Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
A Review Article: Nanotechnology
Authors: Prathmesh Belkhode, Karteek Kotekar , Zaid Ameen , Linata Madame , Dr. Ravi Kalsait , Sheetal Dabre
DOI Link: https://doi.org/10.22214/ijraset.2023.55690
Certificate: View Certificate
Abstract
Nanotechnology is the exploitation of the unique properties of material at the nano scale. It is the engineering of functional system at the sub atomic level covers a broad range of topic and is focused on controlling and utilizing Nanotechnology is applicable to a large variety of sector like energy, environmental science, homeland security, medicines. Cardiovascular and cancer has been combat through nano medicines. (Application of nanotechnology in medicine) .
Introduction
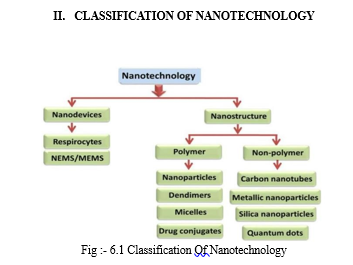
I. INTRODUCTION
Over the past decade, there has been considerable research interest in the field of developing nano technology by using nanoparticles. Norio Taniguchi was the man who first initially utilized the term 'nanotechnology' (Professor of Tokyo Science University) in 1974. He started his research on the free abrasive mechanisms of high exactness machining of hard and brittle materials. Kim Eric Drexler is the founder and father of nanotechnology. He is the man behind theorizing nanotechnology top to bottom and promoting the subject. He is an American engineer best known for promoting the capability of sub-atomic nanotechnology, from the 1970s and 1980s.Nanotechnology deals with dimension and tolerance of less than 100 nanometer, especially the manipulation of individual atom and molecules . [2] A brief and general definition of nanotechnology is the statement by the us nation science and technology is the ability to work at the molecular level, atom to create large structure with fundamentally new molecular organization. The aim is to exploit these properties by gaining control of structures and devices at atomic, molecular, and supramolecular level and to learn to efficiently manufacture and use the devices. Nanotechnology is very diverse ranging from extensions of conventional devices physics to completely new approaches based upon molecular self-assembly, from developing new material with dimension on the nanoscale to direct control of matter on the atomic scale. At this scale the properties approach involving field such as applied physics, material science, chemistry, biology, surface science, robotics engineering, electrical engineering and biomedical engineering. At this scale the properties of matter is dictated and there are few boundaries between scientific disciplines [1]. The use of nanomaterial in everyday products can be generally divided into two types. First, nanomaterial can be merged or added to a pre-existing product and improve the composite objects' overall performance by lending some of its unique properties. Otherwise, nanomaterials such as nanocrystals and nanoparticles can be used directly to create advanced and powerful devices attributed to their distinctive properties. The benefits of nanomaterials could potentially affect the future of nearly all industrial sectors. The beneficial use of nanomaterial can be found in sunscreens, cosmetics, sporting goods, tyres, electronics and several other everyday .[11]


- Multi Lamellar Vesicals (MLV’S)
This type have several lipid bilayer separated from one another by aqueous space. They have difference in size ranging from few hundreds to thousands of nm in diameter [3] .
2. Small Uni Lamellar Vesicles (Suv’s) And Large Uni Lamellar Vesicles (LUV’S)
These consist of single bilayer surrounding the aqueous space. LUV’s have diameter of more than 100 nm SUV’s have diameter of less than 100 nm. Drug is entrapped in the aqueous space as well as intercalated intolipid bilayer of liposomes, depending upon the physiochemical characteristics of drugs.On the basis of material from which liposomes are prepared, they are classified as pH -sensitve liposomes, conventional liposomes, immune liposomes, long circulating liposomes(LCL).
