Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Solvent Free Synthesis, Characterization and Application of Zinc Oxide Nano Particles in Tracer Diagnostic Techniques
Authors: Preeti Nitin Joshi
DOI Link: https://doi.org/10.22214/ijraset.2022.46804
Certificate: View Certificate
Abstract
In this work, we report the synthesis of ZnO nanoparticles using Moringa Oleifera (Drumstick) leaves as natural precursor via precipitation method. The formation and characterization of ZnO nanoparticles was established by UV-VIS spectroscopy, Fourier transform infrared spectroscopy and Particle Analyzer. The absorbance at 361nm proves the formation of ZnO. The IR spectra results proved the capping and stabilizing agents. The size of the particle was found to be 246.7 nm. Further, the ZnO was used as a nutrient in improvement of plant growth. Background: Analgesia, one of the components of triad of anaesthesia, has now extended to relief of postoperative pain, chronic pain and cancer pain. The spinal cord has taken the center stage in analgesia practice and Spinal anaesthesia is the commonly used technique for lower limb surgeries as it is easy to administer, economical and causes less hemodynamic variation than general anaesthesia. Hence different additives can be used to increase the duration of postoperative analgesia. Since there are no studies comparing Buprenorphine and Nalbuphine, we have selected this study to evaluate the effect of intrathecal Bupivacaine with Buprenorphine compared with Nalbuphine for postoperative analgesia. (10) Materials and Methods: Collection of Sample: Fresh Moringa Oleifera leaves were collected from Farm house. Leaves were washed thoroughly by water and were allowed to dry in air at Sun light. Chemicals, Solvents and Starting Materials: Zinc acetate dihydrate, ammonium carbonate, sodium hydroxide pellets and other chemicals were purchased from LOBA Chemie, Aatarinstra chem, and Chemco (India). All chemicals were used without further purification. Preparation of Moringa Oleifera Leaf Extract: 5 grams of Moringa Oleifera leaves were washed thoroughly with distilled water. These leaves were heated for 60 min in 200 ml of distilled water at 50O C. Then the extract was filtered with Whatman 41 filter paper. The filtrate was stored in a cool and dry place. Results The present study reports the use of dried leaf for the synthesis of nanoparticles, which are free from external stabilizing and accelerating agents, and does not require continuous agitation. The appearance of yellow precipitate is a clear indication of the formation of ZnONPs formed in the reaction mixture.
Introduction
I. INTRODUCTION
In modern research era of any branch of science, nanotechnology has found to be of enormous interest. Nanoparticles (NPs) play an essential role as building blocks of nanotechnology. Nowadays, nanoscience as well as nanotechnology is widely applied in different fields mainly in sensor, electronic, antibacterial, water purification, cosmetic, biomedical, pharmaceutical, environmental, catalytic and material applications. The size, crystallinity and morphology of the nanomaterial can greatly influence their catalytic, magnetic, electronic and optical properties [1]. The main advantages of nanoparticles synthesis at room temperature and from plant extracts are partly to fulfill the green synthesis [2]. The metal and metal oxide nanoparticles are synthesized by physical, biological, chemical and very recent green approaches. Recently, green synthesis has become popular way to synthesize NPs due to its low cost, environment compatibility, synthesizable in ambient atmosphere and non-toxicity [3]. The noticeable importance of Zinc oxide and ZnONPs is they are very good semiconductors with wide range of band gap (3.37) having room temperature dependent high excitation binding energy (60 meV) [4]. Zinc oxide and ZnONPs have excellent thermal and chemical stability with exceptional optical behavior [5]. Now a day the considerable interest was seen in biosynthesis of different nanoparticles as well as photo degradation properties. Recently different metal and metal oxide nanoparticles were reported for their efficient photo degradation effect in presence of visible light illumination [6].
Since, different plant extracts are the main source of large scale synthesis of NPs via green way which includes the synthesis of several noble metal nanoparticles like Au, Ag and Pd. For this extracts from different plant organs e.g. geranium leaves, lemongrass, neem leaves, aloe-vera and others have been reported [7, 8, 9, 10, 11]. Moreover the important applications of ZnONPs towards environmental and biological fields are like drug delivery, biological sensing, biological labeling, gene delivery and nano medicine are reported [12, 13, 14, 15]. ZnONPs also have applications for antibacterial, anti-diabetic, antifungal, acaricidal, pre-diculicidal, larvicidal activities [16, 17, 18, 19, 20]. Different medicinal plant and their leaf and calyx extract are found to have important properties such as diuretic, antioxidant, blood pressure suppressive, chemo protective, hypotensive, anti-tumor and anti-cancerous effect [21, 22, 23, 24, 25]. Karunakaran et al. [26] showed the antimicrobial properties of ZnONPs towards both gram positive and gram negative bacteria. Mahdiyeh et al. [27] synthesized ZnO nanowires via hydro-thermal process at 90 °C from zinc acetate dehydrate. Moj et al. [28] have bio-synthesized silver nanoparticles using Mine Soil Bacteria as natural precursor that showed antibacterial activity.
