Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- References
- Copyright
Inside the Oral Microbiome: How Periodontitis May Increase the Risk for Alzheimers Disease
Authors: Benjamin Bowman
DOI Link: https://doi.org/10.22214/ijraset.2021.39378
Certificate: View Certificate
Abstract
The oral microbiome is the site of the second most abundant microbiota after the gastrointestinal tract. The expanded Human Oral Microbiome Database (eHOMD) that was last updated on November 22, 2017, contains the information of a number of bacterial species, scientists have struggled to categorize and understand every species due to their difficulty of laboratory study. While this much is known, the scientific community still has minimal knowledge of the entire oral microbiome and how it may relate to potential manifestations of various diseases. Periodontitis is one disease that is directly related to the oral microbiome. Periodontitis is the infection of the gums that is associated with poor dental hygiene. This oral malady has been hypothesized to be a potential precursor to neurological Alzheimer’s disease (AD.) Since AD is difficult to study until postmortem, there has been limited availability to study pathways where this may occur. The proposed method by which periodontitis, while primarily hypothesized, includes a shift in the microbiota of the oral cavity to a more pathogenic state. The shift to pathogenicity is mainly believed to be due to an increase in the bacterium Porphyromonas gingivalis. P. gingivalis produces protein plaques on the teeth that can travel through the bloodstream to the brain. The proposed mechanism is hypothesized to weaken the blood-brain barrier and allow for plaque aggregation on the brain seen in AD-infected brains. While more research is necessary to conclude the pathogenesis from periodontitis to Alzheimer’s disease definitively, strides are being made that may help give scientists, and healthcare workers begin preventative measures.
Introduction
I. INTRODUCTION
A. Oral Microbiome
The oral microbiome is the second most abundant microbiota after the gastrointestinal tract. According to a study, a healthy human body contains roughly 3.8 x 1013 bacterial and other microorganismal cells, thus making up the human microbiome (Sender et al. 2016). Narrowing in on the oral microbiome specifically, there has been ever-expanding data regarding the inhabitants of the human oral cavity. Since researcher Antonie van Leeuwenhoek first observed dental plaques under a microscope roughly 400 years ago, and J.K. Clarke isolated the first bacterium in 1924, the scientific community has characterized and isolated approximately 772 prokaryotic species in the oral cavity. While no doubt it is impressive that these species of bacteria have been discovered, nearly 30% of them have not been cultivated due to limitations of either knowledge or appropriate conditions. Of the currently 70% cultivable prokaryotic species, 57% have been completed (Verma et al. 2018). This knowledge gap lends itself to an expansive and untouched field of human health that may lead to advances in preventative and curative medicine. The bacteria that inhabit the oral cavity have been proven to have significant interaction with the host cells. These interactions provide solid and direct influences upon the host, ranging from physiological, metabolic, and immune responses (Bik et al. 2006). For the oral microbiome, the preferred targets of these bacteria include hard and soft palates, sub-/supragingival surfaces, teeth, lips, cheeks, and tonsils (Paster et al. 2001). Though the sites of colonization are primarily indirectly associated with the oral cavity, the implications are far beyond the limitations of oral maladies. Dangerous diseases such as diabetes, bacteremia, cancer, AIDS, endocarditis, and autoimmune diseases all display alterations to their bacterial communities compared to the oral cavity of a healthy individual (Verma et al. 2018).
B. Periodontitis
Periodontitis is a highly prevalent inflammatory gum tissue/oral disease that affects nearly 50% of the population worldwide. Defined as gum disease, periodontitis can lead to tooth loss, the breakdown of soft tissue that makes up the oral cavity, and even jawbone degradation. Periodontitis etiology is bacterial. Specifically, these caustic bacteria accumulate on the dental surfaces and inside the periodontal pockets, where they form a complex biofilm that is commonly known as plaque (Dioguardi et al. 2020). There are a handful of known culprits that are believed to be specific periodontal pathogens. These include A. actinomycetemcomitans, P. gingivalis, T. forsythia, T. denticola, P. intermedia, F. nucleatum, C. rectus, etc. (Dioguardi et al. 2020). Though it is not entirely clear how periodontitis might lead to the manifestation of Alzheimer’s disease, there are several hypothesized pathways that include P. gingivalis.
C. Alzheimer’s Disease
Known as a neurodegenerative disorder, Alzheimer’s disease (AD) affects millions of people worldwide, with a rapidly rising frequency as life expectancy increases and the world population becomes older (Liccardo et al. 2020). AD possesses a recurrent sign common of neurodegenerative pathologies represented by dementia, defined as “a relevant or complete loss of intellectual capacities, beginning gradually and progressively, differentially caused in previously normal patients” (Dioguardi et al. 2020). Neurodegenerative diseases such as AD are classified into two groups: 1. Primary or idiopathic degenerative dementia. 2. Dementia in the presence of other neurodegenerative pathologies. Since this analysis focuses on AD, it is essential to note that roughly half of dementia patients relate to AD, with nearly 25% of the elderly patients affected (Dioguardi et al. 2020).