Different methods are used for preparation of liposomes like freeze drying, sonication , micro Empulsification , french pressure cell, lipid film hydration, ethanol injection membranes extrusion, double emulsification method ,ether lyophilization [8]
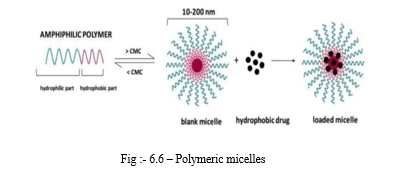
F. Polymeric Micelles
Micelles are amphiphilic surfactant molecule that consist of lipid and amphiphilic molecules.In polymeric miceles,amphiphilic and copolymer orient into nano scopic supra molecular core shell structure known as polymeric micelles. Size of polymeric micelles is less than 100 nm. Their are hydrophilic surface protects there non specific uptake by reticulo endothelial system. In micelles, lipid molecules or polymers orient itself in such a way that hydrophilic and orient towards aqueous phase and lipophilic in towards oily phase.
These micelles are used for systemic delivery of aqueous insoluble drugs. Drug molecule entrapped in hydrophobic core [9] .
III. EXPERIMENTAL
*In order to illustrate the various characteristics of common analytical techniques, two sets of analyses were carried out.
- A series of commercially available nanomaterials were acquired. These were then subjected to a range of tests to investigate their composition.
- A MEMS sample with a monolayer-range thickness antistiction fluorocarbon coating that showed excessive stiction was also analyzed.
The following measurements will be discussed and illustrative examples shown:
- TEM (Transmission Electron Microscopy)
- XPS (X-ray Photoelectron Spectroscopy)
- XRD (X-ray Diffraction)
- AES (Auger Electron Spectroscopy)
- TOF-SIMS (Time of Flight Secondary Ion Mass Spectrometry)
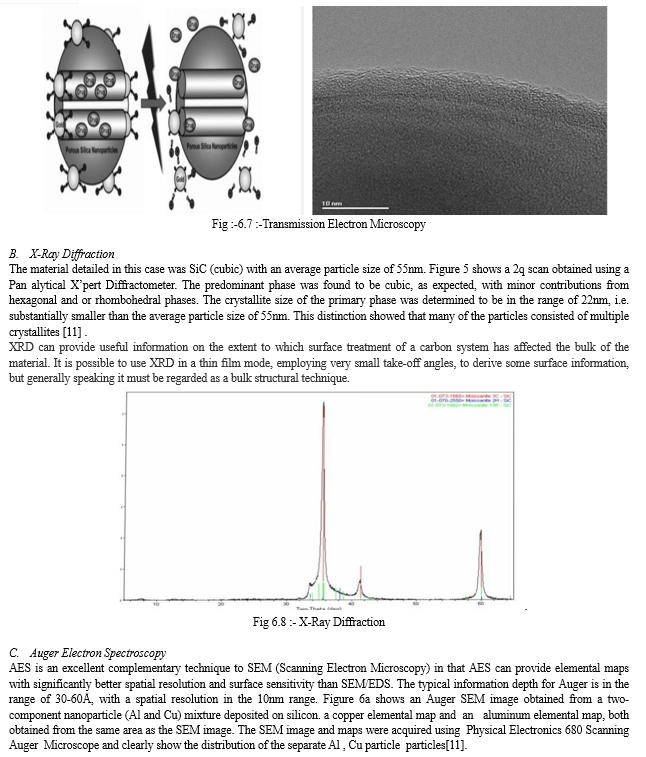
A. Transmission Electron Microscopy
The particles were actually not all spherical and, in fact, a range of shapes were observed. These positively charged species then attached on the negatively charged PSNPs through the electrostatic interaction to produce a PR-Au NP-capped PSNP system, the structure of which was later confirmed by Transmission Elecrton ha Microscopy(TEM) [ JEOL 2010 , 200 kV]
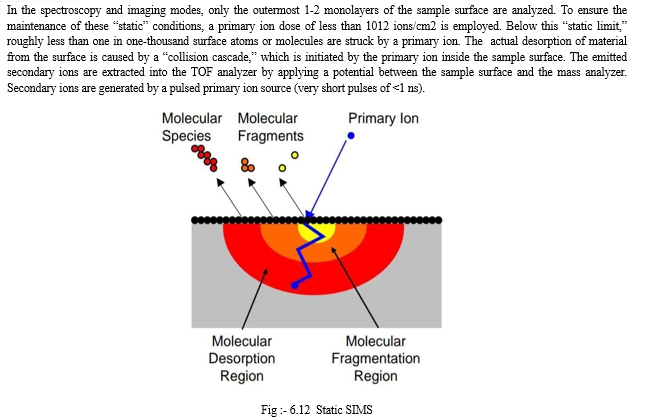
The different methods are :-
- Ampiphillic macromolecule cross linking.
