Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
A Web Application for Drug Traceability in Healthcare System using Blockchain
Authors: Varshad V, Vinay Prasad M S
DOI Link: https://doi.org/10.22214/ijraset.2022.46221
Certificate: View Certificate
Abstract
In the current situation, we come across various problems in the pharmaceutical supply chain in India which can be solved with different ideas. Since a drug’s ownership changes from manufacturer to distributor and then to the pharmacist before reaching the customer. The manufacturers don’t know how their drug is being used. At the same time, consumers don’t know whether the drug has come from the rightful source including inaccurate information, lack of transparency and limited data provenance. It is difficult to detect counterfeits because these drugs pass through different complex distributed networks, thus forming opportunities for counterfeits to enter the authentic supply chain. Most existing track and trace systems are centralized leading to data privacy, transparency and authenticity issues in healthcare supply chains. In this project work, an end-to-end drug tracking system across the pharmaceutical supply chain is built as a Web Application. It attempts to prevent drug counterfeiting, and add traceability, security, and visibility to the pharmaceutical supply chain using Blockchain technology and AWS S3 service. The system guarantees data provenance, eliminates the need for intermediaries, and provides a secure immutable history of transactions to all stakeholders. We developed a Web application that provides traceability of drug manufacturers and other information to emulate the data tampering scenario. We also create a tampering application to exhibit the false transactions of a node. We perform testing and validation to recover the original data to evaluate its effectiveness to enhance traceability within the pharmaceutical supply chain.
Introduction
I. INTRODUCTION
Counterfeit drugs are a major worldwide problem which not only has serious adverse impact on human health but also causes severe economic loss to the healthcare industry. There are many problems associated with the healthcare data handling, particularly with the access control, sharing and storage of data. The main issues with drug safety in the counterfeit medicine supply chain, are to do with how the drugs are initially manufactured. The traceability of right and active pharmaceutical ingredients during actual manufacture is a laborious process. Detecting drugs that do not contain the intended active ingredients can ultimately lead to end consumer patient harm or even death. Healthcare supply chain is a convoluted network of various independent constituents that include raw material suppliers, manufacturer, distributor, pharmacies, hospitals and patients. Tracking supplies through this network is paramount due to various factors containing lack of information, centralized control and competing behavior between stakeholders. According to the Health Research Funding Organization, up to 30% of the drugs sold in developing countries are counterfeit. The counterfeiting of medicines causes a severe menace to the society. Further, a recent study by World Health Organization (WHO) defined counterfeit drugs as one of the important reasons of deaths in developing countries and children are the victims in most cases. Counterfeiting of various products creates muddle to the manufacturing industries and it causes severe menaces to pharmaceuticals products. This menaces the public health and also causes revenue loss to the recognized manufacturing organizations. Most existing track and trace systems are centralized leading to data privacy, transparency and authenticity issues in healthcare supply chains, which are prone to various attack such as replay, man-in-the-middle attack.
To eliminate counterfeits and to ensure product safety an end- to-end drug tracking system across the pharmaceutical supply chain is pre-dominant. Blockchain technology stands out when it comes to preventing counterfeit drugs in the drug supply chain. It corroborates a permanent chain of transaction ledger, which helps to track each step of the supply chain at the individual drug level.
II. LITERATURE SURVEY
Blockchain was initially invented for achieving secured digital money transactions but the technology is garnering ultimate buildup in the healthcare sector. Healthcare industry consists of expeditiously growing delicate data which need to be conserved from confidentiality menaces and integrity menaces. An Ethereum blockchain-based approach leveraging smart contracts and decentralized off-chain storage for coherent drug traceability in healthcare supply chain is presented.
