Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Microbial Enhanced Oil Recovery (MEOR) Surfactant from Pseudomonas aeruginosa isolated from Automobile Garage soil.
Authors: Niraj Prasad, Sumita Dasgupta, Mousumi Chakraborty, Smita Gupta
DOI Link: https://doi.org/10.22214/ijraset.2022.45784
Certificate: View Certificate
Abstract
Microbial enhanced oil recovery (MEOR), is gaining attention today for being environmentally friendly, economically attractive, demonstrating improvement in recovery of oil entrapped in porous media. It is considered to be more efficient than other EOR methods when applied to carbonate oil reservoirs. In the current study oil degrading bacteria was isolated from soil of automobile garage. The biosurfactant producing organism was screened and characterized. 16SrRNA sequence of the most potent bacterial strain suggests it to be belonging to the genus Pseudomonas and species aeruginosa. The biosurfactant produced by the bacteria was detected as rhamnolipid with emulsification index of 69%, foaming of 57.69% and interfacial surface tension of 0.6 mN/m. The MEOR column assay revealed that the additional oil recovery for sand1 and sand2 was 62.142% and 52.173 respectively.
Introduction
I. INTRODUCTION
The current global energy production and consumption from fossil fuels is about 80-90% that contribute 60% of total oil and gas production [1]. Though primary oil recovery during the oil production recovers 30-40% oil and additional 15-25% can be recovered by secondary oil recovery methods like water injection, steam injection etc., 35-55% of oil is left as residual oil in the reservoirs [2].Thus this residual oil becomes the target of enhanced oil recovery technologies and it amounts about 2-4 trillion barrels that is about 67% of the total oil reserves [3,4]. The techniques used for recovery of residual oil is commonly termed as Enhanced Oil Recovery (EOR). Enhanced oil recovery (EOR) is a tertiary recovery process that involves application of different thermal, chemical, and microbial processes to recover additional 7–15% of Original oil in place (OOIP) at an economically feasible production rate from poor performing and depleted oil wells [5]. EOR can significantly impact oil production, as increase in the recovery rate of oil by even a small margin could bring significant revenues without developing unconventional resources [6]. Microbial enhanced oil recovery (MEOR), the use of microorganism and their surface active metabolites in recovery of residual oil after secondary recovery is an alternative oil recovery approach which is claimed in the literature to be very promising in recovering up to 50% of residual oil and also reported to improve petroleum extraction efficiency [7-10]. In 1926, it was suggested for the first time that microorganisms could be used to release oil from porous media [11]. This technology has some advantages. Economically attractive for marginally producing oil fields; a suitable alternative before the abandonment of marginal Cells. According to a statistical evaluation (1995 in U.S.), 81% of all MEOR projects demonstrated a positive incremental increase in oil production and no decrease in oil production. The implementation of the process needs only minor modifications of the existing field facilities. It is less expensive to install and more easily applicable than another EOR method. MEOR products are all biodegradable and will not be accumulated in the environment, so environmentally friendly [12].
Biosurfactants produced by microorganisms from various taxonomic groups and diverse habitats, have attained the special status of a chemical in demand in recent years, have many potential applications including enhanced oil recovery [13,14]. Soil of the automobile garages are contaminated with oil. Contaminants usually found at the garages include brake fluid, engine/dirty oil, petrol, kerosene and bitumen [15]. In the current study the microorganisms were isolated from soil of automobile garage in Udhana area of Surat, Gujarat. The isolates were screened for their ability to produce biosurfactant. The most potent microorganism and the chemical nature of the biosurfactant produced by it was identified. The applicability of the surfactant in enhanced oil recovery was evaluated by analysing its emulsification capacity, foaming characteristics and its ability to reduce the water surface tension. Finally, the oil recovery ability of the biosurfactant was assessed.



Emulsification index E24 gives the measure of the emulsification capacity of any surface active molecule with different hydrocarbons [35]. Stabilizing the emulsion is a function of the biosurfactant concentration [36,37] The emulsification index was found to be 69% that represent a great ability of biosurfactant for formation of emulsion. Total height was 42mm and emulsion height was found to be at 29mm. This method provides better option for screening microorganisms that produce biosurfactant. This result was in accordance with other publications [34,38]. In the present study, foaming was observed after shaking that shows presence of surface active compound. Foaming was found to be 57.69%.The foaming characteristic of surfactant solutions is one of the most important property. Occurrence of foaming, indicates the presence biosurfactant [39,25].
