Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Isolation and Screening of Biosurfactant Producing Bacteria from Soil and Water for their Potential Applications
Authors: Deepak Mundrikeri, G Sanjana, Abhishek Kannur, Suman Bishnoi, Dr. Sneha Chinta, Dr. Badarinath Kulkarni, Dr. Geetanjali Kamble
DOI Link: https://doi.org/10.22214/ijraset.2022.48314
Certificate: View Certificate
Abstract
Biosurfactants are amphiphilic compounds produced on the membrane of cells which tends to reduce surface tension. This study was done in order to isolate, screen for potential biosurfactant producing bacteria and its applications in plant growth and antimicrobial activity which were isolated from soil and water contaminated with used engine oil. Mineral salt medium(MSM) was used to isolate the bacterial strains. The isolated strains were screened for biosurfactant production by microplate assay, oil spread assay, blood haemolysis, CTAB agar assay, emulsification index test . The strains were followed through biochemical tests to study their composition. The isolated strains were run through 16s rRNA sequencing. Assessment of the properties of biosurfactant showed string antimicrobial activity against S.typhi, B.subtilis, E.coli. The biosurfactant effect on plant growth was examined and it showed improved growth in plants. The antimicrobial activity and the enhancement in the plant growth can lead to a revolution in the field of agriculture.
Introduction
I. INTRODUCTION
Past few years we have been seeing crisis of oil spillages in the oceans which have been creating havoc all over the world. This has led to keen interest towards improving the biosurfactants11.
Organo-chemical synthesis is used to produce the dispersants that have been licenced and are kept in stockpiles around the world for use in the event of an oil spill11 . Surfactants are a class of compounds having a diverse and significant role in various segments of the market including petroleum industry, soaps and detergent industry, environmental pollution abatement, and even in the food and beverage industry3. The term "biosurfactants" refers to a class of modern surfactants that are derived from microorganisms and are equally diverse in terms of structure and function. They are attracting more attention due to their biodegradability and environmentally favourable qualities15.
Diverse micro-organisms are known to produce a number of surface-active agents primarily in order to adapt and grow on a variety of substrates among other natural functions1. The glycolipid class of rhamnolipids includes some of the most researched biosurfactants2.
II. MATERIALS AND METHODS
A. Sample Collection
The water sample and soil sample were collected from the surroundings of petrol bunk vidyanagar , Hubballi in a sterile 50ml tube at 10:05am. The sample was immediately stored at 4°C till usage to prevent the microbial contamination of the sample.
B. Isolation of Microbial Consortium
The samples were enriched using inoculating in sterile Mineral Salt Medium (MSM). 1 ml of sample was inoculated in 100 ml of minimal salt medium containing (in g/L): 15g NaNO3, 1.1g KCl, 1.1g NaCl, 0.00028g FeSO4.7H2O, 3.4g KH2PO4, 4.4g K2HPO4, 0.5g MgSO4.7H2O, 0.5g yeast extract at 37°C in shaker incubator (100 rpm). The medium was incubated for 24hrs and were selected based on the colony morphology on the nutrient agar and the selected microbes were gram stained (Table no.1) . The selected isolates were screened for the production of biosurfactants using the following screening methods9.
C. Screening of Biosurfactant Producing Isolates
Bacteria were cultivated aerobically in a 500 ml Erlenmeyer flask with 100 ml mineral salt media comprising 1.0 K2HPO4, 0.2 MgSO4.7H2O, 0.05 FeSO4.7H2O, 0.1 CaCl2.2H2O, 0.001 Na2MoO4.2H2O, 30 NaCl, and crude oil (1.0 percent, w/v) .Flasks containing sterilised mineral salt medium were inoculated with a loopful of bacterial culture cultured in crude oil containing nutrient agar plates and kept in a shaker at 200 rpm and 30°C for 7 days. After 7 days of incubation, each flask's culture broth was centrifuged for 15 minutes at 6000 rpm and 4°C, and the supernatant was filtered through 0.45m pore size filter paper (Millipore). The drop collapse assay, oil spreading assay, emulsification assay, BATH assay and surface tension assay were all performed using this cell-free culture broth9 .
D. Oil Spreading Test
The approach outlined by Morikawa et al, 2000 was used to conduct the oil spreading experiment7. In a nutshell, 20 ml of distilled water was placed to a plastic Petri dish, followed by 20 ml of crude oil added to the water's surface. The oil surface was then sprayed with 10 ml of cell-free culture broth. The negative control was distilled water (without surfactant), while the positive control was Triton X-100.