It overcomes the existing track and trace systems which are centralized leading to data privacy, transparency and authenticity issues in healthcare supply chain in [1]. A blockchain based reliable framework for healthcare chain supply among stakeholders is presented [2]. The storage of transaction data constitutes the alikeness with Bitcoin transaction data. Based on digital signature and PKI the system can prevent replay and man-in-middle attack. The shared data between the stake holders is in encrypted QR code format. The author has failed to implement a complete decentralization system which also lacks transparency of the supply chain. Similarly, a blockchain-based solution for COVID-19 medical equipment achieved by automating forward supply chain processes is presented in [3]. To securely store, fetch and share the COVID-19 related information they have used interplanetary file systems (IPFS) with the integration of Ethereum blockchain. The author has failed to incorporate vaccination data and its supply chain into the system proposed. A pharmaceutical supply chain where only reliable parties can join the network and store or retrieve the information is developed in [4]. Blockchain technology is incorporated to the drug supply system to add security, visibility and traceability. The system developed in [6] Hyperledger Fabric and Hyperledger composer for development purposes. The supply chain exhibits the data regarding the recognition of the medicines produced in the Manufacturing plant. It also exhibits the data of motion patterns of those medicines in the supply chain to corroborate the verification and legitimacy of the medicines which are transported to the pharmacies. However, smart contracts have not been created and access lists are not used and no tests have been carried out by the author. A decentralized and distributed track and trace system has been established in [8], using RFID and barcodes. This system allows tracking from manufacturers to the serialization process to drugs distribution. A data management system for clinical trials is proposed in [9]. By using Ethereum blockchain data related to new cases, recovered cases and deaths collected from external trusted sources are stored. The developed system by the author lacks implementation of DApps, which makes the digital applications to run by relying on a single computer. An Ethereum based monitor and tracking system is implemented on the ropsten network using smart contracts for COVID-19 vaccine supply management in [10]. In this paper [11], present a Blockchain model for medicine supply in a hospital. The model is digital, decentralized, and continuous. Minimal resources have been used to increase the performance and to reduce latency of the system. The authors have used Proof of Ownership in [13], using blockchain to develop a transparent and traceable system. Real-time monitoring of drugs being transported are provided to logistic companies. Only the parties with ownership have the option to hand over their authorities to some other stakeholders of the supply chain.
III. PROPOSED WORK
A. Block diagram
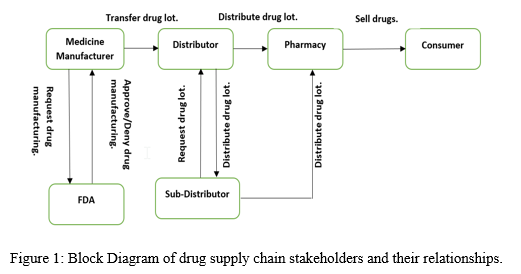
The Drug supply chain stakeholders and their relationships is depicted in the figure 1. The FDA, drug manufacturing companies and pharmacies are registered by system manager. The manufacturer requests FDA for drug patent approval. The FDA can either approve or reject the manufacturer’s request. Once the FDA approves the manufacturer’s request, drug manufacturing is carried out. The manufacture registers distributor and the distributor register sub-distributor. The distributor can place a medicine order request to the manufacturer and once approved he can distribute the drugs to either sub-distributor or pharmacies medicine order requests placed. The pharmacies can place medicine order either to distributor or sub-distributor. The pharmacies can sell drugs to the consumers.
In proposed system, pharmaceutical company will manufacture the medicines with details such as drug name, ingredients, timestamp, usage of drug and get authorized by regulatory authorities of healthcare industry. The Block diagram of the project is as shown in the ?gure 2. In order to do this, the manufacturing company has to get medicine patent approval from FDA for manufacturing medicine. So, the medicine ingredients details are encrypted using SHA, AES Rijndael algorithm and is written to QRCode image and stored in AWS S3 service for security purpose. The Regulatory department now gets the medicine details by extracting data from QRCode image stored in AWS S3 service and decrypts data using SHA, AES Rijndael algorithm. The FDA veri?es the medicine ingredients details and approve or reject medicine patent.
