Ijraset Journal For Research in Applied Science and Engineering Technology
- Home / Ijraset
- On This Page
- Abstract
- Introduction
- Conclusion
- References
- Copyright
Pharmaceutical Micropellets: A Multi-particulate Drug Delivery System
Authors: Ankit Agrawal, Sourabh D Jain, Ashutosh K Das, Arun K Gupta
DOI Link: https://doi.org/10.22214/ijraset.2023.49427
Certificate: View Certificate
Abstract
The major target of Pharmaceutical Sciences is to design a successful and suitable dosage forms for effective therapy, considering individual patient needs and compliance. Development of new technology or development of new dosage form using the existing technology is growing in importance and attracting increased interest, as they are specifically effective at a comparably low dose. Pellets falling in the size range of 1-1000?m are of great interest in the pharmaceutical industry for a variety of reasons which are not only offer flexibility in dosage form design and development, but are also utilized to improve the safety and efficacy of bioactive agents. The most important factor responsible for the proliferation of pelletized products is the popularity of controlled release technology in the delivery of drugs. Moreover, controlled release pellets are less susceptible to dose dumping than the reservoir-type single unit formulations. In case of oral products these pellets solve difficult taste-masking problems, disperse freely in the gastro intestinal tract, invariably maximize drug absorption, reduce peak plasma fluctuations and minimize potential side effects without appreciably lowering drug bioavailability, and also reduce variations in gastric emptying rate and overall transit time, thus, intra and inter subject variability of plasma profiles, which are common with single unit regimens, are minimized.
Introduction
I. INTRODUCTION
Optimization of the drug release of peroral dosage forms and adjusting it to the absorption window of the active ingredient is the fundamental point in the development of a new pharmaceutical product [1, 2]. However, there may be different aspects in cases when local effect is required. In this type of therapy the concept of the dissolution of active ingredients should be revaluated [3]. The enhancement of the bioavailability according to the Biopharmaceutical Classification System (BCS) [4] cannot be applied invariably [6], since importance of the absorption and the permeability is minor. Conversely, there are other essential parameters which should be taken into account during the development of these preparations, including the adhesivity to the mucous membrane, large contact surface of the drug particles, the profile and the time of drug release [7]. Among several diseases, gastrointestinal candidiasis (GIC) [8] was chosen to formulate and optimize a medicinal product taking into consideration the enhancement of the bioavailability of local effect.
Application of pellets in this therapy offers even more advantages. They are suitable to optimize drug release by their particle size, coating thickness or material.
Pellets among multiparticulate dosage forms offer several advantages generally in the therapy related to safety and effectiveness of the medicinal product such as individual reproducibility of gastric emptying, more regular absorption, increasingly stable, predictable plasma levels and a reduced risk of high concentrations [9,10]. This basic concept of multiple-unit systems is the fact that the dose of the active ingredient is released by the individual subunits, and the functionality of the entire dose depends on the quality of the subunits [11]. Pellets are suitable for further processing in order to optimize drug release by combining different particle size fractions and coating thickness [12] in capsules or compressing them into tablet.
Manufacturing of pellets requires specialized equipments, technologies and excipients. Pellets are usually prepared by wet agglomeration of fine powders of active ingredient and excipients into spherical units in closed granulating systems, i.e. in rotor-fluid granulators or high-shear mixers [13]. To produce pellets in a high shear mixer, the process involves distinct phases: homogenization of powders, granulation, spheronization and drying. The primer nucleus of future pellets is formed by binder spraying and dispersing during the agitation. Being a multivariate process, it is important to identify and control the process variables, i.e. the appropriate agitation prevents the development of too large particles [14,15]. Since agglomerates undergo densification as mixing and spraying, the process time is expected as a critical parameter influencing quality of pellets.
Although various experiments were drawn up to investigate the effect of the formulation variables on the physical characteristics of pellets, only a few reports can be found in the pharmaceutical literature investigating the large number of process variables together during the pelletization concerning both the physical properties of granules and the drug release profile of the dosage form [16].
Modelling the effect of process variables with factorial designs and analysis of the response surfaces is a powerful, efficient and systematic tool that shortens the time required for the development of pharmaceutical dosage forms and improves research and development work [20].