In our present work we have synthesized ZnONPs by using Moringa Oleifera (Drumstick) leaves as natural precursor. The prepared nanoparticles were characterized by different techniques and further to be utilized for various applications.
II. MATERIAL AND METHODS
- Synthesis of Zinc Oxide Nanoparticles: For the synthesis of zinc oxide nanoparticles by precipitation reaction process, 10 ml of Moringa Oleifera leaf extract was mixed with 5 ml 5% NaOH solution[29]. Then solutions were taken in a 250 ml beaker and kept stirring for 1 h. Then zinc acetate (2.1g in100ml water), ammonium carbonate (0.96g in 100ml) solutions were added drop wise into the beaker simultaneously with constant stirring[30]. After the completion of reaction, the suspension was kept stirring for 1h at room temperature. Finally, precipitate was filtered, washed with distilled water for several times[31]. Then the precipitates were dried under hot air oven. Then zinc oxide nanoparticles were collected and stored in vacuum for further use.
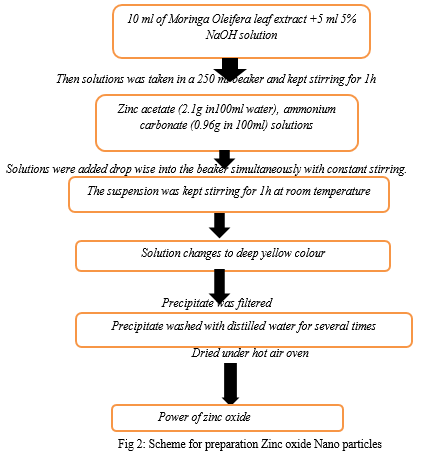
III. RESULT
The present study reports the use of dried leaf for the synthesis of nanoparticles, which are free from external stabilizing and accelerating agents, and does not require continuous agitation. The appearance of yellow precipitate is a clear indication of the formation of ZnONPs formed in the reaction mixture.
IV. DISCUSSION
A. UV-VIS Spectra Analysis
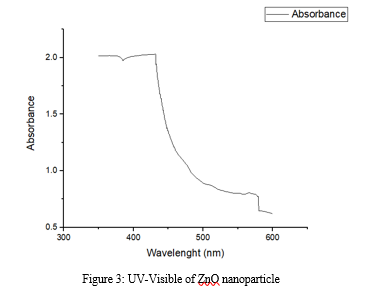
This UV graph shows the absorption spectrum of the synthesized ZnONPs with the absorption peak around 360 nm. It indicates that ZnONPs displays excitation absorption (at 360 nm) due to their large excitation binding energy at room temperature. The sharp bands of zinc colloids were observed at 360 nm, which proves that the zinc ion is efficiently reduced by the Moringa Oleifera leaf extract. The wavelength of 360 nm absorption peak confirms the occurrence of blue-shifted absorption spectrum with respect to the bulk value (365 nm) of the ZnONPs, due to the quantum confinement effect, which is in good agreement with the previous report.[33]
B. FTIR Analysis
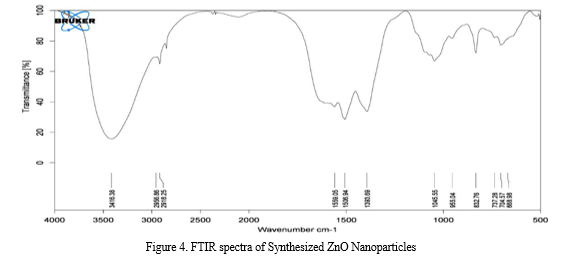
FTIR measurement (Figure 4) was carried out to identify the possible biomolecules in Moringa Oleifera leaf extract responsible for capping leading to efficient stabilization of the ZnONPs. The FTIR spectra shows the presence of bonds due to −OH stretching frequency around 3416.38 cm-1. However, peaks at 2956.86 cm−1 and 2918.25 cm−1 are attributed to the asymmetric and symmetric stretching vibrations of −CH2 group respectively. The peak at 1045.55 cm-1 results from the stretching bands of C=O functional groups. The peak at around 1393.69 cm-1 present in ZnO signified amide band of the random coil of protein [34]. The peak at 688.98 cm-1 indicates the stretching vibrations of ZnO nanoparticle, which is consistent with that, reported earlier [35]. The region between 400 and 600 cm-1 is assigned for metal-oxygen bond. In addition to the absorption bands of the biomolecules used as reduction and stabilization (capping agents), the absorption peak at 440cm-1 indicate the presence of ZnONPs [36].
