Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Parkinson\'s Disease Detection Using Deep Learning Technique
Authors: Kalaiyarasi. I, Amudha. P, Sivakumari. S
DOI Link: https://doi.org/10.22214/ijraset.2023.51916
Certificate: View Certificate
Abstract
Parkinson’s Disease is a degenerative nervous system ailment primarily impacting middle-aged and older persons. Tremors, stiffness of the muscles, and slow, clumsy movement mark it. Parkinson\'s Disease is believed to be caused by genetic and environmental factors, while its precise cause is yet unclear. Levodopa can aid patients\' quality of life and manage their symptoms, but there is no proven treatment for Parkinson\'s Disease. A unique deep-learning approach is developed to determine whether a person has PD based on premotor traits. This study has looked explicitly at several signs to identify PD at an early stage using spiral drawing. Measuring the changes in the handwritten spiral drawing allows for the early and accurate diagnosis of PD. Deep learning algorithms have been used to track the development of the illness and the effectiveness of treatment in PD patients in addition to diagnosis and prediction. By utilizing efficient treatments and medications, the findings will showcase how early illness detection can enhance a patient\'s life expectancy and enable them to live peacefully. One commonly utilized deep learning technique in PD research is Convolutional Neural Networks (CNNs). This work uses Modified Convolutional Neural Networks (MCNN) to predict the normal and abnormal of Parkinson\'s Disease. The complete model\'s performance after being trained on data from 36 patients was 96% overall accuracy, with average recall, precision, and f1 scores of 96.73%, 96.05%, and 96%, respectively.
Introduction
I. INTRODUCTION
Parkinson's Disease (PD) is a severe central nervous system degenerative condition that impairs the quality of life for millions of older people worldwide. Due to the Disease's heterogeneity, each person's PD symptoms may develop uniquely. When at repose, people with Parkinson's Disease may exhibit symptoms, notably tremors[1].The primary symptoms of PD are motor symptoms, while non-motor symptoms are secondary symptoms. The initial symptoms of PD are Bradykinesia refers to slowness of movement and muscular stiffness. Loss of smell, trouble sleeping, mental illness linked to depression, speech problems, and altered vision are some non-motor symptoms. The number of persons with PD over 50 was between 4.1 million and 4.6 million in 2005, and there is a probability that the number may quadruple by 2030, according to PD prediction research[4].
Many health informatics tools have detected and monitored significant illnesses in recent years. PD, typically found in adults over 60, is observed using information systems built on artificial learning[2]. Neuronal death is the typical cause of PD. Dopamine is a neurotransmitter produced by neurons, and its primary function is modulating how the body moves. Hence, as neurons die, the brain produces less dopamine. As a result, this neurological disorder develops extremely gradually and affects many brain communication types[3].
The Activities of Daily Living (ADL) Part 14, the Freezing of Gait Questionnaire (FOG-Q), the Unified PD Rating Scale (UPDRS) [7], and other clinician-mediated questionnaires are still the mainstays of current clinical evaluation approaches for PD patients. In actual practice, trained doctors frequently assess a PD patient's severity concerning how well they complete tasks specified in questionnaires. This subjective, labor-intensive approach has a history of delivering erratic findings. Thus, its use in diagnosing and monitoring is limited. As a result, the diagnosis in modern clinical practice needs routine assessment since specific "red flags" characteristics, such as fast advancement or early falls, might indicate a related disease comparable to developing supranuclear palsy or various systems atrophy [5].
A person with PD (PD) and a person without PD may be easily separated from one another using non-invasive techniques such as drawing shapes like spirals or waves or other handwritten words. The early stages of Parkinson's Disease (PD) have been associated in some manner with spiral doodles and handwriting. The primary problem of this diagnosis is that accurate handwriting and drawing interpretation are required.
In the past, translators who were specialists in the topic manually read the writing or sketches made on paper. Digitally doing these tasks is more accessible, and machine evaluations are more accurate and precise than those made the old-fashioned way [6].