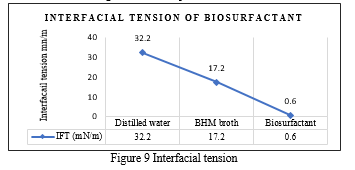
The interfacial tension was measured against paraffin oil. The interfacial tension for distilled water against water was found to be 32.2 mN/m and of BHM broth was found to be 17.2mN/m and this served as a blank for measuring interfacial tension for supernatant containing biosurfactant. The interfacial tension was found to be 0.6mN/m ( Figure 9). This showed that the biosurfactant produced by isolated strain identified as P. aeruginosa is capable of reducing interfacial surface tension. A higher biosurfactant concentration in the test sample provides a lower surface tension until the critical micelle concentration (CMC) is reached [40]. The ability of the biosurfactant to reduce the surface tension of the water and a good emulsifying capacity indicates a promising application in microbial enhanced oil recovery (MEOR) [34].
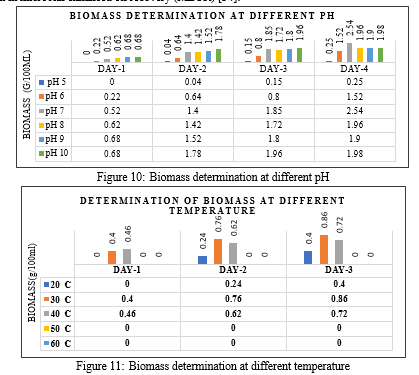
In the current study maximum biomass production was observed at pH 7.0 ( Figure10) and temperature 300C ( Figure 11). Temperature is a critical environmental factor affecting microbial growth and reproduction [41]. Similar result for optimal growth conditions of temperature and pH for strain belonging to the genus Achromobacter was reported [32]. In our study slow growth was observed at 20 ? C which gradually increases and maximum biomass production (0.86/100ml) was observed at 30oC ( Figure 11). Production was found to decline at temperature beyond 30oC.
The characteristic of the biosurfactant observed in this study with respect to Emulsification index (E24),Foaming,Interfacial tension was found to be in co-relation with previous study by [42] where biosurfactant rhamnolipid was isolated from Pseudomonas aeruginosa. In the present study also the biosurfactant was detected to be rhamnolipid.
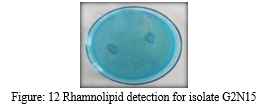
After incubation of 48 hours, blue coloured ring surrounding the colony was observed, indicating biosurfactants have the ability to form a coloured complex with the cationic indicator methylene. If anionic surfactants are secreted by the microbes growing on the plate, they form a dark blue- purple, insoluble ion pair with cetyltrimethylammonium bromide and methylene blue. Accordingly, rhamnolipid-producing strains can be recognized by the dark blue halos formed around the colonies and, ideally, the amounts of rhamnolipids produced could be correlated with the areas of the halos [29]. The dark blue halo around the colonies in the present study (Figure 12) confirmed the presence of rhamnolipid compound produced by the isolate identified as Pseudomonas aeruginosa.
It was reported that the biosurfactants produced by Pseudomonas aeruginosa are mainly rhamnolipids[43].]
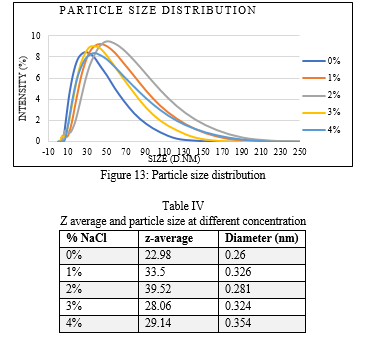
Particle size distributions in microemulsions at 0-4 wt% NaCl solutions are shown in Figure 13. The mean particle diameter and poly dispersity have been calculated from intensity, mass and number bimodal distribution. Size of the dispersed particles in microemulsions plays an important role in EOR because as the size decreases, the colloidal interaction between the dispersed particles increases.
The Z average diameter of dispersed droplet is the mean hydrodynamic diameter and is calculated according to the International Standard on dynamic light scattering ISO13321. The Z-average diameter is intensity weighted and is therefore sensitive to the presence of large particles. Z-average diameter is found to be strong function of the salinity of the microemulsion. Particle size and IFT of the microemulsion is dependent on the salinity and hence oil recovery by microemulsion flooding is also function of salinity of microemulsion [30]. In our study Z-average diameter of the microemulsion was detected at optimal salinity of 3 % NaCl with particle size about 0.324 nm diameter ( Table IV). Which is in accordance with previous report [30].