Definition of pellets is not uniform, sometimes they are discussed together with granules and often the terms pellet-micropellet are not distinguished. Their preparation can be carried out in many ways with similarities to the preparation of granules [21]. According to the literature there are a few fix points which can characterize the pellet dosage form as spherical particles with almost smooth surface and compact texture independently to the method of preparation. If their size is below 1 mm, the term “micropellet”, between 1 and 2 mm, the term ”minipellet” can be used.
According to the structure, as seen on Fig. 1 we can distinguish pellets containing the API in the
- Matrix and in the
- Coating.
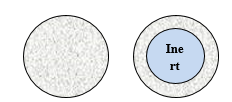
Pellets have several pharmaceutical technological advantages, such as excellent flowability, easily optimizable particle size distribution, good mechanical stability, abrasion-resistant structure and optimizable coating due to the regular, uniform, smooth surface [13].
A. Interaction Between Particles, Distribution Of Binder Liquid
Interaction between solid particles during granulation was first scientifically investigated by Rumpf in the 50s [23]. In his work the following forces were listed:
- Attrition Forces: Gravitational force, magnetic force, electrostatic force or the van der Waal force play less role in the interactions between particles, than capillary forces,
- Solid Bridges: They are developing during the formation of inorganic bonds, chemical reactions, crystallisation and melting phenomena,
- Shape Closing Bridges: they are developing due to the different shape and size of particles during pelletization getting into physical contact to each other,
- Liquid Bridges: They are forming due to capillary forces which ensures the cohesion of particles and decrease the attrition forces.
Newitt and Conway-Jones distinguished four phases during the development of spherical particles according to the distribution of the binder liquid: the pendular state with liquid bridges, the funicular state in which the amount of liquid bridges increases capillary state and the droplet state [24].
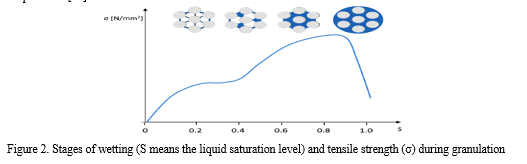
During pelletization right before the addition of any liquid small size particle-aggregates can be observed due to the secondary attrition forces. After the beginning of addition of liquid first drops start to connect the primary particles at the contact points. At the next, funicular stage, the liquid bridges may reach each other and more than two primary particles can be connected via this liquid bridge. Gaps are also started to be filled with liquid at this stage. At the next stage the structure of the pellet is formed and due to the capillary state the liquid added to the system is absorbed by the aggregated structure. The mechanical stability of the pellet is the highest at this point. Increasing the amount of the added liquid the droplet state can be reached, when the structure weakens and the system turns into paste or suspension.
Basically we can distinguish two types of binder liquid additions:
a. the size of the liquid drop is bigger than the size of the primary particle (drop addition),
b. the size of the liquid drop is smaller than the size of the primary particle (spray addition).
During the wetting of the powder mixture the contact angle (Θ) is the most important physical property which describes the wetting mechanism on the solid/liquid/gas boundary surface, which is the angle of solid/liquid and liquid/gas boundary surface.
We can distinguish:
- contact wetting (Θ>0°),
- film wetting (Θ=0°).
In case of Θ<90° we can state, that the substance is lyophilic, in case of Θ>90° it is lyophobic. After the equilibrium of solid/liquid/gas phases the relationship between the surface tension and the contact angle can be expressed by the Young-Dupré equation:
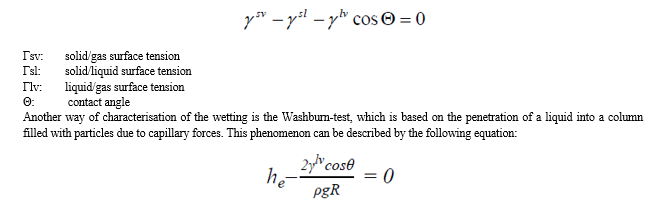
he: height of the liquid after the equilibrium
R: average radius of capillaries in the particle agglomerate
p: density of the liquid
g: gravitational force
γlv Θ: adhesion tension
B. Particle Growth And Attrition Mechanism During Pelletization
During pelletization the balance of particle growth and attrition mechanisms is very fundamental. We can distinguish four types of these mechanisms:
- Nucleation: is the first step during particle growth, when primary particles are getting into contact to each other and stick together in the presence of the binder liquid forming a nucleus.