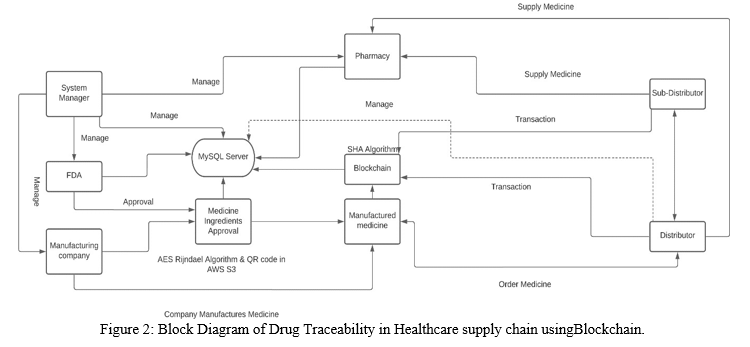
Once the medicines are manufactured, they are distributed to the pharmacies through distributors and sub-distributors. To overcome lack of transparency among participants of supply chain, the transactions between the manufacturing company, distributors, sub-distributors, pharmacies and the consumers are encrypted and secured. When stakeholders like FDA, manufacturing companies, distributors, sub-distributors, and pharmacies tries to fetch the transaction data, if the information is modi?ed, then the application shows that the data is tampered. The FDA, manufacturing companies, distributors, sub-distributors, and pharmacies can all recover the original data from the temporary database whose data hasn’t been altered.
The high-level architecture for drug traceability in healthcare system using blockchain is as shown in ?gure 3.
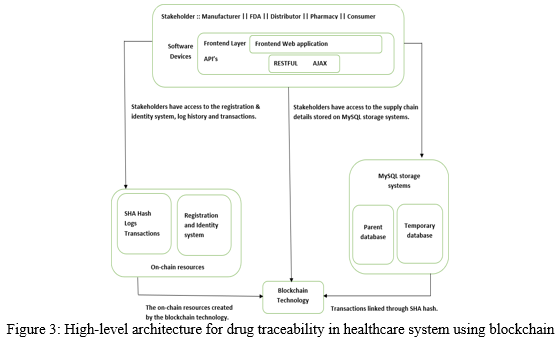
The system components are as follows:
- Stakeholders include regulatory agencies such as FDA, manufacturers, dis-tributors, pharmacies, and consumers. These stakeholders are assigned speci?c functions based on their role in the supply chain. They are also given access to the on-chain resources such as history and log information to track transactions in supply chains.
- MySQL Storage system provides a low-cost off-chain storage to store supply chain transactions data to ensure reliability, accessibility, and integrity of the stored data. The integrity of data is maintained by generating a unique hash using SHA algorithm for every medicine transaction done. The system has a parent database and a temporary database. When the transactions in the primary database are tampered, original data can be recovered from the temporary database whose data isn’t tampered.
- On-chain resources are used to store the logs and events that are created for track and trace operation.
Figure 4 illustrates the relationship among the different entities with the MySQL. Attributes like CompanyId, FDAId, MId, SeriesId, Qty, PHV, CHV, Filepath, DOId, SDOId, SDMOId are considered for mappings for the authorized entities that are allowed to access certain functions within the developed system. It also has several functions needed to carry out the manufacturing process and sale process of drugs. The medicine manufacturer has the option of uploading encrypted QRCode images to the AWS using S3 service.

B. Design Flow
The design ?ow of the project is as shown in Figure 5. This project involves the following main steps to achieve required results.
- The system manager registers the FDA, manufacturing companies and pharmacies by logging into the web application.
- The Manufacturing Company places a request for medicine patent to the FDA for approval. During the medicine patent request submission by the manufacturing company, the Hash codes are generated using SHA-256 algorithm considering the Company Id, Medicine name, Log date and Previous Hash value.
- The Medicine ingredient details are encrypted using AES Rijndael algorithm. The ?rst 2 characters of the hash value generated during medicine patent request are converted to ASCII and are used as key for encryption.
- The encrypted data is written into QR Code image and stored by creating a folder using AWS S3 service. The ?lepath of the QR Code image and the hash value are stored by creating ledgers dynamically using blockchain technology.
- When the FDA log in to the application and tries to fetch the data, the ap- plication checks if the record is tampered or not. If the record is tampered then the original data is recovered from the neighboring node whose data hasn’t been tampered.
- When the FDA clicks on view report, the QR code image is downloaded from the AWS S3 service and extracted for retrieval of data. The extracted data is sent to AES Rijndael algorithm for decryption process to get the original data. The original data is displayed in the dashboard of the FDA.