a. Heat cross linking.
b. Chemical cross linking.
2. Polymerization Based Methods.
a. Polymerization of monomers in-situ.
b. Emulsion (micellar) polymerization.
c. Dispersion polymerization.
d. Interfacial condensation polymerization.
e. Interfacial complexation.
3. Polymer Precipitation Methods
a. Solvent extraction or evaporation.
b. Solvent displacement (nanoprecipitation). C) Salting out.
- Amphiphillic Macromolecule Cross Linking
The materials used are Amphiphillic macro molecules, proteins and polysaccharides. These should have affinity to both aqueous and lipid solubility.
It occurs in 2 steps:-
a. Aggregation of amphiphillic.
b. Stabilization by heat denaturation or chemical crosslinking.
The aggregation takes place in o/w or w/o emulsion type. These sub-divide the amphiphiles prior to aggregative stabilization. The aggregation may also takes place in-aqueous amphiphillic solution through removal, extraction, diffusion of solvent. The amphiphiles are aggregated as tiny particles and subsequently rigidised via chemical cross linking[13].
2. Polymerization Based Methods
a. Polymerization of Monomers in-situ
The polymers used are polymethacrylate, polyacrylamide, polybutyl cyanoacrylate, N-N’ methylene- bis-acrylamide etc.
The two different approaches generally adopted for the precipitation of nano spheres using in-situ technique are:-
- The monomer to be polymerized is emulsified in a non- solvent phase (emulsion polymerization).
- The monomer is dissolved in a solvent that is non-solvent.
In emulsion polymerization, the monomer is dissolved in internal phase. In dispersion polymerization, it is taken in the dispersed phase. In both the cases the polymer is insoluble, thus results in a ordered suspension of nanospheres .
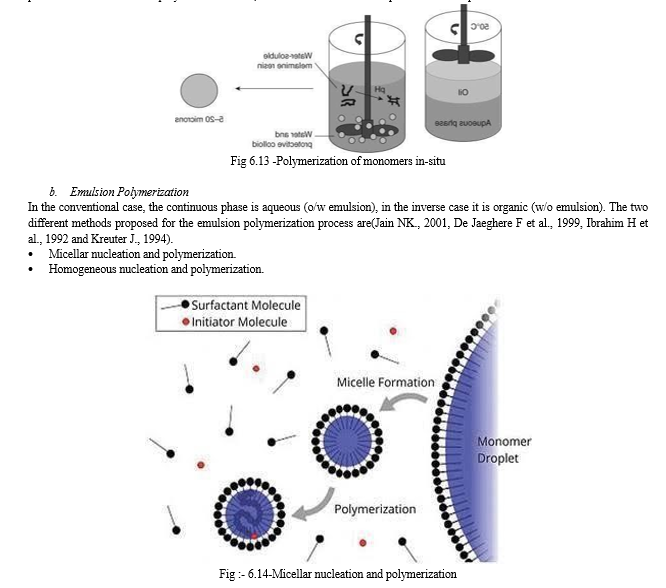
- Micellar Nucleation and Polymerization
The monomer is emulsified in the non-solvent phase with the help of surfactant molecules. This leads to the formation of monomers – swollen micelles and stabilized monomer droplets. Swollen micelles exhibit size in Nano metric range and this have more surface area than monomer droplets. The polymerization occurs in the presence of a chemical or physical initiator. The energy provided by the initiator creates free reactive monomers in the continuous phase, which collide with surrounding un-reactive monomers and initiative polymerization chain reaction [15].