Artificial intelligence (AI) will make it possible to create an effective early detection system. Artificial intelligence (AI) aims to mimic certain aspects of human intelligence. The two main subfields of artificial intelligence are machine learning (ML) and deep learning, and they are applied in almost every sector (DL). Several CDSS, telemonitoring, and telediagnosis systems are available, including AI for PD diagnosis using non-invasive data modalities [4].While the earlier study used single-type feature sets for PD classification using CNN, such as EEG data, Spiral images, and sensor activity data. A CNN's fundamental building blocks are its convolutional layers, consisting of filters (kernels) dragged over the input picture to extract characteristics like edges, corners, textures, or forms. The convolutional layer's output is a series of feature maps showing where the learned features are across the picture. The pooling layers minimize the feature maps' spatial dimensions by condensing the data into small areas. Max pooling, which chooses the highest value inside each local window, is the most used pooling procedure. At the network's edge, fully connected layers use the collected features to perform the classification or regression task.
In CNN, weights are learned via backpropagation and gradient descent on a sizable labeled dataset to reduce the prediction error. In various image and video identification tasks, including object detection, picture segmentation, facial recognition, and scene understanding, CNNs have attained cutting-edge performance [1].CNN with several parallel layers has yet to be applied for categorization. To reduce the training time without reducing any essential features of CNN modified convolution neural network was created. Experiment findings on publicly accessible datasets demonstrate that the suggested MCNN architecture performs better than single-layered CNN classifiers, which use parallel convolution branches.
This section discusses, the theoretical foundations of the research, starting with an explanation of PD and moving on to symptoms of PD, neural networks, related work, and PD diagnosis challenges. The following is the breakdown of the essay: Existing works are presented in Section 2. The model's approach, components, steps, and architecture are covered in Section 3 of this article. The study's findings are presented in Section 4. The study's commentary is provided in Section 5. The study's findings and recommendations for further research are provided in Section 6.
II. LITERATURE SURVEY
Specific machine learning and deep learning research discoveries have been provided below to aid in our study. These approaches may be used to build an automated system using various datasets linked to Parkinson's illness.
Memedi et al., suggested research based on Spiral data acquired by telemetry touchscreen devices in the home surroundings. To use machine learning algorithms to separate off episodes and peak dosage dyskinesia, The machine learning classifiers received several attributes from the data. Various techniques have been utilized to train automated system features, including Support Vector Machine, Logistic Regression, Random Forest, and Multilayer Perceptron (MLP). With an accuracy of 84%, MLP performed the best of all the classifiers[7].
A?uro?luww et al., the study is the first and only one to emphasize predicting the severity of PD symptoms more than making a binary diagnosis. This study developed a locally weighted random forest (LWRF) to exploit correlations between gait signals. Time and frequency information are collected and fed into the LWRF model from gait data. They provided data for the Mean Absolute Error (MAE), the Root Mean Square Error (RMSE), and the Correlation coefficient (CC) of symptom severity predictions, and they outperformed past studies on binary classification systems [8].
Zeng et al., implemented a method to predict PD, and a multiphase classifier based on gait signals was built. Essential characteristics like left-right sensor value differences and output aggregation are part of the feature extraction step. A neural network with a radial foundation is fed these feature vectors [9].
Luukka et al., discovered how to achieve excellent results using fewer elements than in the first informative indexes. Furthermore, grouping accuracy was significantly improved by adding informational collections on Parkinson's and dermatology. Using only two highlights from a unique 22, the mean characterization precision using Parkinson's informational index was 85.03%. With 29 highlights as opposed to 34 different highlights, the informative dermatological gathering achieved a mean accuracy of 98.28%. The results are pretty impressive [10].
Chen et al., suggested a PD diagnosis system that uses Fuzzy KNN for Classification and PCA for feature extraction [11]. Priya et al., investigated several local binary patterns approaches' effects on diagnosing PD. They used LBP techniques to alter the gait data. They then retrieved statistical characteristics from these patterns after modification. They compared the classification abilities of KNN, SVM, and Logistic Regression models in their tests. They said the logistic regression model incorporating LBP patterns attained maximum accuracy, precision, and recall [12].
Jie Mei et al., utilized all fundamental deep-learning algorithms to find PD. For example, Radial Basis Function Networks (RBF), Deep Belief Networks (DBN), Decision Trees, Radial Basis Function Networks, ANN, and KNN. PD must be diagnosed as soon as possible. Using a data mining approach, the identification may be carried out. This paper provides a theoretical justification for the PD detection methods, including Decision tree, Multilayer Perceptron Neural Network, Support Vector Machine, and Naive Bayes. To forecast PD, this study analyses voice input from acoustic equipment. This study looks at people from different backgrounds and speech characteristics to forecast PD in patients. Using multilayer perceptron and logistic regression (LR) frameworks, a voice dataset was utilized to identify PD [13].
