Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Insilico Bimolecular Interaction Studies of Natural Compounds for Treatment of Alzheimers Disease
Authors: Kumar Blesso, V Shristi Sharma, Mitali V Shetty
DOI Link: https://doi.org/10.22214/ijraset.2022.46599
Certificate: View Certificate
Abstract
To determine the natural composites that can be used as implicit medicines towards Alzheimer’s complaint. Alzheimer’s complaint is a nonstop progressive and multi-factored neurodegenerative complaint that\'s known to be the sixth leading cause of death and the most common cause of madness in old, aged people. Alzheimer’s complaint is related to increased situations of the amyloid ? peptide( A?) along with loss of neurons and synapses. The complaint process is largely associated with amyloid pillars, neurofibrillary befuddlements, and loss of neuronal connections in the brain. In our study, we will be exercising the structural and natural exertion information on specific ligands and compactly study the vital targets that are believed to be effective against Alzheimer’s complaint. The early onset of the domestic Alzheimer\'s complaint is considered to do due to the mutations in one of three genes those garbling amyloid- beta precursor protein( APP) and Presenilins PSEN1 and PSEN2. utmost mutations in the APP and presenilin genes increase the product of the small proteins known as the amyloid beta( A?) 42 proteins, are the main element of the forming amyloid pillars. There are numerous selective targets for the event of anti-AD( Alzheimer’s complaint) medicines, and thus the multi-factorial nature of this complaint involves multi-target-directed composites which may be salutary for announcement treatment. The complaint which is known to target the hippocampus that is associated with our memory, further this becomes responsible to instigate the first symptoms of memory impairment. As the complaint progresses so does the degree of memory impairment. The natural emulsion models were considered as multi-target leads using molecular docking ways and also completely analysed grounded on the list affinity values, RMSD values, stability and molecular interactivity studies for the determination of implicit medicine composites to make medicinals. Eventually, the foremost implicit multi-target natural composites against Alzheimer’s complaint are determined and farther validated through the corresponding invitro studies.
Introduction
I. INTRODUCTION
Alzheimer’s sickness could be a progressive neurological disorder that causes the brain to shrink (atrophy) and brain cells to die. Alzheimer’s sickness is that the commonest reason for dementedness — never-ending decline in thinking, activity and social skills that affects a person’s ability to perform severally. close to five.8 million individuals inside the North American nation age sixty five and older settle for Alzheimer’s sickness. Of those, eightieth ar seventy five years recent and older. Out of the close to fifty million individuals worldwide with dementedness, between hour and seventieth ar calculable to own Alzheimer’s sickness. the first signs of the sickness embody forgetting recent events or conversations. because the sickness progresses, an individual with Alzheimer’s sickness can develop severe memory impairment and lose the flexibility to hold out everyday tasks. the event of the sickness takes place slowly bit by bit like phases.[49]
AD begins slowly. It first involves the parts of the brain that control thought, memory, and language. People with AD may have trouble remembering things that happened recently or names of individuals they know. A related problem, mild cognitive impairment (MCI), causes more memory problems than normal for people of an equivalent age. Many, but not all, people with MCI will develop AD. In AD, over time, symptoms worsen.
People might not recognize relations. they'll have trouble speaking, reading, or writing. they'll forget the way to brush their teeth or comb their hair. Later, they'll become anxious or aggressive, or stray from home. Eventually, they have total care. this will cause great stress for relations who must look after them. AD usually begins after age 60 and the danger goes up as you grow older. Our risk is additionally higher if a loved one has had the disease.
Most AD cases are sporadic and present considerable heterogeneity in terms of risk factor profiles and neuropathological features. First-degree relatives of Alzheimer patients have a higher lifetime risk of developing AD than the general population or relatives of nondemented individuals both genetic and shared environmental factors contribute to the phenomenon of familial aggregation.