Aside from age being a factor, another critical risk factor is heredity. Pathogenic mutations are seen in the amyloid precursor protein (APP), and the presence of presenilin (I and II) have a 50% increment of the onset risk (Dioguardi et al. 2020). Since AD etiology is not one hundred percent understood, more of the proposed mechanisms are hypothesized. These hypotheses include histopathologic alterations of senile plaques, neurofibrillary tangles, and neural cells caused by amyloid precipitation (Dioguardi et al. 2020). For this review, the focus will primarily be on the role of P. gingivalis and its associated amyloid protein aggregates.
One of the most probable mechanisms by which AD may progress is via the alteration of APP metabolism, which results in an increased deposition of a harmful glycoprotein known as β-amyloid (Hunter and Brayne. 2018). β-amyloid deposition is neurotoxic due to the variation in calcium homeostasis and other factors (Dioguardi et al. 2020). Additionally, these aggregates of plaques seen in the brains of AD patients are dangerous and cause impairments to the functionality of the human brain.
II. PROPOSED MECHANISM BY WHICH P. GINGIVALIS MAY LEAD TO ALZHEIMER’S DISEASE
P. gingivalis and its association with the onset of AD comes from several studies examining the bacterium’s inflammatory properties against microglia. This is mediated by the lipopolysaccharide (LPS) of the microbe. In periodontitis, it is apparent that the large majority of bacteria are the early colonizing gram-positive, followed by the late colonizing gram-negative bacteria (Harding et al. 2017). The P. gingivalis infection, amongst several other periodontitis-associated bacteria, proliferate under conditions of poor oral hygiene. As these bacteria proliferate, they increase the anaerobic environment in the dental plaque. While the environment becomes more and more pathogenic due to this bacterial proliferation, toxic factors are released (Harding et al. 2017). In the case of P. gingivalis, it is hypothesized that the bacterium subverts neutrophilic function, inhibiting phagocytosis, resulting in an uptick in cytokine production and recruitment of inflammatory immune responses (Harding et al. 2017). This sounding of the inflammatory alarm exacerbates the inflammatory response of the environment.
This inflammatory theory essentially claims that the β-amyloids precipitation could be caused by inflammatory processes (Ho et al. 2005). Induction of neurodegeneration via neuroinflammation is proposed to be caused by the presence of periodontitis-associated bacteria and the associated self-triggering of the host’s inflammatory process in the central nervous system (Ho et al. 2005).
While this seems to be a reasonably localized event, it is far from it. Periodontitis-associated inflammation leads to systemic inflammation due to the infiltration of bacteria and their associated virulence factors into the host’s bloodstream (Harding et al. 2017). Moreover, while it has long been known that the blood-brain barrier (BBB) is an immunoprotected environment, it is now believed that system inflammation from periodontitis may lead to impairment of the BBB due to the role of a lipid metabolizing protein essential in neurobiology called ApoE protein (Harding et al. 2017). The mechanism by which P. gingivalis and associated amyloid plaques can progress from the mouth to the brain is still hypothesized, though it is a central focus of the relationship. The method proposed explains that an infection in the gingival pocket leads to the activation of the NF-kB signaling pathway leading to inflammasome activation. Once activated, the bacterium is able to pass through the gingival pocket into the host’s bloodstream, resulting in bacteremia. Once in the blood, more cytokine recruitment is engaged, which leaves the BBB susceptible to becoming leaky (Hafezi-Moghadam et al. 2007). This mechanism of bacterial-BBB interaction where the LPS of bacterium is able to increase BBB permeability is seen in previously done studies in mice (Dioguardi et al. 2020). From here, the bacteria are able to access the brain and develop a biofilm that we know as plaque. Additional support was seen in a study done by Zhan et al. showing that peripheral injection of LPS in mice causes microglia activation, inducing the release of pro-inflammatory cytokines (Zhan et al. 2018).
