Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Introduction
- Conclusion
- References
- Copyright
Nephroprotective Plants
Authors: Mr. Atul Anil Waghmode , Prof. Khade. P, Dr.Hingane.L.D (M.Pharm PhD.Scholar)
DOI Link: https://doi.org/10.22214/ijraset.2021.39564
Certificate: View Certificate
Abstract
Introduction
I. INTRODUCTION
Man and his domesticated animals have since the time immemorial been largely dependent on plants for the essential for their existence by way of food, clothing, shelter and medicines etc, besides various other uses.Since disease, decay and death always coexisted with life, the study of diseases and their treatment must have also been contemporaneous with the dawn of the human intellect. The primitive man must have used as therapeutical agents and remedial measures those things which he was able to procure most easily. There is no authentic record of medicines used by the primitive man. But the Rigveda which is the oldest book in the library of man supplies curious information on the subject. In his work on plants and animal under domestication, Darwin says "From innumerable experiments made through dire necessity by savages of every land, with the result handed down by tradition, the nutritious, stimulating and medicinal properties of the most of unpromising plants were probably first discovered."
The doctrine of signatures would all account for the use of several plants as medicinal agents. The reason for the extensive use of vegetable drugs may be the fact that plants are everywhere at hand, their number is very great and their focus are distinct and peculiar and these are procured without trouble. It is greatly to the credit of people of India, that they were acquainted with a far large no. of medicinal plants than the natives of any other country on the face of the earth. Many Indian fruits, grains and vegetables employed as useful dietary articles form a chief factor in the cure of diseases, as well as preservation of health and good nutrition. Herbs have always been the principle form of medicine in India and they are becoming popular throughout the world, as people strive to stay healthy in the face of chronic stress and pollution and to treat illness with medicine that work in concert with the body's own defaces. Thus medicinal plants play an important role in the lives of rural people. A plant is said to be medicinal when "at least one part possesses therapeutic properties." One may recognize four stages in the development of the implements in the treatment of disease. In the first stage, crude drug were employed, prepared in the roughest manner, such as powered cinchona or metallic antimony. In the next stage, these were converted to more active and more manageable forms, such as extractions or solutions, watery or alcoholic. In the third stage, the pure active principles, separated from the crude drugs were employed Eg: morphine and quinine. In the 4th stage, instead of attempting to extract out medicine from the natural products in which they are contained, such substances are synthesized which possess particular desired actions.2 Medicinal plants have curative properties due to the presence of various complex chemical substances of different composition, which are found as secondary plant metabolites in one or more parts of these plants. The purpose of pharmaceuticals research is to develop new drugs. The discovery of medicinal value of foxglove (Digitalis purpurea) is the case where traditional herbal knowledge led to major advance in medicine. Phytochemical investigations on plants have not only yielded many compounds of medicinal importance, but have also enriched our knowledge of the subject and understanding of natural products. Ayurveda and siddha system of medicine, the traditional heritage of India include many true tested medicinal plants/drugs for various diseases and to which there is no answer in modern medicine till today.
II. ANATOMY & PHYSIOLOGY OF KIDNEY
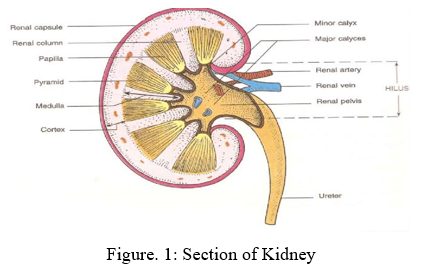
Paired kidneys are reddish bean shaped organs about 10-12cm long, 5-7cm wide, 3cm thick and has a mass of 135-150g.10 The kidneys lie on the posterior abdominal wall, one on each side of vertebral column, behind the peritoneum and below diaphragm. They extend from the level of 12th thoracic vertebrae to 3rd lumbar vertebrae.9 Near the centre of concave boarder is a deep vertical fissure called the renal hilum, through which the ureter emerges from the kidney along with blood vessels, lymphatic vessels & nerves. The kidney consists of two distinct region, outer renal cortex & inner renal medulla. The urine collects to calyx and then to renal pelvis which empties into ureter. The functional unit of kidney is nephron and there are about 1million nephron in each kidney.