E. Microplate Assay
The microplate assay developed and patented by Vaux and Cottingham can be used to determine the surface activity of specific strains in a qualitative manner. A 100ml sample of each strain's supernatant is obtained and placed in a microwell of a 96-mircowell plate for this test. A grid-backed sheet of paper is used to see the plate. Only a modest amount of sample (100ml) is required. In addition, the approach can be used for automated high-throughput screening.16
F. Blood Haemolysis Test
In 5% Hen blood agar plates, a hemolytic assay was performed. Spot injected 50ml of bacterial culture cultured in a mineral salt medium onto blood agar plates and incubated for 48 hours at 37°C. A clear zone (hemolysis) around the colony was visually verified on the plates. A qualitative approach for determining biosurfactant production is the diameter of the clear zone. 6
G. CTAB Agar Plate
Extracellular glycolipids or other anionic surfactants can be detected using the CTAB agar plate method, which is a semi-quantitative assay. Siegmund and Wagner were the ones who came up with the idea14. On a light blue mineral salts agar plate containing the cationic surfactant cetyltrimethylammonium bromide and the basic dye methylene blue, the microorganisms of interest are grown. 16
H. Calculation of Emulsification Index (E24)
Several colonies of pure culture were suspended in test tubes containing 2 ml of mineral salt medium after 48 h of incubation; 2 ml cetaphil seed oil was added to each tube. Then, the mixture was vortexed at high speed for 1 min and allowed to stand for 24 hours. Calculation of emulsification index
Emulsification index=(Height of the emulsion layer/Total height) *10
I. Biochemical Test
Chemical Analysis of Biosurfactants was done by referring to the procedure explained by N.Mahesh8. (Table.no.2)
J. Sequencing
Both the water and soil sample were sent for 16srRNA sequencing. The bacteria in the soil sample was identified as Pseudomonas citronellolis and the bacteria in the water sample was identified as Pseudomonas aeruginosa .
K. Assay of Biosurfactant on Plant Analysis
Four samples were prepared using half cut water bottles with holes on the bottle for aeration. Control sample consisted of Soil with green gram, Sample A consisted of soil with crude oil and green gram, Sample B consisted of soil with crude oil and green gram along with microbes isolated from water sample introduced into the soil, Sample C consisted of soil with crude oil and green gram along with microbes isolated from soil sample introduced into the soil. Keeping all the conditions( sunlight and water ) same, all the samples were allowed to grow and the data was recorded . This was done to observe the growth of plant in every sample and how the biosurfactant microbes enhanced the growth of plants with respect to stem length, leaf length , leaf width .
L. Antimicrobial Activity of Isolated bacterium
The antibacterial activity of crude biosurfactant was tested using the agar well diffusion method. The crude biosurfactant was tested on Salmonella typhi, Aspergillus niger, Staphylococcus aureus, and Escherichia coli along with control was used as 5% ciprafloxaccin The plates were incubated for 24 hours at 37°C. The presence of a clear zone suggests that the crude biosurfactant has antibacterial action. 13
Data Description: Nucleotide sequence
Repository: GenBank
Accession no. OP120781
OP120782
III. RESULTS
- Oil spread test shows a positive results for Pseudomonas aeruginosa and Pseudomonas citronellolis. When the sample is added to the petri plate containing the oil and the water solution clear zone is formed. This shows that the sample as the biosurfactant property and hence it degrades the oil.
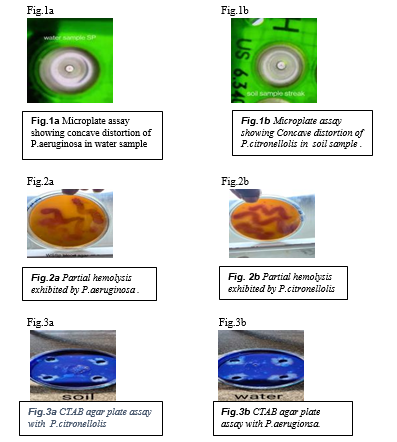
- In 96 well microplate assay when the sample was added to the wells containing oil, a concave surface distortion is formed, this can be viewed using a backing sheet of paper with a grid. This shows that Pseudomonas aeruginosa and Pseudomonas citronellolis have surfactant nature. Fig.1(a) Fig.1 (b)
- Blood haemolysis assay When the samples containing Pseudomonas aeruginosa and Pseudomonas citronellolis were inoculated on the blood agar and incubated at 32C for 48 hours, a clear zone was formed on the blood agar, which indicates the partial hemolysis of the blood agar. The partial hemolysis indicates the presence of biosurfactants producing organsims.