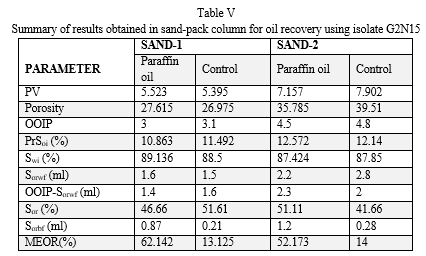
The biosurfactant produced from the isolated strain of Pseudomonas auregiona was subjected to MEOR Column assay. The additional oil recovery for sand1 and sand2 was measured after nullifying the control volume (Table V). The additional oil recovered for sand1 and sand2 is 49.0% and 38% respectively. This result is in agreement with that of previous work [16,44].
Previous investigations have reported that the biosurfactants rhamnolipid isolated from P aeruginosa can be efficiently used for MEOR applications, since it presented expressive results when compared with other biosurfactants including synthetic surfactants [34].
Conclusion
In the current study oil degrading bacterial strain designated as G2N15 was isolated from the contaminated soil of automobile garage located in Surat, Gujarat. Based on the results of Gram staining, biochemical and 16s rRNA sequence analysis the strain was suggested to be belonging to genus Pseudomonas and species aeruginosa. The biosurfactant produced by the strain was detected as rhamnolipid possessing excellent surface activities and emulsifying properties. The MEOR column assay predicted the efficiency of the biosurfactant for MEOR. Our current study demonstrated the possibility of developing an easy and inexpensive technique for MEOR using biosurfactant produced by Pseudomonas aeruginosa isolated from the contaminated soil of automobile garage.
References
[1] W. Graus, M. Roglieri, P. Jaworski, L. Alberio, and E. Worrell, “The promise of carbon capture and storage: evaluating the capture-readiness of new EU fossil fuel power plants” Climate Policy, vol. 11, pp.798-812,2011. [2] R.Cosse, “Basics of reservoir engineering, editions technip, Paris, 343pp.enhanced waterflooding field pilots.,” Dev. Pet. Sci., vol. 39, pp.289-306, 1993. [3] C.Hall, P.Tharakan, J.Hallock, C. Cleveland, and M. Jefferson, “Hydrocarbons and the Evolution of Human Culture,” Nature, vol. 426, pp.318-322, Nov.2003. [4] R.S. Bryant, A. K. Stepp, K. M. Bertus, T. E. Burchfield, and M. Dennis,“Microbial enhanced water flooding field pilots,” Dev. Petrol. Sci., vol. 39, pp. 289-306, 1993. [5] R. T.Bachmann, A. C.Johnson, and R. G. J. Edyvean, (2014). “Biotechnology in the petroleum industry: an overview,” Int. Biodeterior. Biodegradation, vol. 86, pp.225–237,2013. [6] S. Thomas, “Enhanced oil recovery-an overview,” Oil Gas Sci. Technol,. vol. 63, pp. 9–19,2008. [7] A.Rabiei , M. Sharifinik, A. Niazi, A. Hashemi, S. Ayatollahi, “Core flooding tests to investigate the effects of IFT reduction and wettability alteration on oil recovery during MEOR process in an Iranian oil reservoir.” Appl. Microbiol. Biotechnol, vol. 97, 5979-5991,2013. [8] B.Shibulal S.N. Al-Bahry, Y.M. Al-Wahaibi, A.E. Elshafie, A.S. Al-Bemani, and S.J..Joshi, “Microbial enhanced heavy oil recovery by the aid of inhabitant spore-forming bacteria: an insight review,” Sci. World J.,2014.pp.1-12, 2014. [9] S. J. Geetha, I. Banat, M., and S. J. Joshi, (2018). “Biosurfactants: production and potential applications in microbial enhanced oil recovery (MEOR),” Biocatal. Agric. Biotechnol. Vol. 14,pp. 23–32,2018. [10] C. Nikolova, and T. Gutierrez, “Use of Microorganisms in the Recovery of Oil From Recalcitrant Oil Reservoirs: Current State of Knowledge, Technological Advances and Future Perspectives,” Front Microbiol., Vol.10, Jan 2020. [11] J.W.Beckman, “The action of bacteria on mineral oil,” Industrial and Engineering Chemistry, News Edition. Vol.4, pp. 23–26, 1926. [12] I.Lazar, G. Petrisor, and T. F. Yen, “Microbial Enhanced Oil Recovery (MEOR),” Petroleum Science and