II. METHODOLOGIES
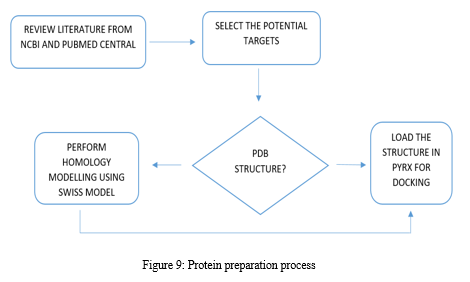
A. Protein Model Preparation For Docking Studies.
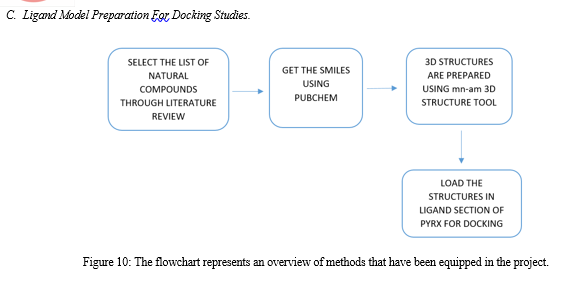
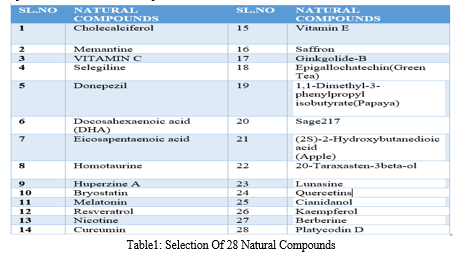
Relevant literature for Alzheimer's disease is collected from pubmed and PMC in the NCBI Database in order to identify the target protein for the disease, the pathways a compound may take to reduce the effects of the disease and accordingly determine the potential compounds. After sufficient literature collection and referencing, the main target protein was determined as “Secretase”, ????-Amyloid A4 protein and Glycogen Synthase Kinase 3(GSK3) along with other β-amyloid peptide pathways from Uniprot. The compounds along with their smile structures along with their isomeric smiles were then tabulated for further molecular docking studies using Pyrx software,wherein about 28 natural compounds as listed in Table 1 were taken for conduction of studies. The PDBE structural model of the target proteins of concern-Secretase,-Amyloid A4 protein and Glycogen Synthase Kinase 3(GSK3) in humans was first retrieved from the Swiss Model online server and the macromolecule structure was then loaded on Pyrx tool.
One must note that the water molecules are not involved in binding, and they must be eliminated before the conduction of any molecular docking and binding studies to make the computations easier and clear the binding pocket of possible water molecules that would distort the pose search. The water molecules and irrelevant chains must be eliminated, depending upon the area of study from the notepad sequence and only then the molecule must be loaded for efficient docking. The ligands of the 3D conformation ligands of the required natural compounds are downloaded from pubchem and then loaded into Pyrx software to carry out molecular docking with respect to Secretase,-Amyloid A4 protein and Glycogen Synthase Kinase 3(GSK3).The Binding affinities consisting of the ligands containing the lower and upper RMSD values as zero are prioritised as shown in Table2. The docked structures of the target protein and the chosen ligands are then saved in PDB format and analysed for their stability towards different conditions of temperature and pressure, reactivity, binding affinity to enable efficient action on the target site for the noted important proteins and minimize effects of Alzheimer's condition.
Several factors concerning Adsorption, Distribution, Metabolism, Excretion and Toxicity[ADMET] are also analysed utilising the pkcsm online web server. Ligands of natural compounds having a binding affinity of -7.0 and above are considered to have potential in targeting the required sites of the target proteins that have earlier been determined using the Cast-p online tool. The IGEMDOCK Software is utilized to obtain the 3D structures of the ligand-protein interactions and the results are now analysed depending upon the ligand binding capacity, hydrogen bonding and hence a crystal structure with reasonable resolution and binding affinity that have a slightly consensus profile in terms of binding residues (if possible) should be the top criteria. The binding residues (not the ligand functional group) biochemical and cellular activity is encoded by the interactions (allosteric/competitive or agonist/antagonist) from such domains/pocket and hence are key determining factors.
B. Protein Model Preparation For Docking Studies.
III. RESULTS AND DISCUSSION
A. Selection Of The Target Proteins And Natural Compounds.
- Target Proteins: Target proteins play a very important role in the study of the disease and computational drug designs. Target proteins control the action and therefore the kinetic behaviour of medicine within the organism. The elucidation of structure, conformational signalling and catalytic properties of particular target proteins facilitates a rational design of medicine and biotechnological processes. The following table lets us look into the set of target proteins chosen for this project and studies.