- CTAB Agar plate assay When the cell free culture is poured into the well on CTAB agar plate and incubated for 48 hours at 37 °C, dark blue colour was formed around the wells. This indicates the presence of biosurfactants producing organsims, Pseudomonas aeruginosa and Pseudomonas citronellolis.
- Emulsification index:
soil sample, Emulsification index=62.5%
water sample, Emulsification index=57.1%
6. Antimicrobial
The antimicrobial activity of the extracts containing surfactin produced by P.aeruginosa and P.citronellolis against various reference, environmental and clinical bacterial was then determined.
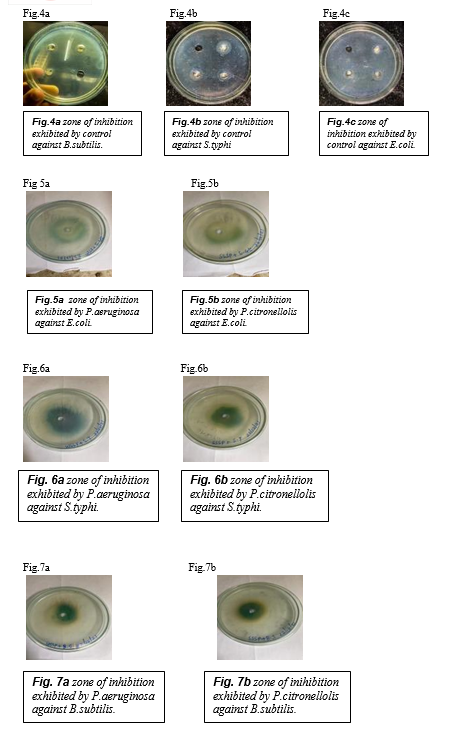
The average zone of inhibition exhibited by control against B.subtilis , S.typhi and E.coli was 0.5cm , 0.8cm and 0.3cm respectively. Fig.4(a) Fig.4(b) Fig.4(c)
The average zones of inhibition, the surfactin extract (WSSP) exhibited an increased antibacterial activity against E.coli strains ( average zone of inhibition of 4cm). Fig.5(a)
The average zones of inhibition, the surfactin extract (SSSP) exhibited an increased antibacterial activity against E.coli strains.( average zone of inhibition of 3cm). Fig.5(b)
The average zones of inhibition, the surfactin extract (WSSP) exhibited an increased antibacterial activity against S.typhi strains.( average zone of inhibition of 5cm).Fig.6(a)
The average zones of inhibition, the surfactin extract (SSSP) exhibited an increased antibacterial activity against S.typhi strains.( average zone of inhibition of 3.8cm).Fig.6(b)
The average zones of inhibition, the surfactin extract (WSSP) exhibited an increased antibacterial activity against B.subtilis strains.( average zone of inhibition of 3cm).Fig.7(a)
The average zones of inhibition, the surfactin extract (SSSP) exhibited an increased antibacterial activity against B.subtilis strains.( average zone of inhibition of 4cm).Fig.7(b)
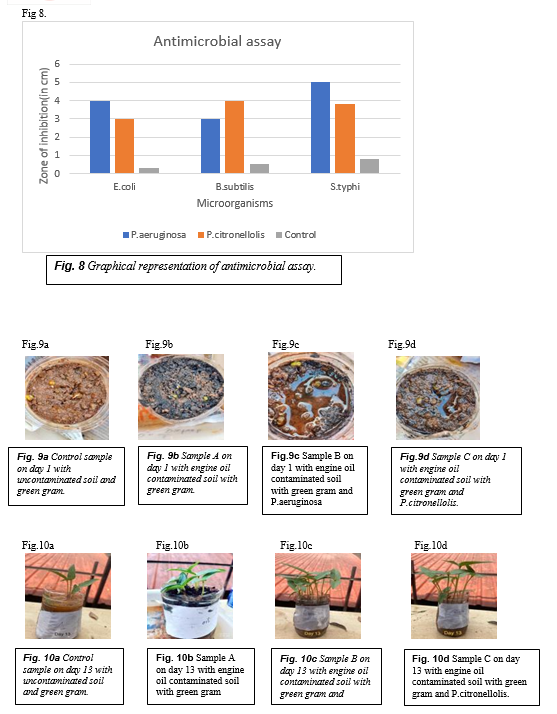
Graphical Respresentation of antimicrobial assay . Fig.8
7. Plant Growth Assay
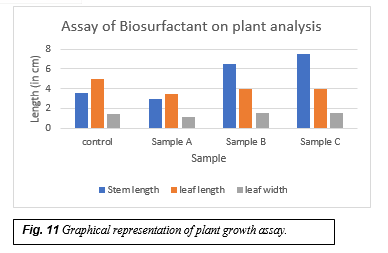
When the cell free cultures of Pseudomonas aeruginnosa and Pseudomonas citronellolis were added to different samples, there was various changes seen in the plant growth. Control sample had a contamination free soil to which the green gram seeds were added( Fig.9 a) .Sample A had the soil contaminated by the engine oil to which the green gram seeds were sowed(Fig.9 b). Sample B had the contaminated soil and the cell free culture of Pseudomonas aeruginosa (Fig.9 c) and the sample C contains the contaminated soil and the cell free culture of Pseudomonas citronellolis (Fig.9d) . The growth of the seeds were observed in these samples regularly. The seeds sprouted in all the samples around 3rd day. The growth of the plant in control sample was normal whereas in the sample A the growth of