- The FDA verify the medicine ingredient details and approve or reject the medicine patent. Once the medicine patent is approved by the FDA, the Manufacturing company starts production of the approved medicine.
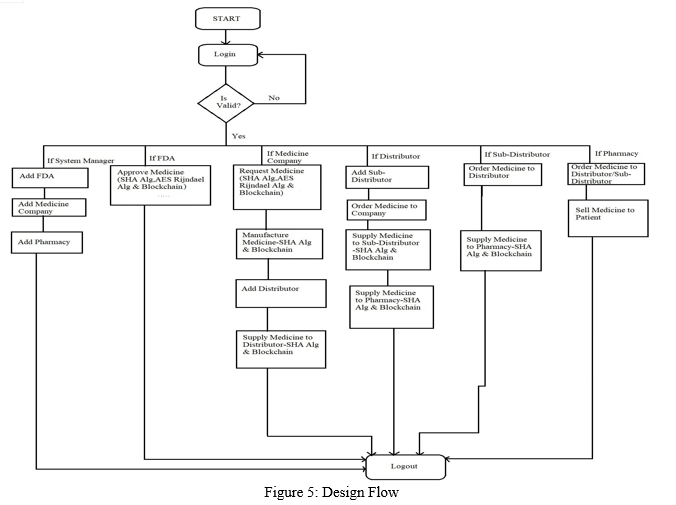
8. During the manufacturing of medicine, the Hash codes are generated using SHA-256 algorithm considering the Company Id, Series Id, Quantity, Log date and Previous Hash value.
9. The Distributor and the sub-distributor places Medicine order request to the manufacturing company indicating the required quantity of the medicines.
10. Based on the availability of the medicine stock, the manufacturing company approves the distributor and sub-distributor’s request for medicine.
11. During the approval of Distributor, Sub-distributor and Pharmacy medicine order requests, the Hash codes are generated using SHA-256 algorithm considering the Medicine Id, Series Id, Quantity, Log date and Previous Hash value. They are stored by creating ledgers dynamically.
12. The transactions performed between the distributor/sub-distributor and the manufacturing company are encrypted using SHA algorithm. If the transactions are tampered, then they are recovered from the neighboring nodes.
13. The pharmacy places medicine order request either to distributor or the sub-distributor based on the requirements. The transactions performed between them are encrypted and updated to the database.
14. The end-to-end transactions between the Manufacturing company and the pharmacy are traced based on the medicine production series ID, which depicts the medicine stock available with the stakeholders and the quantity of medicine sold between the manufacturing company, distributor, sub-distributor and pharmacy.
C. Algorithms For Tampering And Recovering Data Of Medicine Transactions
The following algorithms explains the steps involved in tampering and recovering data of medicine transactions between the stakeholders of the healthcare supply chain.
Algorithm 1 explains the steps involved in tampering the medicine details transaction by modifying the system log date and time and generating a new hash code using SHA-256 algorithm. The generated new hash code is updated to the CHV of the primary database which makes the transaction tampered.
Algorithm 1:: Tampering the medicine details transaction.
Input :: CompanyId, LogDate, MedicineName.
Output:: An event declaring that medicine details transaction is tampered.
Data::
MedicineName :: is the name of the medicine.
CompanyId :: is the medicine manufacturer Id.
LogDate :: is the system log date and time.
Initialization;
Generate a new SHA Hash code by modifying the system log date and time.
Update the CHV using newly created Hash code.
Emit an event declaring that the medicine details transaction is tampered.
Algorithm 2 explains the steps involved in recovering the tampered transactions. Once the stakeholder clicks on recover link button, original data is recovered by updating the CHV of the primary database using the untampered CHV value present in the temporary database.
Algorithm 2 :: Recovering the tampered transactions.
Input :: CHV, TableName, DatabaseName.
Output :: An event declaring that the tampered transaction is recovered and updated with the original data to the primary database.
Data ::
CHV :: is the current hash value of the transaction.
TableName :: is the block name where the transaction is present.
DatabaseName :: is the temporary database name of the system developed.