2. ???? Secretase: Ectodomain shedding of the amyloid precursor protein (APP) by the metalloprotease activity α-secretase may be a key regulatory event preventing the generation of the Alzheimer's disease (AD) amyloid β peptide. Proteases almost like α-secretase are essential for diverse physiological processes, like embryonic development, cell adhesion and neuronal guidance. Previously, several proteases were suggested as candidate α-secretases for APP, especially members of the ADAM family (a disintegrin and metalloprotease). Two recent studies analyzed primary neurons, which are the cell type affected in AD, and eventually demonstrated that the constitutively cleaving α-secretase activity is selectively mediated by ADAM10. An increase in α-secretase cleavage is taken into account a therapeutic approach for AD. However, the molecular mechanisms regulating α-secretase cleavage remain only partly understood. Signaling pathways activating protein kinase C and MAP kinase play a central role in stimulating α-secretase cleavage of APP. Additionally, several recent publications demonstrate that ADAM10 expression and α-secretase cleavage of APP are tightly controlled at the extent of transcription, e.g. by retinoic acid receptors and sirtuins, and at the extent of translation and protein trafficking. This review focuses on the recent progress made in unravelling the molecular identity, regulation and therapeutic potential of α-secretase in Alzheimer's disease. [52]
3. Glycogen Synthase Kinase 3: Glycogen synthase kinase (GSK)-3 has been proposed because the link between the 2 histopathological hallmarks of Alzheimer's disease , the extracellular senile plaques composed of beta-amyloid and therefore the intracellular neurofibrillary tangles formed from hyperphosphorylated tau. Thus, GSK-3 is one among the most tau kinases and it modifies several sites of the tau protein present in neurofibrillary tangles. Furthermore, GSK-3 is in a position to modulate the generation of amyloid-beta, also on answer this peptide. In several transgenic models, overexpression of GSK-3 has been related to neuronal death, tau hyperphosphorylation and a decline in cognitive performance. Lithium, a widely used drug for affective disorders, inhibits GSK-3 at therapeutically relevant concentrations and it's been demonstrated that this is often ready to prevent tau phosphorylation. In the present review, we summarize all these data and discuss the potential of GSK-3 inhibitors for Alzheimer's disease therapy, as well as some of their potential problems. [52]
4. Amyloid ???? peptide: The beta amyloid (A beta) protein may be a key molecule within the pathogenesis of Alzheimer's disease (AD). The tendency of the A beta peptide to aggregate, its reported neurotoxicity, and genetic linkage studies, have led to a hypothesis of AD pathogenesis that a lot of AD researchers term the amyloid cascade hypothesis. In this hypothesis, an increased production of A beta leads to neurodegeneration and ultimately dementia through a cascade of events. In the past 15 years, debate amongst AD researchers has arisen on whether A beta may be a cause or an impact of the pathogenic process. Recent in vitro and in vivo research has consolidated the idea that A beta is that the primary cause, initiating secondary events, culminating within the neuropathological hallmarks related to AD. This research has led to the event of therapeutic agents, currently in human clinical trials, which target A beta.
B. Natural Compounds
A natural product (NP) is usually defined as a compound or substance that's produced by living organisms. NPs are often classified by many criteria and characteristics, like source, biological function, biosynthetic pathway, physical and chemical properties, etc. Nowadays, NPs find a broad spectrum of applications associated with human life, including a crucial role in medicine. Notably, the utilization of natural products as medicines has been described throughout human history in terms of drugs associated with herbal medicines, potions, oils, remedies, and foods. Many of those substances are discovered by trial and error, and through the years they need become standard products in human lives. In the past few decades, natural products are a crucial source of potential drug hits and leads. However, development efforts in NP drug discovery have demonstrated a particular downturn in recent years. Despite this decline, the vast chemical space of natural products continues to supply abundant structural diversity for locating novel lead compounds with low relative molecular mass but 10% of the world’s biodiversity has been explored to seek out potential biologically active compounds. Therefore, numerous lead NPs which will be used against a broad spectrum of diseases await discovery. Many drugs associated with diseases like malaria, tuberculosis, cancer, human immunodeficiency virus (HIV), inflammatory diseases, cardiac diseases, diabetes, etc. are derived from NPs. One area where natural compounds have also been found to possess great research potential and application is neurodegenerative diseases like Alzheimer’s disease (AD), Parkinson’s disease (PD), schizophrenia, dementia, and neuropathic disorders.