- Homogeneous Nucleation and Polymerization
This process applies largely in case of where monomer is sufficiently soluble in the continuous outer phase. The nucleation and polymerization occurs directly in this phase, leading to the formation of primary chains called oligomers. Both micelles and droplets act as monomer reservoirs throughout thepolymer chain length. When the oligomers have reached a certain length, they precipitate and form primary particles, which are stabilized by the surfactant molecules provided by the micelles and the droplets. Depending on bulk conditions and system stability, the end product nanoshperes are formed either by additional monomer input into the primary particles or by fusion of the primary particle [14] .
- Dispersion Polymerization
The term emulsion polymerization is used when the monomer is emulsified in an immiscible (non-solvent) phase by means of surfactants. But in this monomer is dissolved in an aqueous medium, which acts as a precipitant for subsequently formed polymer. In insitu controlled polymerization the drug may be added to monomeric phase or may be added to the formed polymeric nanoparticles dispersion for adsorptive loading.
The polymerization is initiated by adding a catalyst and proceeds with nucleation phase followed by a growth phase (propagation). But in dispersion polymerization, the nucleation is directly induced in the aqueous monomer solution and the presence of stabilizer or surfactants is not absolutely necessary for the formation of stable nano spheres. This is used to prepare biodegradable polyacrylamide and poly methyl- methacrylate (PMMA) nano particles [3] .
- Interfacial Polymerization
In this the pre formed polymer phase is transformed to an embryonic sheath. The polymer that becomes core and drug.molecule to be loaded are dissolved in a volatile solvent. The solution is then poured in to a non solvent for both polymer and core phase. The polymer phase is separated as a co-acervate phase at o/w inter phase. The resultant mixture turns milky due to formation of nano capsules. This is used for encapsulation of proteins, enzymes, anti bodies and cells were employed [14].
- Interfacial Complexation
In this the pre formed polymer phase is transformed to an embryonic sheath. The polymer that becomes core and drug molecule to be loaded are dissolved in a volatile solvent. The solution is then poured in to a non solvent for both polymer and core phase. The polymer phase is separated as a co-acervate phase at o/w inter phase. The resultant mixture turns milky due to formation of nano capsules. This is used for encapsulation of proteins, enzymes, anti bodies and cells were employed [14].
D. Polymer Precipitation Method
In this, the hydrophobic polymer and or a hydrophobic drug is dissolved in a particular organic solvent followed by dispersion in a continuous aqueous phase, the polymer is insoluble. The external phase also contains the stabilizer. Due to the solvent miscibility techniques they are also known as solvent miscibility techniques they are also known as solvent extraction or evaporation method. The polymer precipitation occurs due to solvent extraction or evaporation. Firstly, polymer is dissolved in a suitable good solvent. After mixing in a poor solvent, which is miscible with the good solvent, the good solvent is gradually evaporated.
- Increasing the solubility of organic solvent in the external medium by adding an alcohol.
- By incorporating additional amount of water into the ultraemulsion (extract or diffuse solvent).
- By evaporation of the organic solvent at room temperature or at accelerated temperature or by using vacuum.
- Using an organic solvent that is completely soluble in the continuous aqueous phase (acetone) –nanoparticles [4].
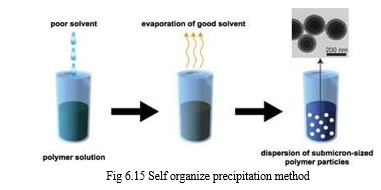
a. Solvent Extraction Method
This method involves the formation of a conventional o/w emulsion between a partially water miscible solvent containing the stabilizer. The subsequent removal of solvent (solvent evaporation method) or the addition of water to the system so as to affect diffusion of the solvent to the external phase (emulsification diffusion method) is two variance of the solvent extraction method. Recently emulsification diffusion method has been used on a regular basis for the solvent extraction purpose [16].
