Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Mechanism of Formation of Smog and some Adverse Effect of Smog on Environment and Human Being
Authors: Dhanraj ., Ram Bilas Meena
DOI Link: https://doi.org/10.22214/ijraset.2023.56235
Certificate: View Certificate
Abstract
The formation of smog is a complex chemical process with significant implications for the environment and human health. This phenomenon primarily results from the interplay of various reactants and products, with its origins tracing back to specific sources. The key contributors to photochemical smog include nitrogen oxides, hydrocarbons, and the energy source that drives the reaction—sunlight. Notably, pollutants like nitrogen oxides and hydrocarbons are byproducts of fossil fuel combustion, primarily emitted from coal-fired power plants and various energy production facilities. Additionally, volatile organic compounds (VOCs) are generated when fossil fuels undergo incomplete combustion, as observed in activities like backyard burning and wood-burning stoves. Smog, characterized by a blend of airborne pollutants, poses significant threats to both human well-being and the environment. It can lead to a range of health issues, including respiratory difficulties, asthma exacerbation, heightened susceptibility to lung infections and colds, and eye irritation.
Introduction
I. INTRODUCTION
The word smog is derived from an elision of smoke and fog, first coined by H.A. Voeux. When atmospheric visibility is partially obstructed by a haze made up of solid particles and liquid aerosols, it is referred to as smog. It is actually fog that is polluted with smoke and was accordingly named by combining the words smoke and fog. In the majority of large urban regions, smog is a major issue. The main reasons behind the occurrence of smog are emissions from industries and automobiles, the burning of wood and coal, and certain weather conditions that lead to the accumulation of pollutants in the air. Pollutants that cause smog can come from a variety of places, including industry, consumer goods, and cars. In urban regions, vehicle emissions contribute to more than half of the smog formation. The occurrence of smog is generally linked to the interplay between weather conditions and high traffic of motor vehicles, as well as emissions from industries and other consumer products. Mostly, the occurrences of smog are associated with the relationship between weather patterns and heavy motor vehicle traffic and industrial and other consumer product emissions. Solvents, paints, plastic containers, and sprays are examples of consumer products.
II. SOURCES OF SMOG
For cities located in river basins and surrounded by mountains, smog is a persistent problem because trapped smog in the valley cannot be carried by the wind. Primary pollutants are the ones that contribute to smog formation and are emitted directly from the source. By chemically interacting with primary pollutants and typical environmental factors, secondary pollutants form in the atmosphere. Oxides of nitrogen, particularly nitrogen oxides, and nitric oxides, which are released into the atmosphere by the burning of fossil fuel from coal power plants, industrial emissions, and vehicle exhausts, are the main pollutants that cause air pollution. When inhaled, nitrogen dioxide, a dangerous air contaminant, produces pulmonary edema (an accumulation of excessive fluid in the lungs). Moreover, it contributes to photochemical smog, seriously harming the ecosystem.
III. TYPES OF SMOG
There are two types of smog:
A. Sulphureous or London Smog
It is a mixture of smoke, fog and sulphur dioxide that affected London so badly after the introduction of coal as a fuel, is chemically reducing mixture, so it is called reducing smog.
Components of Reducing Smog
These are:
- SOx
- Particulates such as soot, ammonium sulphate etc.
- Humidity from fog and aerosol
B. Photochemical Smog
During the 1940s, a novel form of air pollution known as photochemical smog was initially identified in Los Angeles. This unique type of smog is caused by a combination of several major air pollutants, including carbon oxides (CO, CO2), nitrogen oxides and nitric acid (NO, NO2, HNO3), sulphur dioxide and sulphuric acid (SO2, H2SO4), suspended particulate matter (SPM), ozone (O3), and volatile organic compounds (VOCs).
- Components of Photochemical Smog
Vehicular exhaust, unsaturated hydrocarbons, oxides of nitrogen, carbon monoxide, hydrogen peroxide, ozone, some compounds containing sulphur, organic peroxides, hydroperoxides, peroxy acetyl nitrate, peroxybenzyl nitrate, peroxy propionyl nitrate, aldehydes, tertiary butyl hydroperoxide, and particulates constitute components of smog.