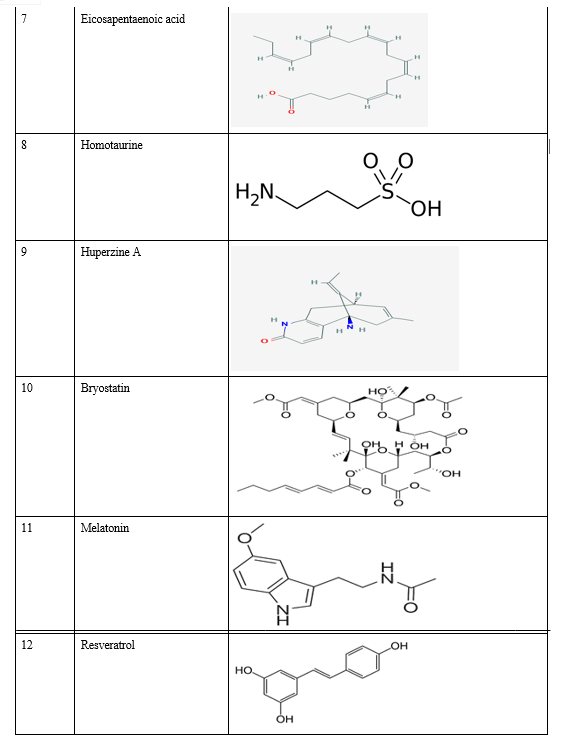
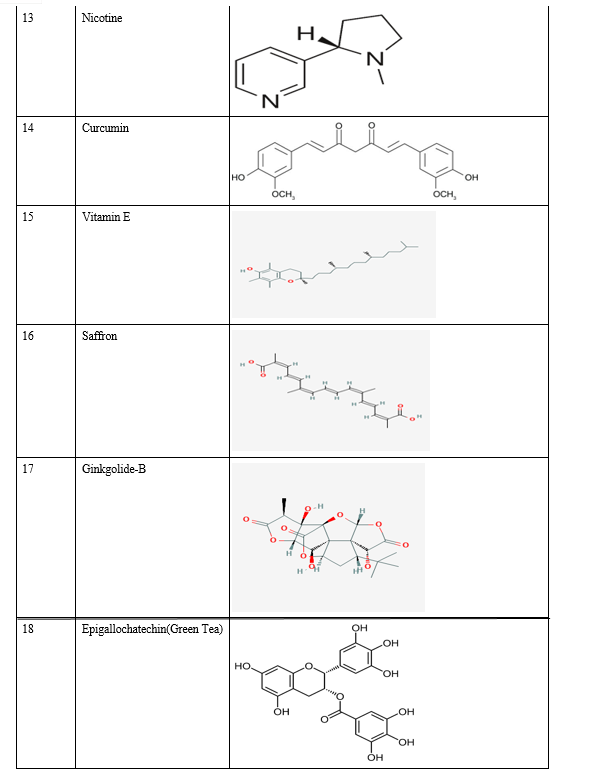
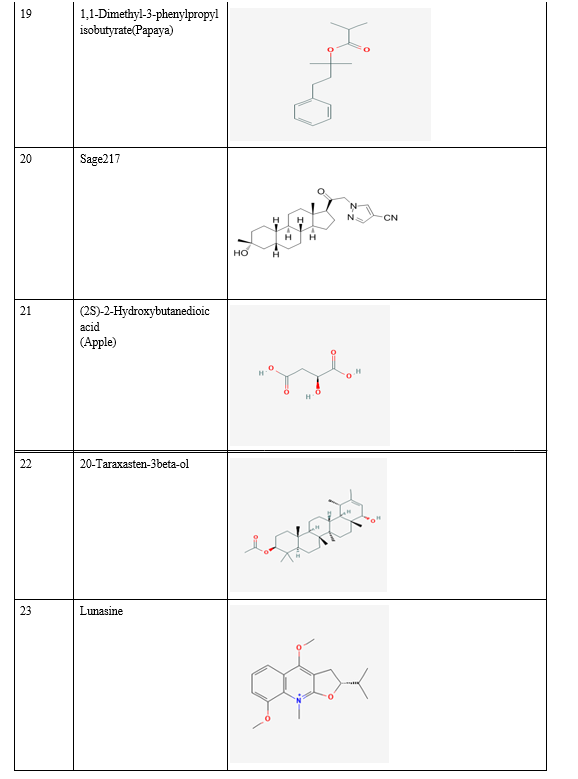
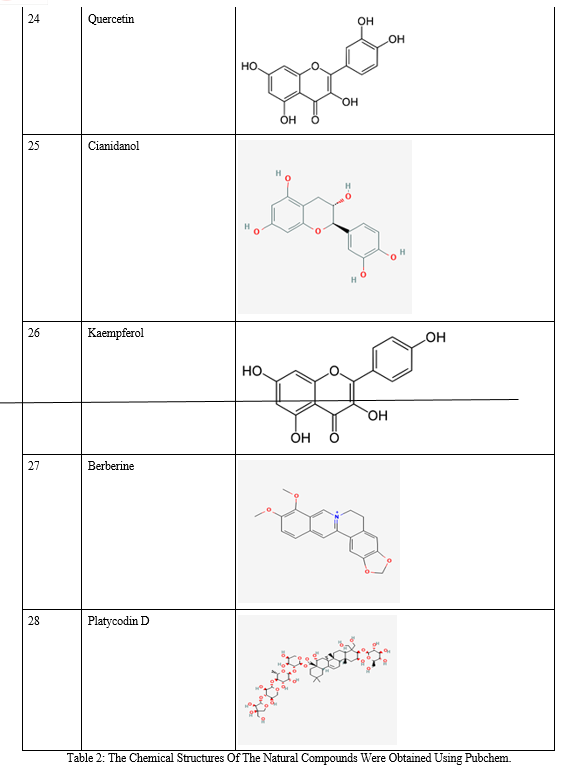

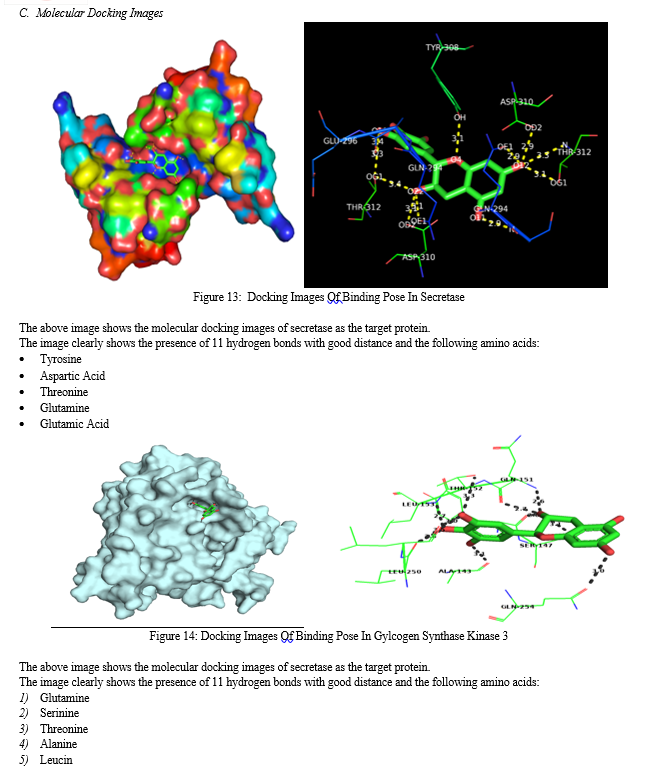
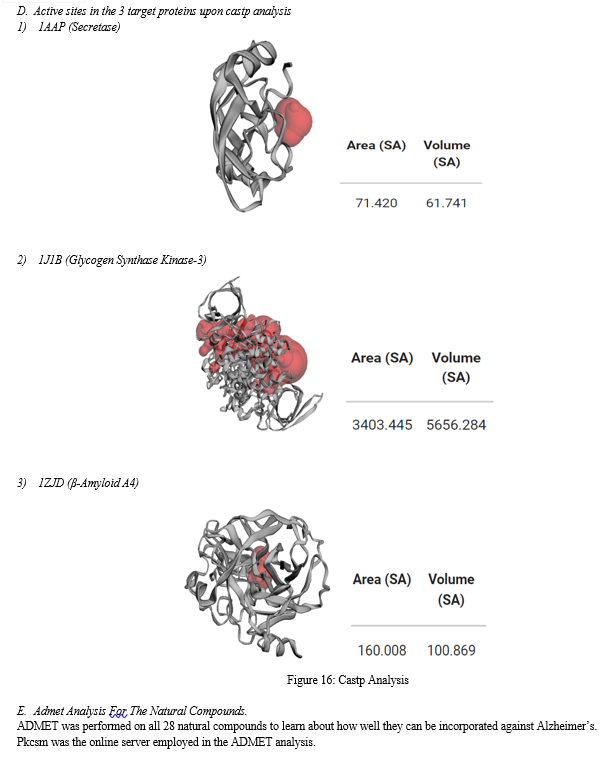
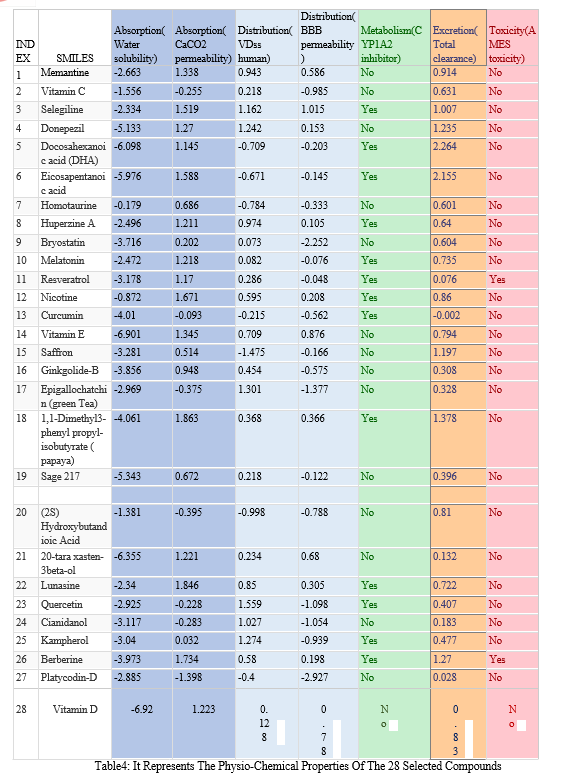
Conclusion
The 3D structure of the three target proteins was elucidated and the active sites of three target proteins was predicted. The 3D structures of the ligand molecules were analysed. Docking results showed that two natural compound Epigallochatechin (EGCC) and 20-taraxasten-3beta-ol are best potential ligands against 3 target proteins with the binding energy of - Therefore these two are best lead molecules to reduce the effects of Alzheimer’s disease after further clinical trials. Pharmacology dynamics through ADMET substantiates the conclusion drawn from our studies.
References
[1] Blennow K, Mattsson N, Schöll M, Hansson O, Zetterberg H. Amyloid biomarkers in Alzheimer’s disease. Trends Pharmacol Sci. 2015;36(5):287–309. [2] Bajda M, Guzior N, Ignasik M, Malawska B. Multi-target-directed ligands in Alzheimer’s disease treatment. Curr Med Chem. 2011;18(32):4949–75. [3] Rathore N, Ramani SR, Pantua H, Payandeh J, Bhangale T, Wuster A, et al. Paired Immunoglobulin-like Type 2 Receptor Alpha G78R variant alters ligand binding and confers protection to Alzheimer’s disease. PLoS Genet. 2018;14(11):e1007427. [4] Drugtargetreview.com.[cited 2021 Jul 14]. Available from: https://www.drugtargetreview.com/news/59528/novel-ligands-developed-with-high-affinity-for-alzheimers-receptors/ [5] Ni R, Gillberg P-G, Bergfors A, Marutle A, Nordberg A. Amyloid tracers detect multiple binding sites in Alzheimer’s disease brain tissue. Brain. 2013;136(Pt 7):2217–27. [6] Unzeta M, Esteban G, Bolea I, Fogel WA, Ramsay RR, Youdim MBH, et al. Multi-Target directed donepezil-like ligands for Alzheimer’s disease. Front Neurosci. 