the plant was very slow and in the sample B and C the growth of the plant was fast and they seemed very healthy. After 13 days the stem length, leaf length and the width was measured, the plant in control sample had a stem length of 5cm, leaf length of 3.6cm and leaf width of 1.4cm (Fig.10a), in sample A the plant was with 3cm stem length, 3.5cm leaf length and 1.1cm leaf width(Fig.10b), in the sample B the plant was with 7.5cm stem length, 4cm leaf length and 1.5cm leaf width(Fig.10c) and the sample C plant was with 6.5cm stem length, 4cm leaf length and 1.5cm leaf width(Fig.10d). This clearly shows that the sample with the biosurfactants have the plants with better growth, the stem length and the leaf length is twice the plants grown in normal control sample. So the biosurfactants acts as plant growth promotor in the contaminated soil and help in the growth of plants and also due to their antimicrobial property they help in boosting the plant immune system.
Graphical representation of plant growth assay (Fig.11)
IV. DISCUSSION
Biosurfactants are the amphiphilic compounds which can amass between phases of liquids. They are usually produced on surface of bacterial cell4 .A wide range of microorganisms have been brought up which exhibit different class of biosurfactant property such as glycolipids, phospholipids, neutral lipids and lipopeptides12. Biosurfactant forms micelles complex with crude oil which leads to emulsion and thus the crude oil can be recovered by breaking the emulsion. In the similar way the biosurfactant binds with the minerals example phosphorous and forms micelles complex and helps in the uptake of minerals in plants. Psuedomonas produce glycolipid group called rhamnolipids. Rhamnolipid is primarily a crystalline acid which is composed of beta-hydroxy fatty acid connected by carboxyl end to rhamnose sugar molecule. 5 Rhamnolipid insertion inside the lipid bilayer does not strongly affect the lipid dynamics but the nature of the phytosterols could influence the effect of glycolipids on plant membrane destabilization. These subtle changes in lipid dynamics could be linked with plant defense. The rhamnolipids trigger the plant physical protective mechanism like callose deposition and stomatal closure and thus helps in plant protection.10
V. ACKNOWLEDGMENT
The authors are thankful to K.L.E. Society’s P.C. Jabin Science College , Hubballi, Karnataka India for providing necessary facilities to carry out the present research. The authors are also thankful to Bio edge Solutions Pvt. Ltd. For supporting in sequencing the microbial isolates.
- Conflict of Interest: The authors declare that they have no conflict of interest in the publication.
- Compliance of Ethical Standard: This article does not contain any studies involving human or animal participants performed by any of the authors.