2. Mechanism of Smog Formation
Smog is typically caused by a variety of pollutants that are emitted from various sources such as factories, vehicles, and consumer products. In many urban areas, more than half of the smog is generated due to vehicular emissions. The occurrence of smog is often linked to the interplay between weather patterns and the heavy traffic of motor vehicles, as well as industrial and consumer product emissions. The latter can include items such as solvents, paints, plastic packaging, and sprays. In the lower region of the atmosphere, oxides of nitrogen play a major role in the formation of smog through a complex set of reactions. It is produced by the reaction of sunlight with hydrocarbons, nitrogen compounds and other gases released in automobile exhaust. When oxides of nitrogen and at least one volatile organic compound (VOC) present in the atmosphere are exposed to sunlight, they react to produce photochemical smog. These photochemical reactions usually occur in summer and lead to the development of ground-level ozone or bad ozone (O3) which is an important smog-causing pollutant. The ozone resulting from photochemical smog can have a harmful impact on human health, particularly on the respiratory and cardiovascular systems, and is known to be a contributing factor to premature deaths.

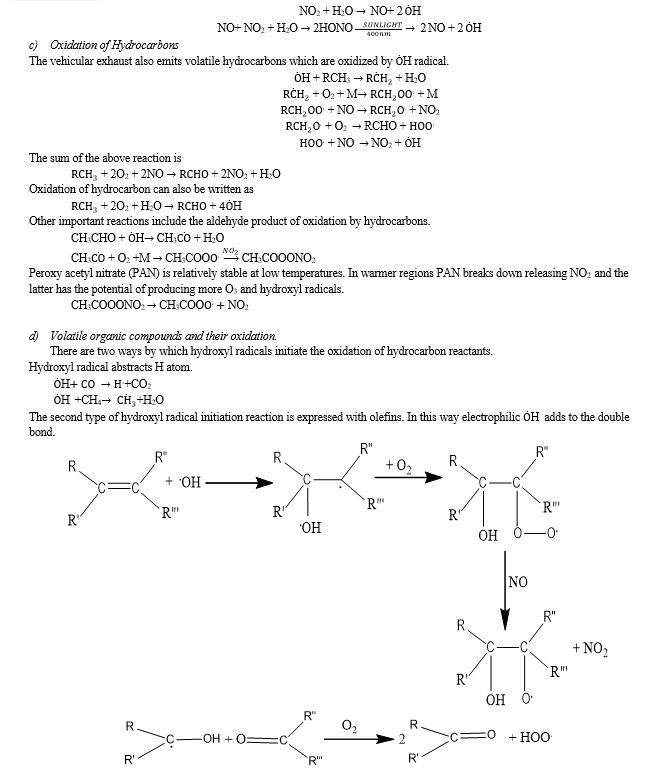
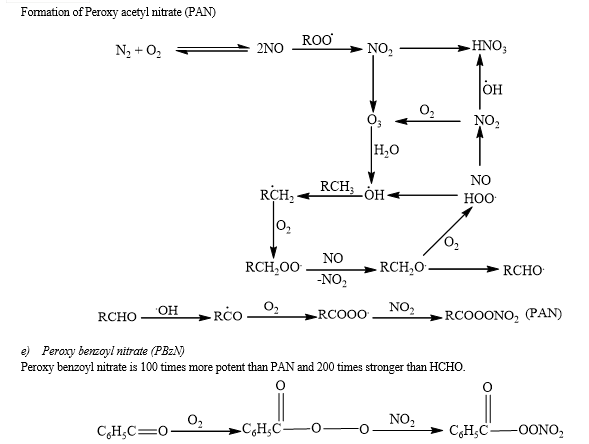
IV. ADVERSE EFFECTS OF PHOTOCHEMICAL SMOG
Smog is primarily composed of ground-level ozone (O3), but it also comprises several other chemicals, such as carbon monoxide (CO), particulate matter like soot and dust, as well as volatile organic compounds (VOCs) such as benzene, butane, and other hydrocarbons. It is important to note that ground-level ozone, which is a harmful pollutant with several adverse health effects, should not be mistaken for the useful ozone layer high in the stratosphere that safeguards the earth from the sun’s harmful ultraviolet rays.
A. Effect On Human Beings
Health effect
Air pollution has been associated with adverse effects. SO2 and coal ducts have been identified as key agents in the past London “killer smog” events. Additives in leaded gasoline were found to cause elevated blood lead levels and related health issues in both children and adults. The World Health Organization has identified particle pollution as a significant contributor to the development of lung cancer. Smog is also known to increase the risk of heart attacks and strokes by introducing harmful pollutants into the bloodstream through inhalation. Additionally, exposure to smog can have a negative impact on fetal development. It can even result in miscarriages. And sometimes, miscarriages do not occur, but it causes the baby to be underweight. Burning of the eyes is another common symptom of Smog. In addition, you may experience swelling, redness, irritation, and itching. Respiratory problems are the most occurring diseases due to Smog. Lung malfunctioning, shortness of breath, bronchitis, asthma, COPD, and breathing problems can all occur due to poor air quality. Photochemical smog cause irritates the eyes, causes obstructive pulmonary disease, cardiovascular disease, and asthma. Exposure to London smog can cause eye irritation, bronchitis, and respiratory problems such as lung issues.