2016;10:205. [7] Ambure P, Bhat J, Puzyn T, Roy K. Identifying natural compounds as multi-target-directed ligands against Alzheimer’s disease: an in silico approach. J Biomol Struct Dyn. 2019;37(5):1282–306. [8] Mori H, Takio K, Ogawara M, Selkoe DJ. Mass spectrometry of purified amyloid beta protein in Alzheimer’s disease. J Biol Chem. 1992;267(24):17082–6. [9] Corey-Bloom J. Galantamine: a review of its use in Alzheimer’s disease and vascular dementia. Int J Clin Pract. 2003;57(3):219–23. [10] Clarke ID, Dirks PB. A human brain tumor-derived PDGFR-alpha deletion mutant is transforming. Oncogene. 2003;22(5):722–33. [11] Lagah S, Tan I-L, Radhakrishnan P, Hirst RA, Ward JH, O’Callaghan C, et al. RHPS4 G-quadruplex ligand induces anti-proliferative effects in brain tumor cells. PLoS One. 2014;9(1):e86187. [12] Blennow K, Mattsson N, Schöll M, Hansson O, Zetterberg H. Amyloid biomarkers in Alzheimer’s disease. Trends Pharmacol Sci. 2015;36(5):287–309. [13] Bajda M, Guzior N, Ignasik M, Malawska B. Multi-target-directed ligands in Alzheimer’s disease treatment. Curr Med Chem. 2011;18(32):4949–75. [14] Rathore N, Ramani SR, Pantua H, Payandeh J, Bhangale T, Wuster A, et al. Paired Immunoglobulin-like Type 2 Receptor Alpha G78R variant alters ligand binding and confers protection to Alzheimer’s disease. PLoS Genet. 2018;14(11):e1007427 [15] Drugtargetreview.com. [cited 2021 Jul 28]. Available from: https://www.drugtargetreview.com/news/59528/novel-ligands-developed-with-high-affinity-for-alzheimers-receptors/ [16] Ni R, Gillberg P-G, Bergfors A, Marutle A, Nordberg A. Amyloid tracers detect multiple binding sites in Alzheimer’s disease brain tissue. Brain. 2013;136(Pt 7):2217–27. [17] Unzeta M, Esteban G, Bolea I, Fogel WA, Ramsay RR, Youdim MBH, et al. Multi-Target directed donepezil-like ligands for Alzheimer’s disease. Front Neurosci. 2016;10:205. [18] Ambure P, Bhat J, Puzyn T, Roy K. Identifying natural compounds as multi-target-directed ligands against Alzheimer’s disease: an in silico approach. J Biomol Struct Dyn. 2019;37(5):1282–306. [19] Kitchen DB, Decornez H, Furr JR, Bajorath J. Docking and scoring in virtual screening for drug discovery: methods and applications. Nat Rev Drug Discov. 