III. STRUCTURE OF NEPHRON
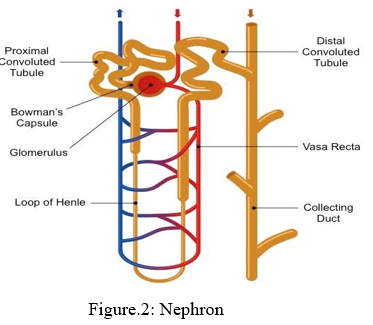
It consists of a tubule closed at one end, and the other end opening into a collecting tubule. The closed end form Bowmann's capsule, which encloses the glomerulus. The remaining parts of nephron is about 3cm long & consist of Proximal convoluted tubule (PCT) Henley's loop Distal convoluted tubule These nephrons are packed tightly to make up the kidney parenchyma.9
IV. FUNCTIONS OF KIDNEY
The main function of kidney can be categorized as Formation of urine Water & electrolyte balance Production of hormones & enzymes In the resting adult, kidney receives 1.2-1.3litres of blood/min. In an adult, the GFR averages 120ml/min. The collecting duct of kidney is an area of fine control of ultrafiltrate composition & volume, where final adjustment in electrolyte composition is made by the action of mineralocortioid & ADH. The hypertonicity of medullary interstitium plays an important role in concentrating the urine. The kidney not only excretes the metabolic substances, but also toxic agents from the body.6 Hence kidney becomes one of the important targets for the toxicity of agents more than other organs in body. Factors that make kidney particularly prone to actions of nephrotoxicity include,High levels of toxins are delivered to the kidney's large blood supply.The large surface area of renal tubular epithelium provide site for toxin interaction & uptake.The availability of specific transport mechanisms that mediate cellular uptakes.The normal concentrating mechanism of kidney can increase concentration of toxins.The presence of the metabolic processes in the renal tubular cell, can release toxic components & induce damage.
A. Renal Failure
The term renal failure primarily denotes failure of the excretory function of kidney, leading to the retention of nitrogenous waste products of metabolism in blood. In addition, there is failure of regulation of fluid & electrolyte balance along with endocrine dysfunction.7 The renal failure is fundamentally categorized into acute renal failure & chronic renal failure.
B. Acute Renal Failure
Acute renal failure is characterized by azotemia that progresses rapidly over several hours or days. It may or may not accompanied by oliguria & there is a sudden & reversible loss of renal function. Histopathology of normal kidney Early recognition of ARF is critical, because it is often asymptomatic. It is detected by measuring serum creatinine level & is more specific than measurement of blood urea nitrogen (BUN). There are many causes of ARF which could be,Pre renal ARF It is due to under perfusion of kidney. It accounted for 21% of ARF cases. It can be thought of as "a good kidney looking at a bad world."
It is quickly reversible with appropriate therapy.Post renal ARF It is caused by obstruction of urinary tract. It accounted for 10% of cases. Intrinsic ARF It is due to disease in parenchyma. It accounted for 69% of cases. Among the renal causes of acute renal failure, acute tubular necrosis is more common accounting for 85% of incidence. ATN occurs due to either ischaemia or toxins. The toxins can be either exogenous or endogenous. The exogenous agents are radiocontrast agents, cyclosporins, antibiotics, chemotherapeutic agents, organic solvents, acetaminophen, & illegal abortifacients.7
C. Chronic Renal Failure
It is a syndrome characterized by progressive & irreversible deterioration of renal due to slow destruction of renal parenchyma, eventually terminating in death when sufficient no. of nephrons have been damaged.8 Various causes are glomerulonephritis, diabetes mellitus, chronic pyelonephritis, hypertension.9 antineoplastic agents like cyclophosphamide, viniristne, cisplatin etc.
V. NEPHROTOXIC AGENTS
Drugs, diagnostic agents & chemical are well known to be nephrotoxic. The following are some of the important nephrotoxic agents.