Shail Raval et al., incorporate all components, such as biological, chemical, and genetic data, to identify PD. In this study, the primary emphasis was placed on the symptoms, such as stiffness, tremor at rest, voice changes, etc. The authenticity check, duplication check, and defective node detection are suggested methods for safe data transfer. Long transmission distances can be covered using the suggested strategy. It also backs the idea of retransmission [14].
One of the most critical issues is Parkinson's disorder prediction, which must be identified in the earliest stages of the Disease's onset to slow the rate at which people develop the condition. There have been numerous attempts to identify the root cause.
Some of these efforts have advanced to the point where they have proposed a system that uses various machine learning techniques to distinguish between healthy individuals and those with any neurodegenerative disorder. Several feature selection, pre-processing, and classification approaches have been applied and developed.
III. METHODOLOGY
The proposed model's block diagram is seen in Figure 1. The testing phase and the training phase are two phases. The testing data is then used to assess the trained model to assess its performance in detecting PD from Spiral drawing data. Each phase is divided into four sections. The first one is, importing datasets for two categories (Normal and Parkinson) collected from a data science website called Kaggle. These data are collected, placed in different folders with respective disease names, and uploaded for pre-processing. After data pre-processing, the CNN training model is created using the Modified Convolution Neural Network (MCNN) Architecture. In the next step, deep, the neural network is designed with an input, hidden, and output layer. After training the datasets, the model file is created. The test image is imported during Classification, pre-processed CNN prediction is made using the model file, and the result is classified.
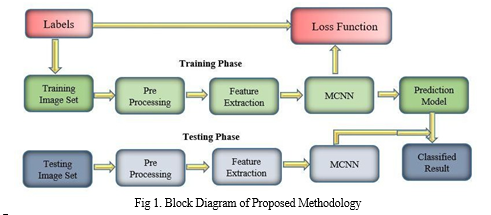
A. Data set Description
For the proposed study, spiral images of normal and affected persons are taken from the Kaggle dataset. Figure 2 and Figure 3 represent the average and affected person's Spiral drawing image data, respectively. Drawings of spirals from 112 people are included in this collection, including 42 healthy individuals and 70 persons with PD. Each person underwent the spiral drawing exam. An A3-sized tablet that is often used was used to record the spiral pattern. A3 paper was put over the tablet, and the Spiral was sketched using an ink pen[6]. Five-time series for each drawing was kept track of. Each recording contains data regarding X, Y, and Z coordinates, pressure, and a grip angle that was collected using the device's API capabilities. The data were gathered in two phases at different sampling rates of 110 Hz and 140 Hz. All recordings were rescaled to the same 110 Hz sampling rate to maintain uniformity. The sample sequence was broken down into 3-second windows (330 samples per window) and spaced apart by 0.5 seconds (resulting in a 2.5-second overlap between two successive windows).

B. Pre-processing
The pre-processing phase for detecting PD using spiral drawing involves several steps to prepare the input data for a modified convolutional neural network (MCNN) model.
- The first step is to collect the data, which typically involves having PD patients draw spirals using a digital pen or a touchscreen device. The spirals are then recorded as a sequence of (x, y) coordinates over time, stored as discrete points.
- The data must be processed in the second stage to get rid of any potential noise or artifacts. This may entail applying a low-pass filter to the data to reduce high-frequency noise or applying methods like smoothing or interpolation to fill in any missing or noisy data points.
- The third step is normalizing the data to ensure all spirals have the same scale and orientation. This can involve centering the spirals around their centroid, scaling them to a fixed size, and rotating them to a standardized orientation.
- The fourth step is to extract relevant features from the normalized spirals, such as curvature, speed, and direction, which are then used as input to the MCNN model. This feature extraction process can be done using Fourier analysis, wavelet analysis, or principal component analysis (PCA) techniques.
Figure 6 shows the pre-processed Spiral image. The pre-processed data is split into training and testing sets, with the training data being used to train the MCNN model. The performance of the trained model in recognizing PD from Spiral drawing data is then assessed using testing data[15].

C. Feature Extraction
- Curvature: The curvature of the Spiral can be an essential feature that can differentiate between healthy and Parkinson-affected individuals. The curvature can be calculated by measuring the change in angle between successive points along the Spiral.