2004;3(11):935–49. [20] Jorgensen WL. Rusting of the lock and key model for protein-ligand binding. Science. 1991;254(5034):954–5. [21] Arcon JP, Turjanski AG, Martí MA, Forli S. Biased docking for protein-ligand pose prediction. Methods Mol Biol. 2021;2266:39–72. [22] Bhandari T. Link between 2 key Alzheimer’s proteins explained [Internet]. Wustl.edu. 2018 [cited 2021 Jul 3 [23] Deshpande P, Gogia N, Singh A. Exploring the efficacy of natural products in alleviating Alzheimer’s disease. Neural Regen Res. 2019;14(8):1321–9. [24] Asai M, Iwata N, Yoshikawa A, Aizaki Y, Ishiura S, Saido TC, et al. Berberine alters the processing of Alzheimer’s amyloid precursor protein to decrease Abeta secretion. Biochem Biophys Res Commun. 2007;352(2):498–502. [25] Cai Z, Wang C, He W, Chen Y. Berberine alleviates amyloid-beta pathology in the brain of APP/PS1 transgenic mice via inhibiting ?/?-secretases activity and enhancing ?-secretases. Curr Alzheimer Res. 2018;15(11):1045–52. [26] Chen X-W, Di YM, Zhang J, Zhou Z-W, Li CG, Zhou S-F. Interaction of herbal compounds with biological targets: a case study with berberine. ScientificWorldJournal. 2012;2012:708282. [27] Khan H, Ullah H, Aschner M, Cheang WS, Akkol EK. Neuroprotective effects of quercetin in Alzheimer’s disease. Biomolecules. 2019;10(1):59. [28] Salehi B, Machin L, Monzote L, Sharifi-Rad J, Ezzat SM, Salem MA, et al. Therapeutic potential of quercetin: New insights and perspectives for human health. ACS Omega. 2020;5(20):11849–72. [29] Chen G-F, Xu T-H, Yan Y, Zhou Y-R, Jiang Y, Melcher K, et al. Amyloid beta: structure, biology and structure-based therapeutic development. Acta Pharmacol Sin. 2017;38(9):1205–35 [30] Ayaz M, Ullah F, Sadiq A, Kim MO, Ali T. Editorial: Natural products-based drugs: Potential therapeutics against Alzheimer’s disease and other neurological disorders. Front Pharmacol. 