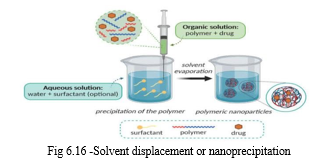
b. Solvent Displacement or Nanoprecipitation
This is based on the interfacial disposition of a polymer following displacement of a semi-polar solvent miscible with water from a lipophilic solution. This method involves the use of an organic phase, which is completely soluble in the external aqueous phase, inducing immediate polymer precipitation because of the complete miscibility of both the phases. Separation and extraction of the solvent is not required for polymer precipitation. After nanoparticles preparation, the solvent is eliminated and the free-flowing nanoparticles can be obtained under reduced pressure. and uniform in size. However, the loading efficiency of lipophilic drugs, such as indomethacin, metripranol, betaxolol in nanoparticles of PLA, PLGA and PECL has been increased using a modified solvent displacement method [17] .
c. Salting Out
The presence of multiple polar functionalities (e.g., amides, alcohols, amines, etc.) generally increases the water solubility of organic compounds, often leading to difficulties in aqueous workups.
To address these situations, a number of techniques that fall under the umbrella of “enhanced extraction” have been developed
- Optimization of the extraction solvent and cosolvent (e.g., 1BuOH or other water-immiscible alcohols) can improve partitioning but usually only to a modest extent, if at all.
- Continuous liquid–liquid extraction overcomes poor partitioning by continual renewal of fresh solvent. Although continuous processing can offer many advantages, it requires significant capital investment and limits the portability of the process [18 ].
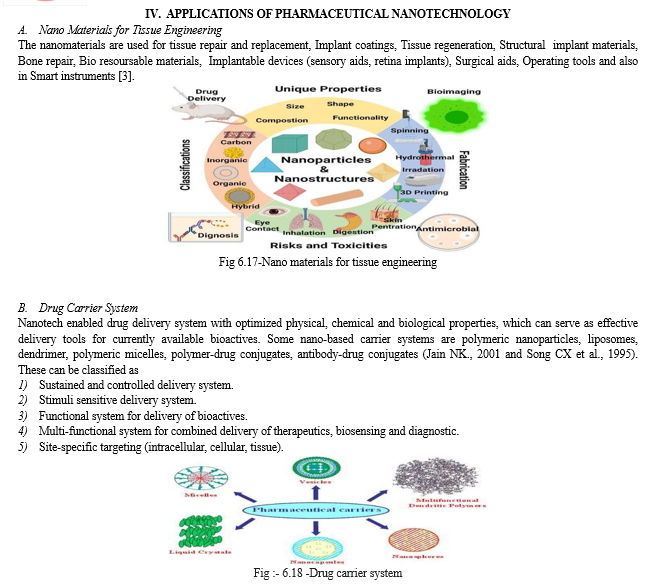
The main advantage deriving from the use of drug carriers is their ability to protect drugs during the overall administration time, enclosing them in external protective barriers of different types and nature; thus, reducing losses of active substances and limiting any side effects in patients Drug delivery systems are generally designed at nanometric and micrometric levels, to combine different properties with specific aims, such as site specificity, longevity, or external stimuli sensitivity[19] .