VI. FIGURES
Conclusion
Biosurfactants have gained a greater interest in past years. With respect to oil and gas sector, the application of biosurfactants in degradation and in dispersant form to treat oil spills are of greater interest. Along with these application, testing on plant for comparative study of the growth gave a different angle to thought process. The antimicrobial activity and the enhancement in the plant growth can lead to a revolution in the field of agriculture. It can also thereby protect environment. In future studies , rhamnolipids , which are the key component of biosurfactant responsible for the forementioned application can be extracted and can be formulated into oil cleanser, plant growth promoter and soil quality improver products.
References
[1] Banat, I.M., Franzetti, A., Gandolfi, I., Bestetti, G., Martinotti, M.G., Fracchia, L., Smyth, T.J. and Marchant, R. (2010) Microbial biosurfactants production, applications and future potential. Applied Microbial Biotechnology 87, 427–444. [2] Burger M. M., Glaser L. and Burton R. M. (1963), The synthesis of rhamnose-containing glycolipid by extracts of Pseudomonas aeruginosa.J. Biol. Chem.238, 2595-2602. [3] Cameotra, S.S., Makkar, R.S., Kaur, J. and Mehta, S.K. (2010) Synthesis of biosurfactants and their advantages to microorganisms and mankind. In Biosurfactants ed. Sen, R. pp. 261–280. [4] Jerome Crouzet,Anthony Arguelles Biosurfactants in Plant Protection Against Diseases: Rhamnolipids and Lipopeptides Case Study 2020 https://doi.org/10.3389/fbioe.2020.01014 [5] Kamaljeet K. Sekhon Randhawa Rhamnolipid biosurfactants—past, present, and future scenario of global market Front. Microbiol., 02 September 2014 https://doi.org/10.3389/fmicb.2014.00454 [6] M.Rajesh et al., Journal of Applied & Environmental Microbiology. 2017, 5(2), 74-78 . [7] Morikawa M, et al. BBA - Molecular and Cell Biology of Lipids. 2000;1488:211. [8] N. Mahesh , S. Murugesh and V. Mohana Srinivasan , 2006. Determination of the Presence of Biosurfactant Produced by the Bacteria Present in the Soil Samples. Research Journal of Microbiology, 1: 339-345. [9] Nayarisseri A, Singh P, Singh SK. Screening, isolation and characterization of biosurfactant producing Bacillus subtilis strain ANSKLAB03. Bioinformation. 2018 Jun 30;14(6):304-314. doi: 10.6026/97320630014304. PMID: 30237676; PMCID: PMC6137570. [10] Noadya Monnier et el., Front. Plant Sci., 08 August 2018 | https://doi.org/10.3389/fpls.2018.01170 [11] Nikolova C and Gutierrez T (2021) Biosurfactants and Their Applications in the Oil and Gas Industry: Current State of Knowledge and Future Perspectives. Front. Bioeng. Biotechnol. 9:626639. doi: 10.3389/fbioe.2021.626639 [12] R.Thasavi et al., Indian J Microbiol. 2011 Jan; 51(1): 30–36. Published online 2011 Jan 25. doi: 10.1007/s12088-011-0076- PMID: 22282625 [13] S. O. Adebajo, P. O. Akintokun, A. E. Ojo, A.K. Akintokun & O.A. Badmos (2020) Recovery of Biosurfactant Using Different Extraction Solvent by Rhizospheric Bacteria Isolated from Rice-husk and Poultry Waste Biochar Amended Soil, Egyptian Journal of Basic and Applied Sciences, 7:1, 252-266, DOI: 10.1080/2314808X.2020.1797377 [14] Siegmund, I., Wagner, F. New method for detecting rhamnolipids excreted byPseudomonas species during growth on mineral agar. Biotechnol Tech 5, 265–268 (1991). [15] Shekhar, S., Sundaramanickam, A. and Balasubramanian, T. (2015) Biosurfactant producing microbes and their potential applications: a review. Crit Rev Environ Sci Technol 45, 1522–1554. [16] Walter V, Syldatk C, Hausmann R. Screening Concepts for the Isolation of Biosurfactant Producing Microorganisms. In: Madame Curie Bioscience Database [Internet]. Austin (TX): Landes Bioscience; 2000-2013.
Copyright
Copyright © 2022 Deepak Mundrikeri, G Sanjana, Abhishek Kannur, Suman Bishnoi, Dr. Sneha Chinta, Dr. Badarinath Kulkarni, Dr. Geetanjali Kamble. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET48314
Publish Date : 2022-12-22
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