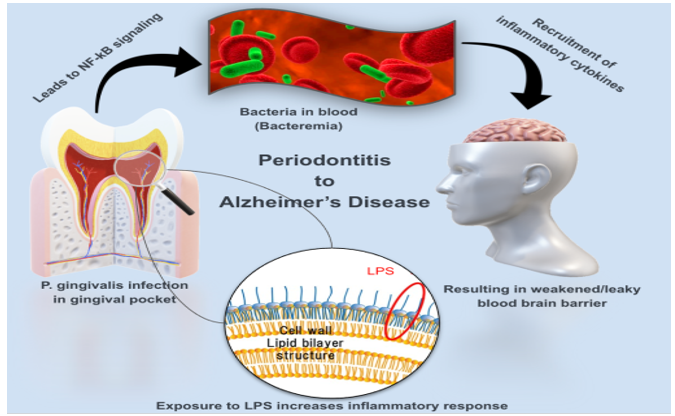
Figure 1: Periodontitis has been loosely linked to Alzheimer’s disease (AD.) In this figure, we can see a representation of the role a P. gingivalis infection may have on the progression of AD. Peripheral inflammation caused by the bacteria results in signaling of a number of pro inflammatory cytokines. IL-1beta, IL-6 and TNF-alpha are all associated with the weakening of the blood brain barrier. Bacteria entering the bloodstream via the oral cavity exacerbate the condition and provide a route of access to the brain. A degraded blood brain barrier allows the brain to become susceptible to amyloids and infection that may cause Alzheimer’s disease. The bacterium’s LPS contributes to the systemic inflammation over the course of exposure.
Pro-inflammatory intermediates themselves have limited access to the brain and CNS. However, once they reach the CNS via the previously proposed method, they can activate endothelial cells in the brain. This would induce cytokines and other factors that eventually induce glial cells (Dioguardi et al. 2020). If the neurodegenerative process has already begun, then bacterial products already present in the brain can increase already high levels of cytokines.
The results of this proposed method have been shown in several studies where LPS co-localized with amyloid proteins in plaque inside the brain of a person with AD. Additionally, LPS, the main component in gram-negative bacteria, can be found in abundance in the brains of those with AD (Harding et al. 2017).
III. EVIDENCE OF THE ROLE P. GINGIVALIS HAS ON THE ETIOPATHOGENESIS OF ALZHEIMER’S DISEASE
A number of studies have shown that periodontitis correlates with cognitive impairments. In 2017, a study conducted by Wu et al. found a strong association between LPS administered to cathepsin B-dependent mice. Cathepsin B (CatB) is a lysosomal protease involved in the breakdown of proteins. CatB is known to play an integral role in the mediation of neuroinflammation and beta-amyloid deposition (Dioguardi et al. 2020). The hypothesis by Wu et al. was that CatB would promote the production of interleukins and amyloids from microglial cells. Over the course of Wu et al.’s 5-week study, chronic exposure to LPS induced a memory deficit in middle-aged mice. Of note, there was no memory deficit in the CatB- mice, only significant increases in CatB+ mice. This suggests that the memory deficit was primarily due to systemic exposure of LPS, as suggested by the previously proposed method of bacteremia (Wu et al. 2017). The targeting of CatB as a therapeutic target for its role in the connection of periodontitis and AD is suggested by both Wu and in another study done in 2019 by Nie et al. Nie et al. also shows the P. gingivalis infection induces the production of beta-amyloid through CatB/NF-kB pathways.
Stein et al. studied antibody levels of seven periodontal pathogens in 158 subjects. Of the 158 participants, 81 developed cognitive decline. Comparing the antibody levels of those with cognitive decline to those with cognitive abilities intact, Stein et al. were able to find that those who have developed AD have a higher antibody response than those without AD. Stein et al.’s study provide data demonstrating that high antibody responses against the bacteria seen in periodontitis may potentially contribute to the risk of AD onset (Stein et al. 2012).
Furthermore, and finally, a bioinformatics study researched 78 genes associated with AD by relating them to periodontitis-associated pathogens, including P. gingivalis (Carter. 2017). One such study found that there was a possible association between periodontal disease caused by P. gingivalis and AD. The study concluded that a return to a healthy oral microbiome might be able to arrest, delay, or possibly even provide remission in patients with AD (Carter. 2017).
IV. REAL-WORLD IMPACTS AND DISCUSSION
The development of mechanisms, while at this point still hypothesized, have massive implications on the future of both periodontitis and Alzheimer’s disease. If it holds true that bacteria such as P. gingivalis are potential culprits when it comes to the manifestation of AD, then scientists may be well on their way to discovering not only preventative measures but potentially treatments for the devastating disease. If evidence continues to grow indicating that plaques that are on teeth are potentially related to the plaques seen in the brains of AD patients, it would potentially be transformative in the prevention and treatment of this devastating disease.
As a society, there are already well-established reasons for dental hygiene, but this could show us additional importance on dental health. P. gingivalis is a bacterium that we can get a better understanding of with the proper measures. Handwashing and brushing teeth twice daily can keep the oral microbiome at healthy levels. And while there are additional risk factors for both periodontitis and AD, the potential for eliminating some level of risk would have both humanistic and financial benefits to families as well as the global healthcare infrastructure. When scientists compile the entirety of the research it also lends itself to the potential studying of new drugs and treatments for the number of illnesses not limited to AD. Healthcare could potentially tackle, or at the very least look into associated dementia class diseases to see if the mechanisms are similar. This could lead to treatments for a number of maladies that plague humans. Overall, the findings of P. gingivalis and its relationship with both periodontitis and AD is an exciting discovery that may have massive impacts on AD progression.