2019;10:1417. [31] Mishra S, Palanivelu K. The effect of curcumin (turmeric) on Alzheimer’s disease: An overview. Ann Indian Acad Neurol. 2008;11(1):13–9. [32] Sawda C, Moussa C, Turner RS. Resveratrol for Alzheimer’s disease: Alzheimer’s disease. Ann N Y Acad Sci. 2017;1403(1):142–9. [33] Durazzo TC, Mattsson N, Weiner MW, Alzheimer’s Disease Neuroimaging Initiative. Smoking and increased Alzheimer’s disease risk: A review of potential mechanisms. Alzheimers Dement. 2014;10(3):S122–45. [34] Ide K, Matsuoka N, Yamada H, Furushima D, Kawakami K. Effects of tea catechins on Alzheimer’s disease: Recent updates and perspectives. Molecules [Internet]. 2018;23(9). [35] Silva Dos Santos J, Gonçalves Cirino JP, de Oliveira Carvalho P, Ortega MM. The pharmacological action of kaempferol in central nervous system diseases: A review. Front Pharmacol. 2020;11:565700. [36] Huperzine A: Can it treat Alzheimer’s? [Internet]. Mayoclinic.org. 2019 [cited 2021 Jul 30]. [37] Buchete N-V, Tycko R, Hummer G. Molecular dynamics simulations of Alzheimer’s beta-amyloid protofilaments. J Mol Biol. 2005;353(4):804–21. [38] Gilles C, Ertlé S. Pharmacological models in Alzheimer’s disease research. Dialogues Clin Neurosci. 2000;2(3):247–55. [39] Rastegari A, Nadri H, Mahdavi M, Moradi A, Mirfazli SS, Edraki N, et al. Design, synthesis and anti-Alzheimer’s activity of novel 1,2,3-triazole-chromenone carboxamide derivatives. Bioorg Chem. 2019;83:391–401. [40] Schachter AS. Department of Psychiatry, Mount Sinai School of Medicine, Mount Sinai Hospital. New York, NY, USA: Mount Sinai Medical Center; [41] MacGill M. Alzheimer’s disease: Symptoms, stages, causes, and treatments [Internet]. Medicalnewstoday.com. 2020 [cited 2021 Aug 1]. [42] Alzheimer’s disease fact sheet [Internet]. Nih.gov. [cited 2021 Aug 1]. [43] Qiu C, Kivipelto M, von Strauss E. Epidemiology of Alzheimer’s disease: occurrence, determinants, and strategies toward intervention. Dialogues Clin Neurosci. 