- Coalescence: due to the collision of particles bigger aggregates are formed.
- Layering – also called as „balling”, when a larger amount of primary particles stick on the surface of a bigger aggregate due to liquid addition.
- Breakage: there are more mechanisms for the breakage of granules/pellets:
a. Crushing: during which smaller granules or pellets crush and get onto the surface of bigger particles. There are several mechanisms of crushing:
- shattering,
- fragmentation,
- abrasion,
b. Abrasion transfer: during which the material transfer occurs between two colliding particles, while both particles stay intact.
These mechanisms are often described mathematically by different models according to different steps of pellet formation. Strength of liquid bridge between two colliding particles determines whether they stay intact or attach together. The greatest force which depends on the relative amount between two particles comparing to their volume was first described by Rumpf [23]:

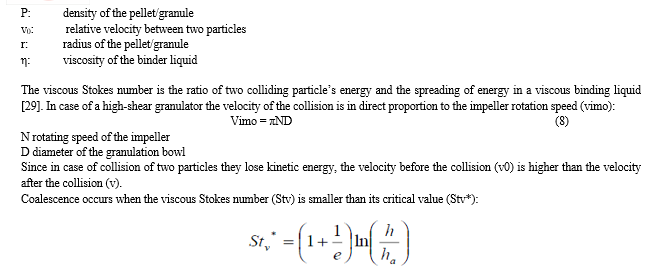
e: rearrangement coefficient, which can be calculated from the difference of velocity before and after the collision
h: thickness of the liquid layer on the surface of the pellet
ha: surface roughness (diameter of the primary particles is also ’ha’)
According to the theory above there are three cases regarding the values Stv and Stv*:
- Stv « Stv* - in this case all collisions are successful
- Stv = Stv* - some collisions are successful
- Stv » Stv* - none of the collisions are successful
From the expressions above we can state, that the collision of the particles and growth by coalescence has higher probability at low particle density, low impeller speed, small particle size, high viscosity liquid binder, small rearrangement coefficient, thick surface liquid layer and smooth particle surface. Since these variables usually change in function of time, above calculations can only be used for retrospective studies.
C. Operation Of Pelletization
Similarly to the granules there are three main types of operations of pelletization:
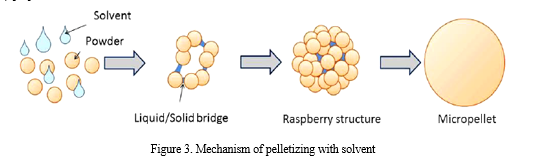
- Solvent Pelletization: The applied liquid dissolves totally or partially one or more components of the powder mixture. During the addition of the liquid a saturated solution develops on the surface of the particles which binds them together. After the evaporation of the solvent the dissolved component gathers on the surface of the pellet and crystallizes, forming a thick coating (Fig.3.) [21].
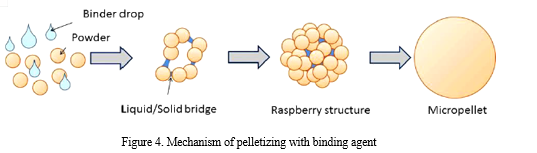
2. Binder Pelletization: Usually high molecular weight polymers are used. The powder mixture is usually wetted by the proper amount of binder liquid and then it is kneaded. The binder material also can be added in its powder form, but these pellets’ mechanical properties are usually poor (Fig.4.) [21].
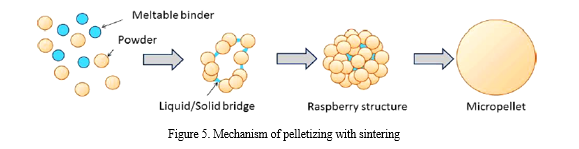
3. Sinter Pelletization: In this case one or more components of the powder mixture has a lower melting point (usually high molecular weight macrogol). Heating the mixture partial melting occurs. Spray drying or freeze drying this mixture result almost perfectly spherical pellets (Fig.5.) [21].
a. Mixing Method
This is the simplest way of pelletisation. This method usually results heterogeneous particle size distribution. During this method the powder mixture and the liquid binder both are placed into a rotating caldron, where the pellets are formed [13].
b. Rotary Disk Method
The powder mixture is placed on the surface of a rotating flat disk or a conical body, while the binder liquid is added from the centre of the rotating axis. Developing particles roll to the edge of the rotating disk due to the centrifugal force, during which they gain their spherical shape. Using this method spheronization of preliminary prepared granules can also be carried out (Fig.6.).