Table 1: Health effects of pollutants involved in photochemical smog
|
Pollutants |
Effects |
|
Nitrogen oxides |
• can contribute to problems with the heart and lungs • links to decreased resistance to infection |
|
Volatile organic compounds (VOCs) |
• eye irritation • respiratory problems • some compounds are carcinogens |
|
Ozone |
• coughing and wheezing • eye irritation • respiratory problems (particularly for conditions such as asthma) |
|
Peroxyacetyl nitrate (PAN) |
• eye irritation • respiratory problems |
B. Effects On Plants
The impact of air pollutants on plants was one of the initially observed outcomes of photochemical smog. Various pollutants enter the plant system via the stomata through which the plant takes carbon dioxide for photosynthesis. When air pollutants are taken up by plants during the process of normal gas exchange, they can trigger harmful chemical reactions within the structure of the leaves. PAN, PBN, etc. are produced photochemically in the air having hydrocarbons, oxide of nitrogen, ozone, etc. These components are phyto-toxicant causing bronzing and gazing on leaf surfaces. The initial collapse in the spongy parenchyma cells surrounding the air space into which the stomata open. Some plants, including tobacco and petunias, can have collapses that go all the way through the leaf thickness. Thus, a cross-leaf banding associated with sequential cell maturation along the leaf blade develops.
C. Effects On Material
Smog, which includes fog, dust, mist, and soot, among other things, impairs visibility and corrodes metals, stones, construction materials, rubber, leather, painted surfaces, and paper. The susceptibility to attack enhances with an increasing number of double bonds in olefins. These reactions create carbon chain breaking and cross-linking. Actually, the long chains of carbon atoms that make up the polymer are broken and the material becomes fluid like losing its tensile strength. Even a trace of ozone results in the formation of new links between parallel carbon chains making the material less elastic and more brittle. The relationship between dye structure and ozone activity has been explored by many researchers. Ozone is an electrophilic agent that can attack the amino group. The nucleophilic properties of the nitrogen in the aryl amino dyes determine the extent of oxidative fading. A structural modification reduces the electron density of the nitrogen and renders it less susceptible to attack by electrophilic agents, whether ozone or oxides of nitrogen. Ozone causes the fading of certain dyed fabrics. Such materials dyed with ozone-sensitive dyes and exposed outdoors in several areas of the U. S. were most impacted in Los Angeles, where there is a substantial amount of ozone present in the ambient "total oxidant" measurement. Compared to comparable materials exposed in a dry area with abundant sunlight, dyed fabrics exposed in humid areas with abundant sunlight exhibited higher fading. Due to the fact that rubber is an organic component, it is delicate to various chemical influences, such as oxidation, reduction, and substitution, which easily break apart the rubber molecule or alter the beneficial qualities of rubber. Before the recognition of ozone as a specific chemical component found in atmospheric photochemical smog, it was already widely known that rubber could crack, lose elasticity, and deteriorate when exposed to ozone.
There have been reports of the effects of ozone on asphaltic materials, indicating that photochemical oxidants can have an impact on materials other than just rubber. Recent studies on the photo-oxidation of asphaltic films in the presence of very high concentrations of ozone (much in excess of any atmospheric photochemical smog concentrations) showed degradation of these films. The oxidative changes or time-to-failure of the films (cracking) were measured by means of an infrared spectroscope. There is limited precise knowledge recorded in the literature about the effects of ozone on materials other than rubber. Researchers indicated that ozone reacts with damp or wet aluminum but has practically no effect on dry aluminum. Platinum, when at high temperatures, was attacked by ozone.
Neither the ozone concentrations nor the exposure periods are provided. Researcher also cited that ozone was not compatible with phenol formaldehyde resins, but provided no further clarification.
V. INHIBITION OF PHOTOCHEMICAL SMOG
To inhibit the formation of photochemical smog, it may be possible to add free radical scavengers to the atmosphere. The major components of photochemical smog, such as ozone, nitrogen dioxide, and peroxyacetyl nitrate, involve free radicals, and by adding scavengers, the chain processes that form photochemical oxidants can be terminated. One such promising scavenger is diethyl hydroxyl amine (DEHA), which is a volatile liquid with a mild odor and has been shown to be non-toxic and non-mutagenic. When only 3 m torr of DEHA is added to a propylene-NO2-O2 mixture with a total pressure of 100 torrs, the oxidation of NO to NO2 is completely inhibited, while the addition of 2.5 torr results in 50% inhibition. The inhibition seems to result from the radical abstraction of hydrogen.