- Smoothness: Parkinson's Disease affects motor control, resulting in tremors and jerky movements. The smoothness of the Spiral can be measured by calculating the variation in distance between successive points along the Spiral.
- Symmetry: Parkinson's Disease can affect the symmetry of the Spiral. The symmetry of the Spiral can be measured by comparing the left and right halves of the Spiral.
- Speed: Parkinson's Disease can slow down motions by affecting movement speed. One may determine its speed by counting the amount of time it takes to complete the Spiral.
- Amplitude: Parkinson's Disease can affect the amplitude of movements, resulting in smaller movements. The amplitude of the Spiral can be calculated by measuring the distance between the center of the Spiral and the farthest point on the Spiral.
These features can be extracted using image processing techniques such as edge detection, line fitting, and curve fitting. In order to categorize the Spiral photos as either healthy or impacted by Parkinson's, the extracted characteristics may then be fed into a deep learning model, such as a convolutional neural network (CNN).
D. Building the MCNN
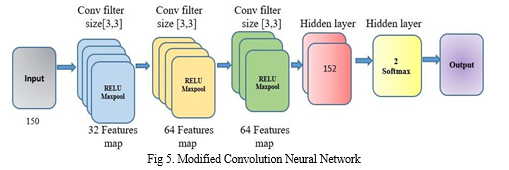
AI and machine learning have brought about technological progress, including medical diagnosis. Identifying Parkinson's Disease using machine learning has shown promise, with CNNs successfully extracting features from medical images. Training time for CNNs has been challenging, but researchers have developed modified architectures to address this issue. The modified convolution architecture presented in Figure 5 is a powerful tool for reducing training time without sacrificing essential features. With three convolutions and two hidden layers, the designers created this model to extract critical features from input images and classify them accurately.
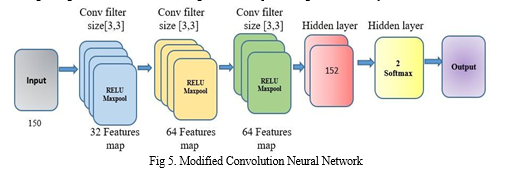
To pre-empt this occurrence, the initial course of action involves transforming the image resolution into a standardized 200*200 dimension, which is then subjected to a tri-layered convolution process, each utilizing a 3*3 kernel with relu activation to interpret the positive part of its argument. We execute the max-pooling strategy to refine the extracted features further. Once the convolutional layers have removed the relevant features, fully connected layers use these features to classify the Spiral image. The two hidden layers have a denso of 128, and the final layer utilizes SoftMax activation to ensure accurate Classification. We train the architecture using a dataset of Spiral images and create an MCNN model file. The result of the fully - connected layers is a probability distribution that assigns the input vision a likelihood of PD or healthy. The modified convolution architecture is a valuable tool in diagnosing PD and can reduce training time and improve diagnostic accuracy.
IV. EXPERIMENTAL RESULTS
The multistage classifier created in this study using convolutional neural network techniques showed some results in identifying Parkinson's Disease from Spiral pictures. The model created for the study produced average f1 scores of 96%, an accuracy of 96%, a recall average of 96.73%, a precision average of 96.05%, and so on. The model's generalizability was further tested using 5-fold cross-validation on the complete dataset. The model showed a constant tendency toward accuracy and remembered throughout random data folds of training, validation, and testing. Additionally, the model generalized rather well between the two classes.
The Confusion Matrix of the proposed system is shown in Figure 6.1. The figure demonstrates that the model's performance is wholly compatible with the primary hypothesis.