Initialization;
Compare the CHV of transaction in both primary and temporary database.
If CHV of transaction in primary database is not equal to CHV of transaction in temporary database.
Highlight the transaction of medicine as tampered and copy the original CHV from temporary database and update the permanent database with the original data.
Algorithm 3 explains the steps involved in approving medicine order requests placed by distributor, Sub-distributor and pharmacies.
Algorithm 3 :: Granting Drug sales.
Output :: An event declaring that the drug is sold. Initialization;
If Requested Medicine order quantity <= Company medicine stock.
Emit an event stating medicine order is approved.
Else
Display as Insuf?cient balance of medicine.
IV. RESULTS & DISCUSSION
Using several algorithms testing and validation are done to recover the original data to evaluate its effectiveness to enhance traceability within the pharmaceutical supply chain.
A. Medicine Ingredients Patent Approval And Tampering Of Transactions And Its Recovery
The web application developed for the traceability of medicine supply has a login form page where validations are done for the user type by entering the credentials and allows the user to navigate to the dashboard as shown in Figure 6 and Figure 7.

The system manager performs the task of registering the FDA, Medicine manufacturing companies and Pharmacies as shown in Figure 8
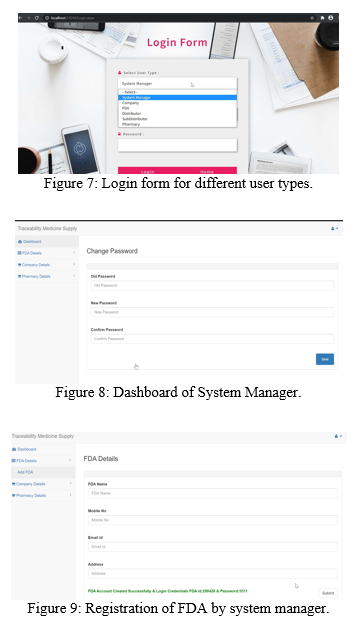
The Figure 9 depicts the generation of the user ID and password for FDA registered by system manager. The user ID and password are generated as a com- bination of a random number chosen (between 100000 and 999999 for user ID and between 1000 and 9999 for password) and the system log date and time at the time of registration. Also, manufacturing company and pharmacies are registered.
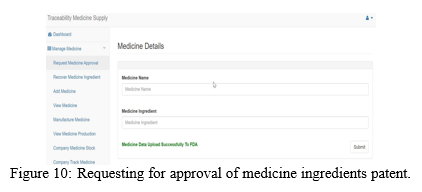
The medicine manufacturing company submits a request for medicine ingredients patent approval to the FDA as shown in the ?gure 10
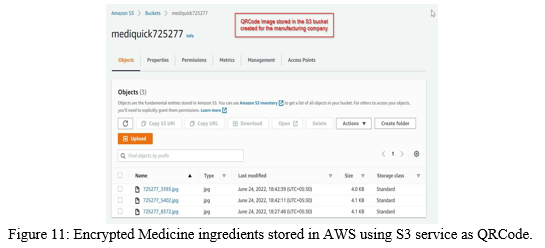
The Medicine ingredient details are written to QRCode Image and stored in AWS using S3 service as shown in the Figure 11
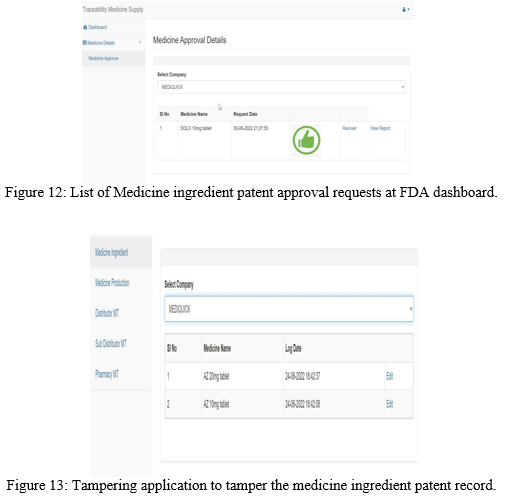
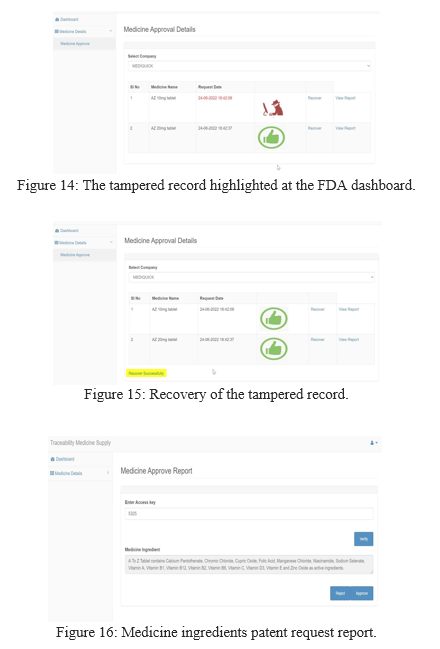
The submitted request shows up in the FDA dashboard as shown in the Figure 12. If the submitted record is tampered by a hacker using a tampering application by modifying the system log date and time as shown in the Figure 13, it leads to change in the hash value of the transaction. In that case the submitted record shows up as tampered in the FDA dashboard as shown in the Figure 14.