2009;11(2):111–28. [44] L. F, Strauss E., Cx Q. Epidemiology of the dementias of old age. In: Dening T, Jacoby R, Oppenheimer C, Thomas A, editors. 4th ed. New York, NY: Oxford. University Press;2008:391–406; [45] R CK, Lr H, Es Y, Zy W, A B, Dp S, et al. The malignancy of dementia. Predictors of mortality in clinically diagnosed dementia in a population survey of Shanghai. Vol. 51. 1994. [46] Helzner EP, Scarmeas N, Cosentino S, Tang MX, Schupf N, Stern Y. Survival in Alzheimer disease: a multiethnic, population-based study of incident cases. Neurology. 2008;71(19):1489–95. [47] Mehta KM, Yaffe K, Pérez-Stable EJ, Stewart A, Barnes D, Kurland BF, et al. Race/ethnic differences in AD survival in US Alzheimer’s Disease Centers. Neurology. 2008;70(14):1163–70. [48] Rc CLA, R G, Ks B, T E, Pa G, M W, et al. Risk of dementia among white and African American relatives of patients with Alzheimer disease. Vol. 287. Green; 2002. [49] Qiu C, Kivipelto M, Agüero-Torres H, Winblad B, Fratiglioni L. Risk and protective effects of the APOE gene towards Alzheimer’s disease in the Kungsholmen project: variation by age and sex. J Neurol Neurosurg Psychiatry. 2004;75(6):828–33. [50] Verdile G, Fuller S, Atwood CS, Laws SM, Gandy SE, Martins RN. The role of beta amyloid in Alzheimer’s disease: still a cause of everything or the only one who got caught? Pharmacol Res. 2004;50(4):397–409. [51] Avila J, Wandosell F, Hernández F. Role of glycogen synthase kinase-3 in Alzheimer’s disease pathogenesis and glycogen synthase kinase-3 inhibitors. Expert Rev Neurother. 2010;10(5):703–10. [52] Binkowski TA, Naghibzadeh S, Liang J. CASTp: Computed Atlas of Surface Topography of proteins. Nucleic Acids Res. 2003;31(13):3352–5.
Copyright
Copyright © 2022 Kumar Blesso, V Shristi Sharma, Mitali V Shetty. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET46599
Publish Date : 2022-09-02
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