Technology, vol.25,pp.1353-1366,2007. [13] D. Haferburg, R. Hommel, R. Claus, and H.P. Kleber, “Extracellular microbial lipids as biosurfactants,” Engineering Biotechnology,vol. 33, pp. 53-59, 1986. [14] I.M.Banat, R.S. Makkar, and S.S. Cameotra,”Potential applications of microbial surfactants,” Appl Microbiol Biotechnol., vol. 53, pp. 495– 508,2000. [15] X. Lang, A. Dalai, N. Bakhshi, M. Reaney, and P. Hertz, 2001. “Preparation and characterization of bio-diesels from various bio-oils,” “Bioresource Technology” vol. 80,pp. 53-62, 2001. [16] E.J. Gudin˜a, L.R. Rodrigues, J.A. Teixeira, J.F. Pereira, J.A. Coutinho, A.L. Soares, M.T. Ribeiro, in: Abu Dhabi International Petroleum Exhibition & Conference Held in Abu Dhabi, UAE, 11–14 Nov, 2012. [17] F. Chaillan, F.A. Le, E. Bury, Y.H. Phantavong, P. Grimont, A. Saliot, J. Oudot,“Identification and biodegradation potential of tropical aerobic hydrocarbon-degrading microorganisms,” Res. Microbiol., vol. 155, pp. 587-595,2004. [18] M. Hassanshahian, and G. Emtiazi,“Investigation of alkane biodegradation using the microtiter plate method and correlation between biofilm formation, biosurfactant production and crude oil biodegradation,” International Biodeterioration & Biodegradation, Vol. 62, pp. 170-178, 2008. [19] M. Morikawa, Y. Hirata, T. Imanaka, “A study on the structure function relationship of lipopeptide biosurfactants,” Biochim Biophys Acta, vol. 1488 pp. 211–218,2000. [20] N. H.Youssef, K. E.Duncana, D.P.Naglea, K. N.Savage, R. M. Knapp, M. J.McInerney, “Comparison of methods to detect biosurfactant production by diverse microorganisms,” J Microbiol Methods., 2004 Mar Vol.56, pp. 339-347, March 2004. [21] C.N. Mulligan, D.G. Cooper, R.J. Neufeld, “Selection of microbes producing biosurfactants in media without hydrocarbons,” J. Ferment. Technol., vol. 62,pp. 311-314.1984. [22] R. Patel and K. R .Patel , Experimental microbiology. vol 1and 2, Third edition Aditya publication Ahmedabad, India, 2004. [23] J. Vater, B. Kablitz, C. Wilde, P. Franke, N. Mehta, S. Cameotra, “Matrix assisted laser desorption ionization time of flight mass spectrometry of lipopeptide biosurfactants in whole cells and culture filtrates of Bacillus subtilis C-1 isolated from petroleum sludge,” Appl Environ Microbiol., vol. 12 pp. 6210–6219,2002. [24] D.G. Cooper, B.G. Goldenberg “Surface active agents from two Bacillus species,” Appl Environ Microbiol., vol. 53pp. 224–229,1987. [25] H.S. El-Sheshtawy, I. Aiad, M.E. Osman, A.A. Abo-ELnasr, A.S. Kobisy, “Production of biosurfactant from Bacillus licheniformis for microbial enhanced oil recovery and inhibition the growth of sulfate reducing bacteria,” Egyptian Journal of Petroleum, Vol. 24, pp.155–162,2015 [26] Production of biosurfactant from Bacillus licheniformis for microbial enhanced oil recovery and inhibition the growth of sulfate reducing bacteria,” Egyptian Journal of Petroleum, Vol. 24, pp.155–162,2015 [27] A.Daverey and K. Pakshirajan, “Sophorolipids from Candida bombicola using mixed hydrophilic substrates: production, purification and characterization,” Colloids Surf B Biointerfaces. Vol. 79, pp. 246–253, August 2010. [28] T.B. Lotfabad, M. Shourian, R. Roostaazad, A.R. Najafabadi, M.R. Adelzadeh, K.A. Noghabi, “An efficient biosurfactant-producing bacterium Pseudomonas aeruginosa MR01, isolated from oil excavation areas in south of Iran,” Colloids Surf B Biointerfaces, vol. 69, pp. 183–193,2009. [29] M.E.M. Mabrouk, E.M. Youssif, S.A. Sabry, “Biosurfactant production by a newly isolated soft coral-associated marine Bacillus sp. E34: statistical optimization and characterization,” Life Sci. J., vol. 11, pp. 756–768, 2014. [30] I.Siegmund, and F. Wagner, “New