In addition, the confusion matrix reveals that one misclassification in the Healthy class was incorrectly identified as Parkinson's Disease. In contrast, four misclassifications in the Affected class were incorrectly identified as Parkinson's Disease. The sketches created by those particular persons were found to be exceedingly distorted, which is pretty akin to the drawings of people with Parkinson's, after carefully studying the likelihood of those misclassified samples.Consequently, since the healthy individuals were all chosen by matching their ages, likely, the Parkinson's patient who is well and predicted to develop the Disease may be a progression subject. Figures 6.2 and 6.3 illustrate the model's performance with and without the dropout layer. It should be emphasized that without the dropout layer, data overfitting is possible. In Figure 6.2, the accuracy of the training set is not significantly different from the accuracy of the validation set. Figure 6.3 shows that the validation set's accuracy is significantly lower than the training data's.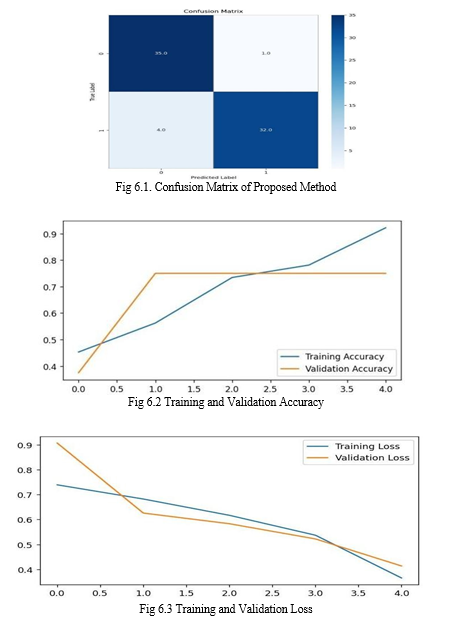
V. DISCUSSION
Kaggle's Spiral image dataset contains drawings by participants with Parkinson's Disease and healthy controls to aid diagnosis through analysis. Analyzing spiral drawings may help detect Parkinson's Disease early, but variability in quality and style among participants poses a challenge. Careful pre-processing and creating robust algorithms are necessary. The dataset's transferability to other populations is still being determined, and further research is required to confirm the accuracy of using Spiral drawings for diagnosis. Pre-processing is crucial for analyzing Spiral drawings to detect Parkinson's Disease. It involves image cropping, filtering, normalization, and feature extraction. These steps remove unwanted elements, improve image quality, standardize images, and extract relevant information for Classification. Curvature, smoothness, symmetry, speed, and amplitude are essential for differentiating between healthy and Parkinson-affected individuals. These features can be extracted using image processing techniques and input to a deep learning model, such as a CNN, to classify Spiral images.AI and machine learning have advanced medical diagnosis, including identifying Parkinson's Disease using CNNs to extract features from medical images. A modified convolutional architecture with three convolutions and two hidden layers can reduce training time without sacrificing essential features. The input image is transformed into a standardized dimension, subjected to convolution, and refined using max-pooling. Fully connected layers use these features to classify the Spiral image, and SoftMax activation ensures accurate Classification. The architecture is trained using a Spiral image dataset to create an MCNN model file, which assigns a likelihood of PD or healthy to the input image. Finally, it was discovered that the spiral image generated the prediction.
Conclusion
In this paper, using Spiral drawing in Modified convolution neural networks for PD detection is a promising development in medical research. Spiral drawing is one of the early symptoms of Parkinson\'s Disease, and assessing drawing motions is simple to undertake because it doesn\'t call for any invasive treatments. The spectrum input idea for an MCNN for PD detection has been essential. The PD Spiral Drawings identification method makes use of a public dataset., MCNN prediction obtained the best results in detecting Parkinson\'s and healthy individuals accurately, where one indicates Parkinson\'s and 0 indicates healthy. This system represents a reliable, non-invasive way to detect PD. We can analyze any hand as a result. Additionally, the readings from the two axes are nearly equal for both hands. Thus, we can consider any one axis for implementing such a system. We must collect sufficient data for a confirmed diagnosis of Parkinson\'s patients. The model created for the study had an accuracy of 96%, an average recall of 96.73%, an average precision of 96.05%, and an average f1 score of 96%. Further studies are needed to improve the model\'s efficiency and expand the datasets for better generalization. Nothing less, this technology has the potential to revolutionize the way PD is detected and, ultimately, improve the lives of those affected by this debilitating condition.