A. Heavy Metal
Mercury, arsenic, lead, bismuth
B. Antineoplastic Agents
Alkylating agents
Cisplatin, cyclophosphamide
Nitrosoureas: Streptozotocin, Carmustine, Lomustine & Semustine
Antimetabolites
High dose Methotrexate, Cytosine Arabinose, high dose 6-thioguanine, 5-flurouracil Antitumour antibiotics Mitomycin, Mithramycin, Doxorubicin
Biologic agents
Recombinant leukocyte and interferon
C. Antimicrobial Agents
Tetracycline, Acyclovir, Pentamidine, Sulphadiazine, Trimethoprin, Rifampicin
AmphotericinB
D. Aminoglycosides
Gentamicin, Amikacin, Kanamycin, Streptomycin
E. Miscellaneous
Radiocontrast agents
Non-steroidal anti-inflammatory agents: Ibuprofen, Indomethacin, Aspirin etc
VI. ANIMAL MODELS USED IN EXPERIMENTAL STUDIES
A. In Vivo Models
- GM treated albino rat
- Cisplatin treated albino rats
- Cisplatin treated rabbits
- GM treated guinea pigs
- Mercuric chloride treated mice
- Ethylene glycol treated mice
B. In Vitro Models
- Vero cells
VII. PLANT SHOWING NEPHROPROTECTIVE ACTIVITY
A. Effect Of Aerva lanata on Gentamicin & Cisplatin Models of Acute Renal Failure
The ethanol extract of entire plant of Aerva lanata was studied for its nephroprotective activity in cisplatin & gentamicin induced acute renal injury in albino rats of either sex. In the curative regimen, the extract at dose levels of 75,150 & 300mg/kg showed dose dependant reduction in the elevated blood urea and serum creatinine & normalized the histopathological changes in the cisplatin model. In the gentamicin model, the rats in the preventive regimen also showed good response to the ethanol extract at 300mg/kg. The findings suggest that the ethanol extract of Aerva lanata possesses marked nephroprotective activity with minimal toxicity and could offer a promising role in the treatment of acute renal failure caused by nephrotoxins like cisplatin & gentamicin.

B. Protective Effect of Pongamia pinnata Flowers Against Cisplatin & Gentamicin Induced Nehrotoxicity in Rats
When ethanolic extract of flowers of Pongamia pinnata (300 &600mg/kg) was administered orally in rats followed by cisplatin (5mg/kg ip), toxicity of cisplatin as measured by loss of body weight, elevated blood urea & serum creatinine declined significantly. Similarly in gentamicin (40mg/kg sc) induced renal injury, the extract 600mg/kg normalized the raised blood levels of urea & serum creatinine levels. Reversal of cisplatin & gentamicin renal cell damage was confirmed on histopathological examination. The results suggested that the protective effects is through antioxidant property of two flavonoids kaempferol and 3,5,6,7,8-penta methoxy flavone.

C. Salviae Radix Extract Prevents Cisplatin Induced Acute Renal Failure in Rabbits
The present study was carried out to determine if Salviae radix extract (SRE) exerts a beneficial effect against cisplatin induced renalfailure in rabbits. Rabbits were pretreated with SRE orally followed by cisplatin injection (5mg/kg ip). Cisplatin injection caused a reduction in GFR, which was accompanied by an increase in serum creatinine levels. The fractional Na+ excretion and lipid peroxidation were also increased. All these changes were prevented by SRE pretreatment. Cisplatin treatment invitro in renal cortical slices increased LDH release and lipid peroxidation, which were prevented by SRE and its effect may be attributed to its antioxidant action

D. Protective Effect of Gycyrrhizin on Gentamicin Induced Acute Renal Failure in Rats
The effects of glycyrrhizin (200 mg/kg/day) on renal function in association with the regulation of aquaporin 2 water channel in rats with gentamicin (100 mg/kg/day)-induced acute renal failure was investigated. Polyuria in rats with gentamicin-induced acute renal failure was associated with down-regulation of renal aquaporin 2 in the inner and outer renal medulla, and cortex. Glycyrrhizin administration restored the expression of aquaporin 2 with paralleled changes in urine output. Changes in renal functional parameters, such as creatinine clearance, urinary osmolality, and solute-free reabsorption, accompanying acute renal failure were also partially restored after administration of glycyrrhizin. Histological changes in rats with gentamicin-induced acute renal failure were also abrogated by glycyrrhizin treatment. The above results suggest that glycyrrhizin treatment could ameliorate renal defects in rats with acute renal failure induced by gentamicin.
E. Ginkgo biloba Extract Ameliorates GM Induced Nephrotoxicity in Rats
The effect of Ginkgo biloba (EGb), a plant extract with an antioxidant effect, has been studied on gentamicin-induced nephrotoxicity in male wistar rats. Ginkgo biloba extract (300 mg/kg BW) was administered orally concurrently with gentamicin (80 mg/kg BW). Estimations of urine creatinine, glucose, blood urea, serum creatinine, plasma and kidney tissue MDA were carried out after gentamicin treatment. Kidneys were examined using histological techniques. Blood urea and serum creatinine were increased with gentamicin. Creatinine clearance was significantly decreased with gentamicin. Changes in blood urea, serum creatinine and creatinine clearance induced by gentamicin were significantly prevented by Ginkgo biloba extract. There was a rise in plasma and kidney tissue MDA with gentamicin, which were significantly reduced to normal with Ginkgo biloba extract. Histomorphology showed necrosis and desquamation of tubular epithelial cells in renal cortex with gentamicin, while it was normal with Ginkgo biloba extract. These data suggest that supplementation of Ginkgo biloba extract may be helpful to reduce gentamicin nephrotoxicity.