The tampered record is recovered by using the recover link button as shown in the Figure 15. Here the current hash value of the tracemedicine database [node 1] is updated with the current hash value stored in the tracemedicine temp database [node 2] whose data wasn’t tampered.
When the FDA clicks on view report link button, random access key is generated. Once veri?ed, the FDA now gets the medicine details from AWS S3 service by extracting the data from QRCode image and decrypts data using SHA, AES Rijndael algorithm as shown in the Figure 16. The FDA can either approve or reject the patent request submitted by the manufacturing company.
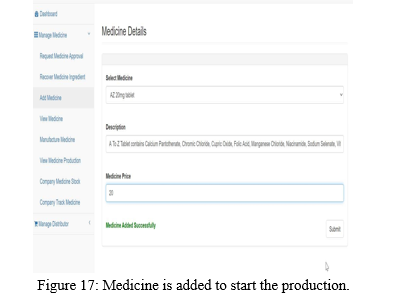
The manufacturing company can start the production of approved medicine by FDA which is shown in the Figure 17 and 18.
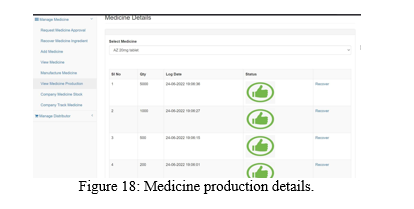
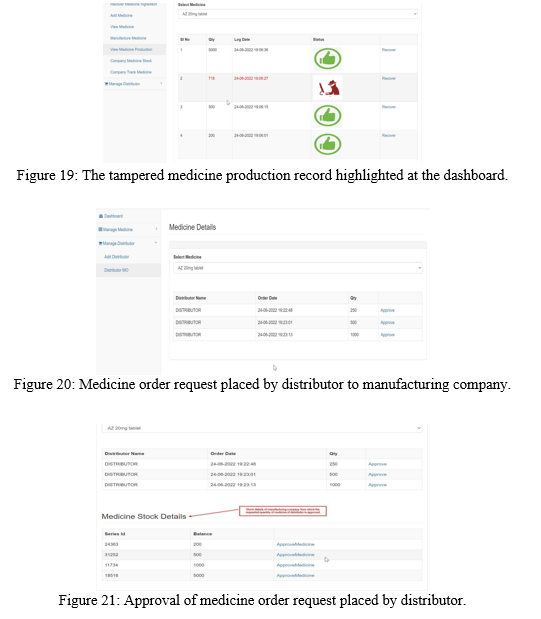
The hacker can also tamper the Medicine production records using the tampering application. It is re?ected in the manufacturing company dashboard as shown in the Figure 19 and the tampered record can be recovered from the node whose data isn’t tampered.
Once the medicine production is successful, the distributor places order for certain quantity of medicine to the manufacturing company. The order is approved by the manufacturing company from the available medicine stock based on their production series ID as shown in the Figure 20 and Figure 21.