method for detecting rhamnolipids excreted by Pseudomonas species during growth on mineral agar,” Biotechnol. Tech., vol. 5, pp. 265-268,1991. [31] A.Bera, T. Kumar, K. Ojha, A. Mandal,”Screening of microemulsion properties for application in enhanced oil recovery,” Fue,l Vol. 121,pp. 198–207,2014. [32] E. J. Gudiña, J.F.B. Pereira, R. Costa, J.A.P. Coutinho, J.A. Teixeira, L. R. Rodrigues, “Biosurfactant-producing and oil-degrading Bacillus subtilis strains enhance oil recovery in laboratory sand-pack columns,” Journal of hazardous materials, Vol. 261, pp. 106–113,2013. [33] Z. Deng, Y. Jiang, K. Chen, J. Li, C. Zheng, F. Gao, X. Liu, “One Biosurfactant-Producing Bacteria Achromobacter sp. A-8 and Its Potential Use in Microbial Enhanced Oil Recovery and Bioremediation,” Frontiers in Microbiology, vol.11, pp 247, February 2020. [34] D.K. Jain, D.L. Collins-Thompson, H. Lee, J.T. Trevors, “A drop-collapsing test for screening surfactant-producing microorganisms,” Journal of Microbiological Methods,Vol. 13, pp. 271-279,1991. [35] J. M. D. A. Câmara, M. A. S. B. Sousa, E. L. Barros Neto and M. C. A. Oliveira, “Application of rhamnolipid biosurfactant produced by Pseudomonas aeruginosa in microbial-enhanced oil recovery (MEOR),” J Petrol Explor Prod Technol., Vol. 9, pp. 2333–2341 2019. [36] S. Jagtap , S. Yavankar, K. Pardesi, B.Chopade, “Production of bioemulsifier by Acinetobacter sp. from healthy human skin of tribal population,” Ind. J. Expt. Biol., vol. 48, 70–76, 2010. [37] D. Cooper, and B. Goldenberg, “Surface active agents from two Bacillus species,” Appl. Environ. Microbiol., vol. 53, pp. 224-229,1987. [38] K.V.Deepika S.Kalam, P.Ramu Sridhar A.R.Podile P.V.Bramhachari,”Optimization of rhamnolipid biosurfactant production by mangrove sediment bacterium Pseudomonas aeruginosa KVD-HR42 using response surface methodology,” Biocatal Agric Biotechno., vol. 5, pp. 38–47. 2016. [39] V. Alvarez, Diogo Jurelevicius, J. Marques, P. M. de Souza, Livia Vieira de Araújo, T. G. Barros, R. D. de Souza, D. Freire, L. Seldin, “Bacillus amyloliquefaciens TSBSO 3.8, a biosurfactant-producing strain with biotechnological potential for microbial enhanced oil recovery,” Colloids Surf B Biointerfaces, Vol. 136, pp. 14–21, December 2015. [40] S. Akbari, A.H. Nour, R.M. Yunus, and A.H. Farhan, A.H. (2018). “Biosurfactants as Promising Multifunctional Agent: A Mini Review, ” International Journal of Innovative Research and Scientific Studies, vol. 1, pp. 1-6, 2018. [41] A.Salihu, A. Ibrahim, and M.N. Almustapha, “An investigation for potential development on biosurfactants,” Biotechnology and Molecular Biology Reviews, vol. 3, pp. 111-117, October, 2009. [42] D. Ratkowsky, J. Olley, T. McMeekin, and A. Ball, (1982). “Relationship between temperature and growth rate of bacterial cultures,” J. Bacteriol., VOL. 149, PP. 1–5, 1982. [43] S.A. Shamaa, and S. Bahjat, “Detection of Rhamnolipid Production in Pseudomonas aeruginosa,” Journal of Physics: Conference Series, vol. 1294. 062083, 2019. [44] M. Hassan, T. Essam, A.S. Yassin ,A. Salama, “Optimization of rhamnolipid production by biodegrading bacterial isolates using Plackett–Burman design,” Int J Biol Macromol., vol. 82: , pp. 573–579, 2016. [45] H. Suthar, K. Hingurao, A. Desai, A. Nerurkar,“Evaluation of bioemulsifier mediated Microbial Enhanced Oil Recovery using sand pack column” Journal of microbiological methods, vol. 75, pp. 225-320, 2008.
Copyright
Copyright © 2022 Niraj Prasad, Sumita Dasgupta, Mousumi Chakraborty, Smita Gupta. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET45784
Publish Date : 2022-07-19
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