F. Effect of Cassia auriculata Root Extract on Cisplatin & GM Induced Renal Injury
The ethanol extract of the roots of Cassia auriculata was studied for its nephroprotective activity in cisplatin- and gentamicin-induced renal injury in male albino rats. In the cisplatin model, the extract at doses of 300 and 600 mg/kg body wt. reduced elevated blood urea and serum creatinine and normalized the histopathological changes in the curative regimen. In the gentamicin model, the ethanol extract at a dose of 600 mg/kg body wt.
Reduced blood urea and serum creatinine effectively in both the curative and the preventive regimen. The extract had a marked nitric oxide free-radical-scavenging effect. The findings suggest that the probable mechanism of nephroprotection by C.auriculata against cisplatin- and gentamicin-induced renal injury could be due to its antioxidant and free-radical-scavenging property.

G. Aged Garlic Extract Attenuates GM Induced Renal Damage and Oxidative Stress in Rats
Aged garlic extract (AGE), an antioxidant, has a protective role in this experimental model of male Wistar rats were studied. AGE was given at a dose of (1.2 mL/kg/12 hours) followed by GM (70 mg/kg/12 hours). Nephrotoxicity was made evident by:

- The increase in blood urea nitrogen and plasma creatinine
- The decrease in plasma glutathione peroxidase (GPx) activity and the urinary increase in N-acetyl-beta-D-glucosaminidase activity and total protein
- Necrosis of proximal tubular cells
- Increase in the renal levels of oxidative stress markers: nitrotyrosine and protein carbonyl groups and the decrease in manganese superoxide dismutase (Mn-SOD), GPx, and glutathione reductase (GR) activities.
These alterations were prevented or ameliorated by AGE treatment. Furthermore, AGE prevented the GM-induced The protective effect of AGE was associated with the decrease in the oxidative stress and the preservation of Mn-SOD, GPx, and GR activities in renal cortex. These data suggest that AGE may be a useful agent for the prevention of GM-nephrotoxicity.
H. The Effects OF Nigella sativa OIL ON GM Nephrotoxicity In Rats
In this work, tested whether oral treatment of rats with N. sativa oil (0.5, 1.0 or 2.0 ml/kg/day) would ameliorate nephrotoxicity of GM (80 mg/kg/day im) concomitantly with the oil. Nephrotoxicity was evaluated histopathologically and by measurement of concentrations of urea, creatinine and total antioxidant status (TAS) in plasma and reduced glutathione (GSH) and TAS in kidney cortex. The results indicated that GM treatment caused moderate proximal tubular damage, significantly increased the concentrations of creatinine and urea, and decreased that of TAS and GSH. Treatment with N. sativa oil produced a dose-dependent amelioration of the biochemical and histological indices of GM nephrotoxicity that was significant at the two higher doses used, and it increased GSH and TAS concentrations in renal cortex and enhanced growth. The results suggest that N. sativa may be useful in ameliorating signs of GM nephrotoxicity in rats.

I. Flavonoid of Drynaria fortunei Protects Against ARF
The flavonoid fraction (FF) from Drynaria fortunei was investigated to determine its biological activity expression in three acute renal failure animal models Guinea pigs & mercuric chloride treated mice. Guinea pigs received 100 mg/kg of gentamicin & 10 mg/kg of FF. FF treatment prevented the GM toxicity, ie; the increase in BUN and creatinine levels. Mice were treated once with 6 mg/kg of mercuric chloride, followed by 10 mg/kg of FF. BUN and creatinine levels were found to be significantly higher on the mercuric chloride treatment and are ameliorated by FF treatment. In conclusion, the present study suggests that FF prevents nephrotoxicity, improves kidney function and promotes kidney primary epithelial tubular cell regeneration.

VIII. DISCUSSION
This study was conducted to establish the nephroprotective activity of plants. Various models have been used to substantiate the nephroprotective activity of herbals. They were GM in albino rats, cisplatin in rabbits, mercuric chloride in mice, ethylene glycol in mice etc. These nephrotoxic agents caused nephropathy mainly due to their free radical generation in kidney tissues. And the kidney damage was indicated by changes in renal function parameters like creatinine, BUN, and the enzymes suchn as GPx, SOD and was also confirmed histopathologically. Above works certified that, by ameliorating all the allied effects, mainly due to antioxidant property the plants like A.lanata, P.pinnata, C.auriculata, S.radix, G.glabra, G.biloba, N.sativa, D.fortunei, T.terrestris, C.nurvala, O.sanctum, S.nigrum, V.vinifera have nephroprotective activity.