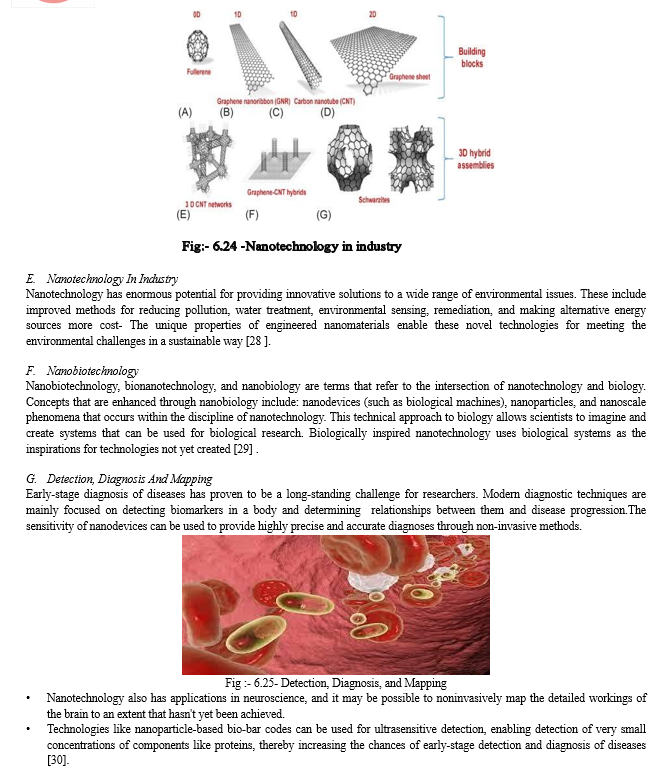
C. Molecular Diagnostics:- (Molecular Imaging)
It is representing, characterizing and quantifying sub cellular biological processes include gene expression, protein-protein interaction, signal transduction, cellular metabolism. They are used in magnetic resonance imaging, optical imaging, ultrasonic imaging and nuclear imaging. Other applications are specific labeling of cells and tissues, useful for long-term imaging, multicolor multiplexing, dynamic imaging of sub cellular structures and fluorescence resonance energy transfer (FRET) and magnetic resonance imaging (MRI).
MRI agents are replaced by nanomaterials like dendrimer, quantum dots, carbon nanotubes and magnetic nanoparticles. They are very efficient, stable, intense, M clearer image due to high intensity, photostability, resolution, resistance (Gupta P.k et al., 1987).Quantum dots, iron oxide nanocrystal and metallic nanoparticles [20] .




Conclusion
As a conclusion to this topic I would like to say that Nanotechnology is a brand new technology that has just began, it is a revolutionary science that will change all what we knew before. The future that we were watching just in science fiction movies will in the near future be real. This new technology will first of all, keep us healthy because of Nano robots that will repair every damage that we have in our body. Nanotechnology will give us an abundant energy because it will transform energy more effectively. Nanotechnology covers a lot of domains today and will cover a lot more in the near future, it is infinitely big and will make a lot of inventions come true like teleportation for example which scientists are working on today.
References
[1] https://www.researchgate.net/publication/289871657_Nanotechno logy_A_Review [2] https://www.sciencedirect.com/science/article/pii/B978012818154 600010X [3] Journal of Applied Pharmaceutical Science 01 (02); 2011: 08-16 [4] https://www.peertechzpublications.com/articles/IJNNN-7141.php [5] Sinha N, Yeow J.T.W. Carbon Nanotubes for Biomedical Applications. IEEE transactions on nanobioscience. 2005 4(2) 180195. [6] Bailey, R. E., Smith, A.M., Nie S. Quantum dots in biology and medicine. Physica E (2004), 25, 1–12 [7] Kreuter J. (1983) International Journal of Pharmaceutics, 14, 43. [8] Maincent P., Marchal-Heussler l., Sirbat D., Thouvenotp., Hoffman M. and Vallet J.A.(1992), Proceedings of International Symposia, Control Release Bioactive Materials, 18, 226. [9] Jain S., Jain N.K. Liposomes as drug carrier, In: Jain NK, editor. Controlled and novel drug delivery, 2nd edition, CBS publisher, New Delhi, 2002, 304-52. [10] Ferrari, M. Cancer nanotechnology: opportunities and challenges. Nature Reviews/Cancer. (2005) 5, 161-171 [11] Nanotech 2007 , www. nsti .org , ISBN 1420063766 vol. 4, 2007 [12] Brindle , Evans and Wilson , “ Encyclopedia of material characterization” Butterworth-Heinmann, 1992. [13] Settle , “Handbook of instrumental techniques for analytical chemistry, prentice-Hall , 1997. [14] Gupta P.k, Gallo J. M, Hung C.T and Perrier D.G. (1987a) Drug Development and Industrial Pharmacy. 