C. Particle size Analysis
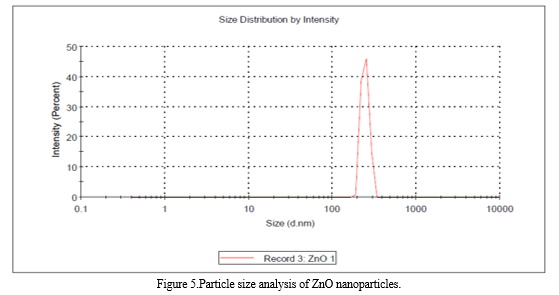
The particle size distribution (PSD) curve of the synthesized Zinc oxide particles from different zinc precursors is given in figure 3. The PSD plot for sample ZnO shows that all the particles are within the size range of around 246.7 nm.

Conclusion
A. Effect on Plant Growth The zinc oxide nanoparticles obtained by green synthesis method was applied to evaluate the growth of plants. Initially soil was taken and mixed with cow dung and further Zinc oxide was added to the soil to check the improvement in growth of plants. The growth of the plants was evaluated at regular intervals B. Weekly Growth Analysis of Sorghum Plant Soil Soil + dung Soil +dung + ZnoNPs Week 1 14.5 (cm) 14.6 15.2 Week 2 30.5 30.9 32.1
References
[1] Cui, Y., Lieber, C. M. (2001). “Functional nanoscale electronic devices assembled using nanowire blocks”, Science., 29: 851-853. [2] Jones, M. R. (2011). “Templated Techniques for the Synthesis and Assembly of Plasmonic Nanostructures”, Chemical Reviews., 111: 3736-3827. [3] Bala, N., Saha, S., Chakraborty, M., Maiti, M., Das, S., Basub, R., Nandy, P. (2015). “Green synthesis of zinc oxide nanoparticles using Hibiscus subdariffa leaf extract: effect of temperature on synthesis, anti-bacterial activity and anti-diabetic activity”, RSC Adv., 5: 4993–5003. [4] Roduner, E. (2006). “Size matters: why naomaterials are different”, Chemical Society Reviews., 35: 583-592. [5] Iravani, S. (2013). “Green synthesis of metal nanoparticles using plants”, Green Chemistry., 13: 2638-2650. [6] Mohamed, R. M., Mkhalid, I. A., Baeissa, E. S., Al-Rayyani, M. A. (2012). “Photocatalytic Degradation of Methylene Blue by Fe/ZnO/SiO2 Nanoparticles under Visiblelight”, Journal of Nanotechnology., 2012-2016. [7] Shankar, S. S., Ahmad, A., Sastry, M. (2003). “Geranium leaf assisted biosynthesis of silver nanoparticles”, Biotechnol. Prog., 19: 1627. [8] Shankar, S. S., Rai, A., Ahmad, A., Sastry, M. (2004). “Rapid synthesis of Au, Ag, and bimetallic Au core-Ag shell nanoparticles using Neem (Azadirachta indica) leaf broth”, J. Colloid Interf. Sci., 75: 496. [9] Shankar, S. S., Rai, A., Ahmad, A., Sastry, M. (2005). “Controlling the Optical Properties of Lemongrass Extract Synthesized Gold Nanotriangles and Potential Application in Infrared-Absorbing Optical Coatings”, Chem. Mater., 17: 566. [10] Chandran, S. P., Chaudhary, M., Pasricha, R., Ahmad, A., Sastry, M. (2006). “Synthesis of gold nanotriangles and silver nanoparticles using Aloe vera plant extract”, Biotechnol. Prog., 22: 577. [11] Huang, J., Li, Q., Sun, D., Lu, Y., Su, Y., Yang, X., Wang, H., Wang, Y., Shao, W., He, N., Hong, J., Chen, C. (2007). “Green Synthesis and Characterization of Gold Nanoparticles Using Onion (Allium cepa) Extract”, Nanotechnology., 18: 105. [12] Rasmussen, W. J., Martinez, E., Louka, P., Wingett, G. D. (2010). “Zinc oxide nanoparticles for selective destruction of tumor cells and potential for drug delivery applications”, Expert Opin. Drug Delivery., 7: 1063–1077. [13] Yoon, H. S., Kim, J. D. (2006). “Fabrication and Characterization of ZnO Films for Biological Sensor Application of FPW Device”. Applications of ferroelectrics., 15th IEEE international symposium., 3: 322–325. [14] Xiong, M. H. (2013). “ZnO Nanoparticles Applied to Bioimaging and Drug Delivery”, Adv. Mater., 25: 5329–5335. [15] Nie, L., Gao, L., Feng, P., Zhang, J. (2006). “Three-Dimensional