Conclusion
As we gone through various studies on treatment of kidney disorders, we can conclude that herbal plants play a unique role in medicine. There is no synthetic drug which relieves overall insufficiency of kidney. But indigenous plants possess tissue rejuvenator property which is anyway unavoidable. To Indians, who are brought upon Indian food, soul & climate with Indian habits of life and environment, Indian drugs naturally suit better and safer than European constitution built upon their peculiar food, climate, habits and manner of life. This may perhaps be the reason why in numerous cases,where synthetic medicines fails, Indiginous system of medication succeed.
References
[1] Singh N.P., 1988. Flora of Eastern Karnataka,1. Published by: Mittal Publications, India. PP: 1-3. [2] Kirtikar K R & Basu B D, Indian Medicinal Plants. Vol.1 pp: 5-6 [3] Dr.K M Nadkarni & A K Nadkarni Indian Materia Medica. Vol. 1 pp:1-4 [4] Prajapathi, Purohit, Sharma & Kumar, A Hand Book of Medicinal Plants [5] Brenner B M, The Kidney, 6th ed1, Published by W B Saunders Co. USA pp:1563-1564 [6] Williams P L 1995, The Anatomical Basis of Medicine & Surgery. 38th ed, Published by ELBS with Churchill Livingstone Publication, Britain, 1814-45 [7] Eugene B & Stephen L 2001, Principles of International Medicine, 15th ed, Mc Graw-Hill Medical Publishing division 1535-1626 [8] Harshmohan, Text Book of Pathology, 5th ed, 675-679 [9] Ross & Wilson, Anatomy and Physiology in Health & Illness, 340-346 [10] Gerard I Tortora, Sandra Reynolds Grabowski, Principles of Anatomy & Physiology, 10th ed, 950 [11] Schrier R W, Gottschalk C W 1993, Disease of kidney, 5th ed, 2, Published by Little Brown & Co, 1031-1165 [12] Davison A M, Cameron J S, Grunfeld J P, Kerr D N S 1988, Oxford Textbook of Clinical Nephrology 2nd ed. 3, Published by Oxford University Press, 2650-53 [13] Stratta P, Canavese C, Dogliani M Experimental Evidences of Mesangiolysis, 37-41 [14] Cheeseman K H, Slater t f,1993.Free Radical in Medicine 49(3)4 [15] Stybbe 1990.Journal of Biological Chemisty, 265,5329-32. [16] Simmons C F,Bogusky R T, Humes H D, 1980 J of Pharmacology & Experimental Therapeutics 214,709-15 [17] Shirwaikar A, Issac D, Malini S, Effct of Aerva lanata on cisplatin & GM models of ARF, Ethnopharmacol 2004 Jan: 90(1)..81-6 [18] Shirwaikar A etal, Protective Effect of P.pinnata flowers against cisplatin & GM induced nephrotoxicity in rats, Indian J Exp.Biol 2003 Jan; 41(1), 58-62 [19] Jeong etal, S.radix extract prevents cisplatin induced ARF in rabbits, Nephron 2001 Jul 88(3), 241-6 [20] Sohn E J etal, Protective Effect of Glycyrrhizin on GM induced ARF in rats, Pharmacol Toxicol 2003 Sep 93(3), 116-22. [21] Naidu M U etal, Ginkgo biloba extract ameliorates GM induced nephrotoxicity in rats, Phytomedicu\\ine 2000 jun 7(3), 191-7 [22] Annie S, Rajgopal P L, Malini S, effect of C.auriculata root extract on cisplatin & GM induced renal injury, Phytomedicine 2005 Aug 12(8), 555-60. [23] Maldonado P D etal, Aged garlic extract attenuates GM inducwd renal damage & oxidative stress in rats, Life Sci. 2003 Oct 3; 73(20) : 2543-56 [24] Ali B H, The effect of N.sativa oil on GM nephrotoxicity in rats, Amj Chin Med 2004 32(1) 49-55.
Copyright
Copyright © 2022 Mr. Atul Anil Waghmode , Prof. Khade. P, Dr.Hingane.L.D (M.Pharm PhD.Scholar). This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET39564
Publish Date : 2021-12-21
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

