharma Chemica. 3(2): 34-38. [2] Cates, A.L. and Rasheed, S.M. (1984). Pharm. Res. 6: 271- 273. [3] Gupta, J.K., Biplab, D.E. and Saravanan, V. (2006). Indian J. Chem. 42B, 2580-2582. [4] Hadjipavlou-litina, D.J. and Geronikaki, A.A. (1996). Drug Des. Discov.15: 199-206. [5] Hossein, N., Salimi, F. and Rabiei, K. (2006). J. Mol. Catal. 260: 100-104. [6] WAHAB ET AL (2014), FUUAST J. BIOL., 4(1): 27-32 32 [7] Imrie, C., Kleyi, P., Nyamori, V.O., Gerber, T.I.A., Levendis, D.C. and Look, J. (2007). J. Organomet. Chem. 692: 3443-3453. [8] Kshash, A.H., Mohammed, O., Ezzat. and Zaid, K.A. (2011). Baghdad Journal for Sciences 8(3):711-716. [9] Lee, S.K., Zakaria, H., Chuyng, H.L., Kuyengl, L., Games, E.J.C., Mehta, R.J., Kinghorn, D. and Pezzuto, J.M. (1998). Combinatorial Chemistry and High Throughput Screening 1: 35-40. [10] Majed, M.H. (2009). E-Journal of Chemistry 6(3): 629-632. [11] Patil, S., Jadhav, M.B. and Deshmukh. (2011). Arch. Appl. Sci. Res. 3(1): 203-208. [12] Perez, C., Paul, M. and Bazerque, P. (2009). Acta Biol. Med. Exp. 15: 113-115. [13] Popp, F.D. (1961). J. Org. Chem. 26: 1566-1568. [14] Popenoe, W. (1974). Manual of Tropical and Subtropical fruits. Hafner press 432-436. [15] Shah, S., Vyas, R. and Mehta, R.J. (1992). J. Indian Chem. Soci., 69: 590 596. [16] Sherwani, S.K., Nazim, K., Khan, T.M., Ahmed, M., Malik, M.W., Noor, A.A., Khan, M.U., Ali, Q.M. and Alam, S.I. (2012). FUUAST J. Biol., 2(2): 65-68. [17] Sherwani, S.K., Bokhari, T.Z., Nazim, K., Gilani, S.A. and Urooj, S.K. (2013). IRJP, 4(7): 83-86. [18] Solak, N. and Rollas, S. (2006). Arkivoc. 12: 173-181. [19] Verma, M., Pandeya, S.N., Singh, K.N. and Sable, J.P. (2004). Acta Pharm. 54: 49-56.
Copyright
Copyright © 2022 Gangadhar B. Gundlewad. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET45263
Publish Date : 2022-07-03
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