References
Wang, W. et al. (2020) ‘Early detection of parkinson’s disease using Deep Learning and machine learning’, IEEE Access, 8, pp. 147635–147646. doi:10.1109/access.2020.3016062. Gunduz, H. (2019) ‘Deep learning-based parkinson’s disease classification using vocal feature sets’, IEEE Access, 7, pp. 115540–115551. doi:10.1109/access.2019.2936564. Oh, S. L., Hagiwara, Y., Raghavendra, U., Yuvaraj, R., Arunkumar, N., Murugappan, M., & Acharya, U. R. (2018, August 30). A deep learning approach for Parkinson’s disease diagnosis from EEG signals. Neural Computing and Applications, 32(15), 10927–10933. https://doi.org/10.1007/s00521-018-3689-5 Anisha. C. D., & Dr. Arulanand. N. (2022, March 5). An Intensive Review on The Symptoms and Advancements In Diagnosis System Of Parkinson Disorder (PD). International Journal of Scientific Research in Computer Science, Engineering and Information Technology, 66–74. https://doi.org/10.32628/cseit22827 Xia, Y., Yao, Z., Ye, Q., & Cheng, N. (2020, January). A Dual-Modal Attention-Enhanced Deep Learning Network for Quantification of Parkinson’s Disease Characteristics. IEEE Transactions on Neural Systems and Rehabilitation Engineering, 28(1), 42–51. https://doi.org/10.1109/tnsre.2019.2946194 Mr. Zaki Shaikh, Mr. Viraj Tilekar, Mr. Atharva Pawar, Mr. Vedant Suryawanshi, & Mr. Nitin R. Talhar. (2022, November 30). Parkinson Disease Detection from Spiral and Wave Drawings using Machine Learning Algorithm. International Journal of Advanced Research in Science, Communication and Technology, 252–258. https://doi.org/10.48175/ijarsct-7631 Memedi, M., Sadikov, A., Groznik, V., Žabkar, J., Možina, M., Bergquist, F., Johansson, A., Haubenberger, D., & Nyholm, D. (2015, September 17). Automatic Spiral Analysis for Objective Assessment of Motor Symptoms in Parkinson’s Disease. Sensors, 15(9), 23727–23744. https://doi.org/10.3390/s150923727 A?uro?lu, T., Aç?c?, K., Berke Erda?, A., K?l?nç Toprak, M., Erdem, H., & O?ul, H. (2018). Parkinson’s disease monitoring from gait analysis via foot-worn sensors. Biocybernetics and Biomedical Engineering, 38(3), 760–772. https://doi.org/10.1016/j.bbe.2018.06.002 Zeng, W., Liu, F., Wang, Q., Wang, Y., Ma, L., & Zhang, Y. (2016, October). Parkinson’s disease classification using gait analysis via deterministic learning. Neuroscience Letters, 633, 268–278. https://doi.org/10.1016/j.neulet.2016.09.043 Ramig, L. A., Titze, I. R., Scherer, R. C., & Ringel, S. P. (1988, March). Acoustic Analysis of Voices of Patients with Neurologic Disease: Rationale and Preliminary Data. Annals of Otology, Rhinology & Laryngology, 97(2), 164–172. https://doi.org/10.1177/000348948809700214 Chen, H. L., Huang, C. C., Yu, X. G., Xu, X., Sun, X., Wang, G., & Wang, S. J. (2013, January). An efficient diagnosis system for detection of Parkinson’s disease using fuzzy k-nearest neighbor approach. Expert Systems With Applications, 40(1), 263–271. https://doi.org/10.1016/j.eswa.2012.07.014 Priya SJ, Rani AJ, Ubendran N. Improving the prediction accuracy of Parkinson’s Disease based on pattern techniques. In: Proceedings 5th International Conference on Devices, Circuits and Systems (ICDCS), 2020, pp. 188–192. Jie Mei, Christian Desrosiers and Johannes Frasnelli, “Machine Learning for the Diagnosis of Parkinson’s Disease” Front Aging Neuroscience, 2021. ShailRaval,“A ComparativeStudy of Early Detection of Parkinson’s Disease using Machine Learning Techniques”, Dept. Of Information Technology Vishwakarma Government Engineering College Ahmedabad, India, pp. 509-516, 2020. Shah, H., & Mufti, U. R. (2021). Detection of Parkinson\'s Disease through Spiral Drawing Analysis: A Comprehensive Review. Journal of Healthcare Engineering, 2021. https://doi.org/10.1155/2021/6631027 Zhao A, Qi L, Li J, Dong J, Yu H. A hybrid spatio-temporal model for detection and severity rating of Parkinson’s disease from gait data. Neurocomputing. 