Similarly, the sub-distributor places a medicine order request to the distributor and the distributor approves the request from the medicine stock available with him as shown in the Figure 22.
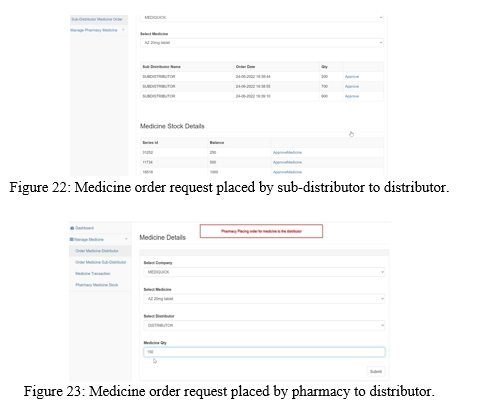
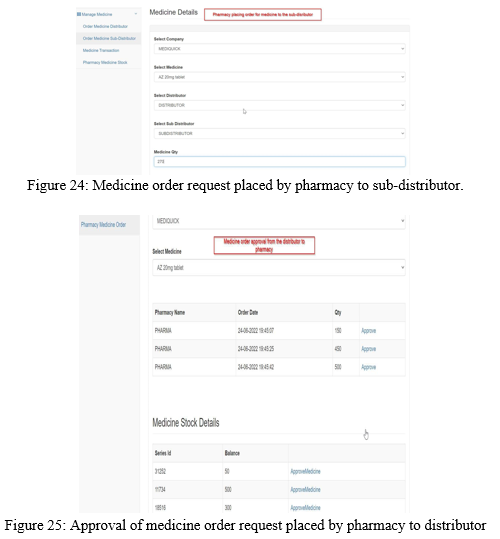
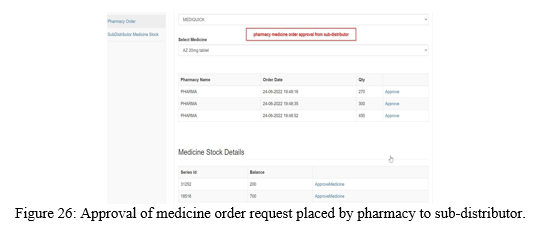
Similarly, the pharmacy can place medicine order request either to distributor or sub- distributor as shown in the Figure 23 and 24. The approval for the medicine order by distributor and sub-distributor is shown in the Figure 25 and Figure 26.
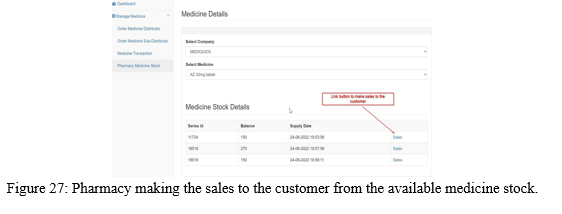
Once the medicine order request of pharmacy is approved, the pharmacy can make sales to the customer from the available medicine stock through the sales link button as shown in the Figure 27.
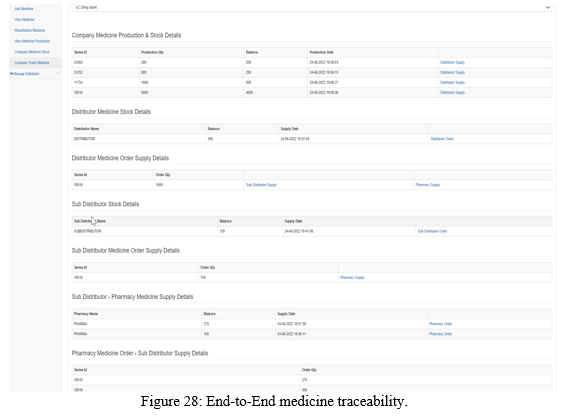
B. End-To-End Traceability Of Medicines
The end-to-end traceability of the medicines, i.e., from the manufacturing company till the pharmacy is achieved as shown in the Figure 28 with the production date and supply date information.