Functionalized Tetrapod like ZnO Nanostructures for Plasmid DNA Delivery”, Small., 2: 621–625. [16] Applerot, G., Lipovsky, A., Dror, R., Perkas, N. (2009). “Enhanced antibacterial activity of nanocrystalline ZnO due to increased ROS-mediated cell injury”, Adv. Funct. Mater., 19: 842–852. [17] Sharmaa, D., Rajputa, J., Kaitha, S. B., Kaurb, M. (2010). “Synthesis of ZnO nanoparticles and study of their antibacterial and antifungal properties”, Thin Solid Films., 519: 1224–1229.118 Pal, Mondal, Maity and Mukherjee [18] Nair, S. (2009). “Role of size scale of ZnO nanoparticles and microparticles on toxicity toward bacteria and osteoblast cancer cells”, J. Mater. Sci.: Mater. Med., 20: 235–241. [19] Kirthi, V. A., Rahuman, A. A., Rajakumar, G., Marimuthu, S. (2011). “Acaricidal, pediculocidal and larvicidal activity of synthesized ZnO nanoparticles using wet chemical route against blood feeding parasites”, Parasitol. Res., 109: 461–472. [20] Alkaladi, A., Abdelazim, M. A., Afifi, M. (2014). “Antidiabetic Activity of Zinc Oxide and Silver Nanoparticles on Streptozotocin-Induced Diabetic Rats”, Int. J. Mol. Sci., 15: 2015–2023. [21] Jiménez-Ferrer, E., Alarcón-Alonso, J., Aguilar-Rojas, A. (2012). “Zamilpa A Diuretic effect of compounds from Hibiscus sabdariffa by modulation of the aldosterone activity”, Planta. Med., 78: 1893-98. [22] Sharaf, A. (1962). “The pharmacological characteristics of Hibiscus sabdariffa L”. Planta. Med., 10: 48–52. [23] Salleh, N., Runnie, I., Roach, D., Mohamed, S. (2002). “Inhibition of low-density lipoprotein oxidation and upregulation of low-density lipoprotein receptor in HepG2 cells by tropical plant extracts”, J. Agric. Food. Chem., 50: 3693-3697. [24] Lin, H. H., Chen, H. J., Wang, J. C. (2011). “Chemopreventive properties and molecular mechanisms of the bioactive compounds in Hibiscus sabdariffa Linne. Curr”, Med. Chem., 18: 1245-54. [25] Ajiboye, O. T., Salawu, A. N., Yakubu, Y. M., Oladiji, T. (2011). “A Antioxidant and drug detoxification potentials of Hibiscus sabdariffa anthocyanin extract. Drug”, Chem. Toxicol., 34: 109-115. [26] Karunakaran, C., Gomathisankar, P., Manikandan, G. (2010). “Structural and Optical Properties of Cu2+ + Ce3+ Co-Doped ZnO by Solution Combustion Method”, Material Chemistry and Physics., 123: 585-94. [27] Mehran, M., Sheikhshoaei, F., Sheikhshoaie, I. (2017). “Effect of Nano-Textured Silicon Substrate on the Synthesize of Metal Oxides Nanostructures”, Int. J. Nanosci. Nanotechnol., Vol.,13(3): 265-274. [28] Khaleghi, M., Madani, M., Parsia, P. (2017). “Biosynthesis Characteristic of Silver Nanoparticles Produced by Mine Soil Bacteria Isolation, Kerman, Iran”, Int. J. Nanosci. Nanotechnol., Vol.,13: 307-313. [29] Bauer, W. A., Kirby, M. M. W., Sherries, C. J., Truck, M. Am. (1996). “Antibiotic susceptibility testing by a standardized single disk method”, J. Clin. Pathol., 45: 493-6. [30] Elumalai, K., Velmurugan, S., Ravi, S., Kathiravan, V., Ashokkumar, S. (2015), “Green synthesis of zinc oxide nanoparticles using Moringa oleifera leaf extract and evaluation of its antimicrobial activity”, Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy., 143: 158–164. [31] Cai, S., Sing, R. B. (2004). “A distinct utility of the amide III infrared band for secondary structure estimation of aqueous protein solutions using partial least squares methods”, Biochemistry., 43: 2541–2549. [32] Ravichandrika, K. (2012). “Synthesis, Characterization and