2018;315:1–8 Abdulhay E, Arunkumar N, Narasimhan K, Vellaiappan E, Venkatraman V. Gait and tremor investigation using machine learning techniques for the diagnosis of Parkinson disease. Futur Gener Comput Syst. 2018;83:366–73 Noella N, Gupta D, Priyadarshini J. Diagnosis of Parkinson\'s disease using gait dynamics and images. Proc Int Conf Recent Trends Adv Comput. 2019. Bouchikhi, S., Boublenza, A., Benosman, A., & Chikh, M. A. (2013). Parkinson’s disease Detection With SVM classifier and Relief-F Features Selection Algorithm. South East Europe Journal of Soft Computing, 2(1) Tu, M., Berisha, V. and Liss, J. (2017) “Interpretable objective assessment of Dysarthric speech based on Deep Neural Networks,” Interspeech 2017 [Preprint]. Available at: https://doi.org/10.21437/interspeech.2017-1222 Oh, S.L. et al. (2018) “A deep learning approach for parkinson’s disease diagnosis from EEG signals,” Neural Computing and Applications, 32(15), pp. 10927–10933. Available at: https://doi.org/10.1007/s00521-018-3689-5 Chakre, R.R. and Patil, D.V. (2021) “Particle rider mutual information and dendritic-squirrel search algorithm with artificial immune classifier for brain tumor classification,” The Computer Journal [Preprint]. Available at: https://doi.org/10.1093/comjnl/bxab194. Seeja K.R., Srishti Grover, Saloni Bhartia, Akshama, and Abhilasha Yadav. “Predicting Severity Of Parkinson’s Disease Using Deep Learning”. Procedia Computer Science, 132:1788–1794 Wingate, J. et al. (2020) “Unified deep learning approach for prediction of parkinson\'s disease,” IET Image Processing, 14(10), pp. 1980–1989. Available at: https://doi.org/10.1049/iet-ipr.2019.1526 Kamble, M., Shrivastava, P. and Jain, M. (2021) “Digitized spiral drawing classification for parkinson\'s disease diagnosis,” Measurement: Sensors, 16, p. 100047. Available at: https://doi.org/10.1016/j.measen.2021.100047 Grover, S. et al. (2018) “Predicting severity of parkinson’s disease using Deep Learning,” Procedia Computer Science, 132, pp. 1788–1794. Available at: https://doi.org/10.1016/j.procs.2018.05.154 [27] Celik, O. (2021) “Detection of impacted teeth using deep learning technique.” Available at: https://doi.org/10.7546/crabs.2021.02.14 [28] J. C. Vasquez-Correa ´ et al., “Multi view representation learning via GCCA for multimodal analysis of Parkinson’s disease,” inProc. Int. Conf. Acoust., Speech, Signal Process., 2017, pp. 2966–2970 Y. Nancy Jane, H. Khanna Nehemiah, and K. Arputharaj, “A Q-backpropagated time delay neural network for diagnosing severity of gait disturbances in Parkinson’s disease,” Journal of Biomedical Informatics, vol. 60, pp. 169-176, 2016/04/01/, 2016 S. Frenkeltoledo, N. Giladi, C. Peretz, T. Herman, L. Gruendlinger, and J. M. Hausdorff, “Treadmill walking as an external pacemaker to improve gait rhythm and stability in Parkinson\'s disease,” Movement Disorders, vol. 20, no. 9, pp. 1109-1114, 2010 A. Zhao, L. Qi, J. Li, J. Dong, and H. Yu, “A hybrid spatio-temporal model for detection and severity rating of Parkinson’s disease from gait data,” Neurocomputing, vol. 215, pp. 1-8, 2018 G. Yogev, N. Giladi, C. Peretz, S. Springer, E. S. Simon, and J. M. Hausdorff, “Dual tasking, gait rhythmicity, and Parkinson\'s disease: which aspects of gait are attention demanding?,” European Journal of Neuroscience, vol. 22, no. 5, pp. 1248-1256, 2010 R. Djaldetti, I. Ziv, and E. Melamed, “The mystery of motor asymmetry in Parkinson\'s disease,” Lancet Neurology, vol. 5, no. 9, pp. 796-802, 2006 B. Su, R. Song, L. Guo, and C. Yen, “Characterizing gait asymmetry via frequency sub-band components of the ground reaction force,” BIOMEDICAL SIGNAL PROCESSING AND CONTROL, vol. 18, pp. 56-60, 2015 [35] Lee S-H, Lim JS. Parkinson’s disease classifcation using gait characteristics and wavelet-based fe
Copyright
Copyright © 2023 Kalaiyarasi. I, Amudha. P, Sivakumari. S. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET51916
Publish Date : 2023-05-10
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