Conclusion
The developed system introduces a non-repudiation process that provides origin of data and proof of probity and traceability for the fast perseverance of any disputation in the pharma supply chain. It helps in detecting medicines that do not contain deliberated active ingredients. The system is evaluated to trace and track medicines in a decentralized manner from manufacturer to end-consumer to achieve end-to- end transparency in the pharmaceutical supply chain. The system is able to accomplish tri?e-proof logs which helps in identifying the counterfeit drug. The proposed solution holds cryptographic fundamentals of Blockchain technology to accomplish secured logs of events within the supply chain ensuring drug safety and obliterate counterfeits.
References
[1] Ahmad Musamih, Khaled Salah (Senior Member, IEEE), Raja Jayaraman, Junaid Arshad, Mazin Debe, Yousof Al-Hammadi and Samer Ellahham, “A Blockchain-Based Approach for Drug Traceability in Healthcare Supply Chain”, Vol 9, IEEE, 2021. [2] Randhir Kumar, Rakesh Tripathi, “Traceability of counterfeit medicine sup- ply chain through Blockchain”, 11th International Conference on Commu- nication Systems and networks, IEEE, 2019. [3] Sudeep Tanwar, Karan Parekh, Richard Evans, “Blockchain-based electronic healthcare record system for healthcare 4.0 applications”, Journal of Infor- mation Security and Applications, Volume 50, Elsevier, 2020. [4] Haq, Ijazul & Muselemu, Olivier, “Blockchain Technology in Pharmaceu- tical Industry to Prevent Counterfeit Drugs”, International Journal of Com- puter Applications, IJCA, 2018. [5] Tseng J. H, Liao Y. C, Chong B, Liao S. W, “Governance on the Drug Supply Chain via Gcoin Blockchain”, Int. J. Environ. Res. Public Health 2018. [6] Bryatov S, Borodinov A, “Blockchain technology in the pharmaceutical sup- ply chain: researching a business model based on Hyperledger Fabric”, In- formation Technology and Nanotechnology, 2019. [7] Shaker Alharthi, Paul R. Cerotti and Shaghayegh Maleki Far, “An Explo- ration of The Role of Blockchain in The Sustainability and Effectiveness of The Pharmaceutical Supply Chain”, Journal of Supply Chain and Customer Relationship Management, Vol. 2020. [8] Shruti Srivastava, Arun Bhadauria, Sunil Dhaneshwar, Suneel Gupta,” Traceability and Transparency In Supply Chain Management System Of Pharmaceutical Goods Through Block Chain” International Journal Of Sci- enti?c & Technology Research Volume 8, Issue 12, December 2019. [9] Alaa A. Abd-alrazaq, Mohannad Alajlani, Dari Alhuwail, Aiman Er- bad, Anna Giannicchi, Zubair Shah, Mounir Hamdi, Mowafa Househ, “Blockchain technologies to mitigate COVID-19 challenges: A scoping re- view”, Computer Methods and Programs in Biomedicine Update, Volume 1, 2021. [10] C. Antal, T. Cioara, M. Antal and I. Anghel, \"Blockchain platform for COVID-19 vaccine supply management”, IEEE Open Journal of the Com- puter Society. [11] Jamil F, Hang L, Kim K and Kim D , “A Novel Medical Blockchain Model for Drug Supply Chain Integrity Management in a Smart Hospital”, Elec- tronics, vol. 8, 2019. [12] Pashkov V and Soloviov O, “Legal implementation of blockchain technol- ogy in pharmacy”, EDP Sciences, p. 01027. [13] Raj R, Rai N and Agarwal S, “Anticounterfeiting in Pharmaceutical Supply Chain by establishing Proof of Ownership”, IEEE. [14] Clark B and Burstall R, “Blockchain, IP and the pharma industry-how dis- tributed ledger technologies can help secure the pharma supply chain”, Jour- nal of Intellectual Property Law and Practice, vol. 13, no. 7, 2018. [15] Shrikant GP, “Traceability and detection of counterfeit medicine supply chain through Blockchain”, Journal of the Gujarat Research Society, vol. 21, no. 8, 2019.
Copyright
Copyright © 2022 Varshad V, Vinay Prasad M S. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET46221
Publish Date : 2022-08-07
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