References
[1] Carter, C.J., 2017. Genetic, Transcriptome, Proteomic, and Epidemiological Evidence for Blood-Brain Barrier Disruption and Polymicrobial Brain Invasion as Determinant Factors in Alzheimer’s Disease. ADR 1, 125–157. https://doi.org/10.3233/ADR-170017 [2] Dioguardi, M., Di Gioia, G., Caloro, G.A., Capocasale, G., Zhurakivska, K., Troiano, G., Lo Russo, L., Lo Muzio, L., 2019. The Association between Tooth Loss and Alzheimer’s Disease: a Systematic Review with Meta-Analysis of Case Control Studies. Dentistry Journal 7, 49. https://doi.org/10.3390/dj7020049 [3] Hafezi-Moghadam A, Thomas KL, Wagner DD. ApoE deficiency leads to a progressive age-dependent blood-brain barrier leakage. Am J Physiol Cell Physiol. 2007 Apr;292(4):C1256-62. doi: 10.1152/ajpcell.00563.2005. [4] Harding, A., Gonder, U., Robinson, S.J., Crean, S., Singhrao, S.K., 2017. Exploring the Association between Alzheimer’s Disease, Oral Health, Microbial Endocrinology and Nutrition. Front. Aging Neurosci. 9, 398. https://doi.org/10.3389/fnagi.2017.00398 [5] Ho, G.J., Drego, R., Hakimian, E., Masliah, E., 2005. Mechanisms of cell signaling and inflammation in Alzheimer’s disease. Curr Drug Targets Inflamm Allergy 4, 247–256. https://doi.org/10.2174/1568010053586237 [6] Hunter, S., Brayne, C., 2018. Understanding the roles of mutations in the amyloid precursor protein in Alzheimer disease. Mol Psychiatry 23, 81–93. https://doi.org/10.1038/mp.2017.218 [7] Liccardo, D., Marzano, F., Carraturo, F., Guida, M., Femminella, G.D., Bencivenga, L., Agrimi, J., Addonizio, A., Melino, I., Valletta, A., Rengo, C., Ferrara, N., Rengo, G., Cannavo, A., 2020. Potential Bidirectional Relationship Between Periodontitis and Alzheimer’s Disease. Front. Physiol. 11, 683. https://doi.org/10.3389/fphys.2020.00683 [8] Nie, R., Wu, Z., Ni, J., Zeng, F., Yu, W., Zhang, Y., Kadowaki, T., Kashiwazaki, H., Teeling, J.L., Zhou, Y., 2019. Porphyromonas gingivalis Infection Induces Amyloid-? Accumulation in Monocytes/Macrophages. JAD 72, 479–494. https://doi.org/10.3233/JAD-190298 [9] Paster, B.J., Boches, S.K., Galvin, J.L., Ericson, R.E., Lau, C.N., Levanos, V.A., Sahasrabudhe, A., Dewhirst, F.E., 2001. Bacterial Diversity in Human Subgingival Plaque. J Bacteriol 183, 3770–3783. https://doi.org/10.1128/JB.183.12.3770-3783.2001 [10] Sender R, Fuchs S, Milo R. Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol. 2016 Aug 19;14(8):e1002533. doi: 10.1371/journal.pbio.1002533. PMID: 27541692; PMCID: PMC4991899. [11] Sparks Stein, P., Steffen, M.J., Smith, C., Jicha, G., Ebersole, J.L., Abner, E., Dawson, D., 2012. Serum antibodies to periodontal pathogens are a risk factor for Alzheimer’s disease. Alzheimer’s & Dementia 8, 196–203. https://doi.org/10.1016/j.jalz.2011.04.006 [12] Wu, Z., Ni, J., Liu, Y., Teeling, J.L., Takayama, F., Collcutt, A., Ibbett, P., Nakanishi, H., 2017. Cathepsin B plays a critical role in inducing Alzheimer’s disease-like phenotypes following chronic systemic exposure to lipopolysaccharide from Porphyromonas gingivalis in mice. Brain, Behavior, and Immunity 65, 350–361. https://doi.org/10.1016/j.bbi.2017.06.002 [13] Zhang, J., Wu, Y., Zhang, Y., LeRoith, D., Bernlohr, D.A., Chen, X., 2008. The Role of Lipocalin 2 in the Regulation of Inflammation in Adipocytes and Macrophages. Molecular Endocrinology 22, 1416–1426. https://doi.org/10.1210/me.2007-0420
Copyright
Copyright © 2022 Benjamin Bowman. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET39378
Publish Date : 2021-12-11
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