Antibacterial Activity OfZnO Nanoparticles”, International Journal Of Pharmacy And Pharmaceutical Sciences., 4: 336- 338, [33] Varghese, E., George, M., (2015). “Green synthesis of zinc oxide nanoparticles”, International Journal of Advance Research In Science And Engineering., 4: 01. [34] Joel, C., Sheik, M., Badhusha, M., (2016). “Green synthesis of ZnO Nanoparticles using Phyllanthus embilica Stem extract and their Antibacterial activity”, Der Pharmacia Lettre., 8: 218-223. [35] Navaza, J., Silva, A. M. (1979). “A geometrical-approach to solving crystal structures”, Acta Crystallographica section A., 35: 266-275. [36] Wei, W., Jiang, X., Lu, L., Yang, X., Wang, X. (2009). “Study on the catalytic effect of NiO nanoparticles on the thermal decomposition of TEGDN/NC propellant”, J. Hazard Mater., 168: 838-842. [37] Comparelli, R., Fanizza, E., Curri, L. M., Cozzoli, D. P., Mascolo, G., Agostiano, A. (2005). “UV induced photocatalytic degradation of azo dyes by organic-capped ZnO nanocrystals immobilized onto substrates”, Appl. Catal. B: Environ., 60: 1–11. [38] 41.Hayat, K., Gondal, A. M., Khaled, M. M., Ahmed, S. (2010). “Kinetic study of laser-induced photocatalytic degradation of dye (alizarin yellow) from wastewater using nanostructured ZnO”, J. Environ. Sci. Health, Part A., 45: 1413–1420. [39] Mai, D. F., Chen, C. C., Chen, L. J., Liu, C. S. (2008). “Photodegradation of methyl green using visible irradiation in ZnO suspensions determination of the reaction pathway and identification,of intermediates by a high-performance liquid chromatography–photodiode array-electrospray ionizationmass spectrometry method”, J. Chromatogr. A., 1189: 355–365. [40] Zhai, J., Tao, X., Pu, Y., Zeng, F. X., Chen, F. X. (2010). “Core/shell structured ZnO/SiO2 nanoparticles: Preparation, characterization and photocatalytic property”, Appl. Surf. Sci., 257: 393–397. [41] Akyol, A., Bayramoglu, M. (2005). “Photocatalytic degradation of Remazol Red F3B using ZnO catalyst”, J. Hazard. Mater., 124: 241–246. International Journal of Nanoscience and Nanotechnology 119 [42] Chakrabarti, S., Dutta, K. B. (2004). “Photocatalytic degradation of model textile dyes in wastewater using ZnO as semiconductor catalyst”, J. Hazard. Mater., 112: 269–278. [43] Behnajady, A. M., Modirshahla, N., Hamzavi, R. (2006). “Kinetic study on photocatalytic degradation of CI Acid Yellow 23 by ZnO photocatalyst”, J. Hazard. Mater., 133: 226–232. [44] Chen, C. C., Liu, F. J., Liu, P., Yu, H. B. (2011). “Investigation of photocatalytic degradation of methyl orange by using nano-sized ZnO catalysts”, Adv. Chem. Eng. Sci., 1: 9–14. [45] Annadurai, G., Sivakumar, T., Rajesh, Babu. S. (2000). “Photocatalytic decolorization of congo red over ZnO powder using Box-Behnken design of experiments”, Bioprocess Biosyst. Eng., 23: 167–173. [46] Amisha, S., Selvam, K., Sobana, N., Swaminathan, M. (2008). “Photomineralisation of Reactive Black 5 with ZnO using solar and UV-A light”, J. Korean Chem. Soc., 52: 66–72. [47] Lu, S. C., Wu, T. Y., Mai, F., Chung, S. W., Wu, W. C., Lin, Y. W., Chen, C. C. (2009). “Degradation efficiencies and mechanisms of the ZnO-mediated photocatalytic degradation of Basic Blue 11 under visible light irradiation”, J. Mol. Catal. A: Chem., 310: 159–165.
Copyright
Copyright © 2022 Preeti Nitin Joshi. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET46804
Publish Date : 2022-09-17
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