Conclusion
Smog is a global issue that adversely affects many major cities. The reliance on fossil fuels to support rapid urbanization and industrialization is a significant contributor to smog occurrences in India. The primary sources of smog in India include vehicular emissions, as well as the burning of crops and solid waste. Smog not only poses health risks to humans but also has negative impacts on animals, tourism, and the economy as a whole. This study has outlined the sources of smog, its associated hazards, and potential preventive measures to combat smog in India. It\'s important to note that there is a lack of specific studies quantifying the economic impact of smog in India. Addressing smog requires the implementation of strict policies and emissions reduction strategies to decrease private car usage, although these efforts are currently insufficient on a national scale. Encouraging the use of public transportation with improved facilities is essential. Legislation pertaining to vehicle maintenance should be enhanced and enforced, while the advantages of hybrid vehicles need to be widely promoted. Industrial emissions from various sources must be closely monitored, treated, and reduced through the adoption of green industrial policies. India is also employing a successful green plantation strategy to mitigate smog severity in its major cities. In addition to government policies, the reduction of smog events can be achieved through community-driven initiatives such as social seminars and public education campaigns that promote behavioral changes. Therefore, it is imperative for both government institutions and individual citizens in India to work together in a coordinated effort to combat and reduce the occurrence of smog.
References
[1] (World Health Organization. Ambient (outdoor) air qua-lity and health. Fact sheet no. 313. (Online) 2016 September (Cited 2016 November 10). Available from URL: http://www.who.int/mediacentre/factsheets/fs313/en/) Fine particulates of 2.5 micrometer or less are also among the pollutants found in smog and haze. [2] (Uzoigwe JC, Prum T, Bresnahan E, Garelnabi M. The Emerging Role of Outdoor and Indoor Air Pollution in Cardiovascular Disease. North American Journal of Medical Sciences, 2013; 5 (8): 445-453. Doi:10.4103/1947-2714.117290.) [3] Y. A. Cengel and M. A. Boles, Thermodynamics: an Engineering Approach, McGraw-Hill, Boston, 6th ed., 2007. [4] J. Jackson, S. Choudrie, D. G. Thistlethwaite, N. Passant, T. Murells, J. Watterson, et al., UK greenhouse gas inventory, 1990 to 2007: annual report for submission under the framework convention on climate change, AEA Technology plc., 2009, http://uk-air.defra.gov.uk/reports/cat07/0905131425 ukghgi-90- 07 main chapters Issue2 UNFCCC CA v5 Final.pdf [5] A. J. Manning and R. G. Derwent, Interpretation of longterm measurements of radiatively active trace gases and ozone depleting substances, Defra Contract: CPEG1 Quarterly Report, The Met Office, Devon, United Kingdom, 2006. [6] A. J. Manning, D. B. Ryall, R. G. Derwent, P. G. Simmonds, and S. O’Doherty, Estimating European emissions of ozone-depleting and greenhouse gases using observations and a modeling backattribution technique, Journal of Geophysical Research, 108 (2003), 4405. [7] R. Milne and D. C. Mobbs, UK emissions by sources and removals by sinks due to land use, land use change and forestry activities report, April 2006. DEFRA contract EPG 1/1/160 CEH No. C02275 [8] Colbeck, I & Mackenzie, AR 1994, Air pollution by photochemical oxidants, Air Quality Monographs Vol 1, Elsevier, Netherlands. White, V 1998, Air emissions inventory for the Adelaide airshed 1995, Environment Protection Agency, South Australia. [9] J. R. Wright and P. G. Campbell, \"Photooxidation of Asphalts in the Presence of Ozone,\" J. Res. National Bur. Standards, 68C (4): 297 (Oct.- Dec. 1964). [10] E. Rabald, Corrosion Guide, p. 588, etc. Elsevier Publishing Co. New York, N.Y. (1951). [11] J. Crabtree and B. S. Biggs, \"Cracking of Stressed Rubber by Free Radicals,\" J. Polymer Science 11: 280 (1953). [12] E. P. W. Kearsley, \"Standardisierung der Kautschukprufungmittels Ozon.\" Kautschuk 6: 244 (1930).
Copyright
Copyright © 2023 Dhanraj ., Ram Bilas Meena. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET56235
Publish Date : 2023-10-20
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