13, 1471 [15] Kreuter J. (1991) Journal of controlled Release, 16, 169. [16] De Jaeghere F., Doelker E., and Gurny R. (1999) In: Encyclopedia of controlled drug delivery, Mathiowitz, E. (Ed.), vol II, Jhon Wiley and sons, New york, 641. [17] Nahar M, Dutta T, Murugesan S, Asthana A, Mishra D, Rajkumar V, Tare M, Saraf S, Jain NK. Functional polymeric nanoparticles: an efficient and promising tool for active delivery of bioactives. Critical Reviews in Therapeutic Drug Carrier Systems. (2006) 23(4), 259318. [18] Quintanar-Guerrero D, Fessi H, Alleman E. and Doelker E. (1996) International Journal of Pharmaceutics, 143, 133. [19] Allemann E.,Gurny R. and Doelker E. (1992) International Journal of Pharmaceutics, 87, 247 [20] Song C.X., Labhaasetwar V., Gujman L., Topol E. and Levy R.J. (1995), Proceedings of International Symposia, Control Release Bioactive Materials, 22, 444. [21] Gupta P.k, Gallo J. M, Hung C.T and Perrier D.G. (1987a) Drug Development and Industrial Pharmacy. 13, 1471. [22] Khopde AJ, Jain, NK. Dendrimer as potential delivery system for bioactive In: Jain NK, editor. Advances in controlled and novel drug delivery. CBS publisher, New Delhi, 2001, 361-80. [23] https://link.springer.com/book/10.1007/978-3-030-11864-8 [24] https://www.academia.edu/22325030/APPLICATIONS_OF_NA NOTECHNOLOGY_IN_CONSTRUCTION_INDUSTRY [25] https://link.springer.com/article/10.1007/s11356-016-6457-z [26] https://www.frontiersin.org/articles/10.3389/fmicb.2017.01501/f ull [27] https://www.mdpi.com/2310-2861/8/3/173 [28] https://www.nanowerk.com/spotlight/spotid=16203.php [29] Industrial applications of nanotechnology – Wikipedia [30] https://www.nano.gov/about-nanotechnology/applicationsnanotechnology [31] https://www.cancer.gov [32] https://www.frontiersin.org/articles/10.3389/fbioe.2021.758121/f ull#:~:text=Nanomaterials%20for%20the%20Therapy%20of%20COVID%2D19,-Nanomaterials%20provided%20a&text=Based%20on%20the%20in teraction%20between,used%20to%20treat%20COVID%2D19. [33] https://www.degruyter.com/journal/key/ntrev/html [34] Fessi H., Puisiex F.,Devissaguet J.P., Ammoury N. and Benita S. (1989). International Journal of Pharmaceutics. 55, R1. [35] Gupta P.k, Hung C.T, Lam F.C. and Perrier D.G. (1987b) International Journal of Pharmaceutics, 43, 167. [36] Gupta P.k, Gallo J. M, Hung C.T and Perrier D.G. (1987a) Drug Development and Industrial Pharmacy. 13, 1471. [37] Ferrari, M. Cancer nanotechnology: opportunities and challenges. Nature Reviews/Cancer. (2005) 5, 161-171. [38] http://crnano.typepad.com/crnblog/2006.03/new_risks_new_f.ht ml [39] http://www.gkss.de/templates/images_d/werkstoff/micelle.jpg. 39)https://www.rroij.com/open-access/a-review-on-application-ofnanotechnology-in-pharmaceuticals.php?aid=90378 [40] https://www.japsonline.com/admin/php/uploads/159_pdf.pdf
Copyright
Copyright © 2023 Prathmesh Belkhode, Karteek Kotekar , Zaid Ameen , Linata Madame , Dr. Ravi Kalsait , Sheetal Dabre. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET55690
Publish Date : 2023-09-10
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