c. Extrusion-spheronization method
This is one of the most commonly used pellet preparation method [27]. Powder components are kneaded with the liquid, then the wet mass is extruded through a 0.5-2.0 mm diameter hole size disk. After this step the plastic and wet extrudate is spheronized (Fig.7.) [30].

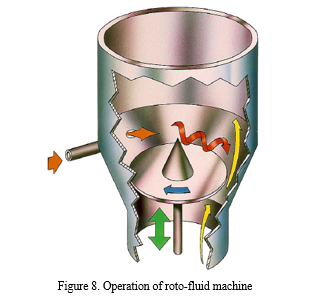
d. Roto-fluid method
Applying fluid-bed granulation, spheronization and drying is carried out in one complex step. There is a rotating disk at the bottom of the machine with a conical body in the centre (Fig. 8.). Due to the fluid air, the rotating disk and the conical body particles circulate around the wall of the working area and roll on the surface of the disk. The liquid is added towards the centre of the working space spraying the liquid to the rotational direction. Reaching the proper particle size the liquid addition is stopped and from this point only spheronization and drying is carried out. Roto-fluid pelletization is one of the most power consuming and expensive methods, its advantage is the high productivity and the one-step production [21].
e. Spray drying and freeze drying
API is dispersed in the vehicle’s solution, and then it is sprayed. Perfectly spherical droplets formed during this preparation are then dried by hot or cold air [21].
f. High-shear pelletization
This method unifies the main steps of the classic wet granulation (homogenization, wetting, kneading and particle forming) and the spheronization. The production is carried out in one working space similarly to the fluidisation in one complex step (Fig. 9.). During high-shear granulation intense mixing of the components and the presence of the shearing forces results the smooth and compact spherical particles. The mechanical parameters of the end-product depend on the operation variables. Spray or drop form of liquid is immediately dispersed due to the high rotation speed of mixer elements, thus reaching a quick and perfect wetting. Main mixer elements consists the impeller and the chopper. The impeller is at the bottom of the working space reaching rotation speed of 500-2000 rpm. The impeller moves the mass around the working space and in radial direction outwards from the centre to the wall. The mass produces a whirling movement and continuously falls back towards the centre. Chopper blades can be placed at the bottom, top or tangentially to the rotation axis of the impeller. This mixer element is composed of more smaller, thinner blades (3-6), which enhance the shearing forces during the production. Their speed also varies between 500-2000 rpm. Pellets produced in the high-shear machine can be dried by microwave or heating double-jacket as well. The production method is extremely quick, there is low risk of contamination due to the closed working area. Choosing the proper operation variables and applying the proper excipients almost all pharmaceutical technological and biopharmaceutical parameters can be optimized and controlled [13].
Most important variables during high-shear production related to the equipment:
1. Mixing bowl
a. shape
b. size
c. material
2. Impeller
a. shape
b. size
c. speed
3. Chopper
a. shape
b. size
c. speed
4. Temperature of the working space
5. Operation time
Most important variables during high-shear production related to the material are:
- Powder composition (particle size distribution, physical properties)
- Weight
- Liquid binder
a. viscosity
b. density
c. amount
d. dosing speed
e. dosing method
- Forces During High-Shear Pelletization
During pelletization presence and equilibrium of integration and reduction effects leads to the formation of pellets, which are based on the following forces:
- gravitational force
- acceleration force
- centrifugal force
- resistance of the pelletizing bowl
- fluid effects developed by the impeller
For the characterisation of the forces effecting the preparation, the tip speed of the impeller is commonly used, which is directly proportional to its shearing force. The perimetric speed is responsible for the movement of the wet mass. This value can be calculated by the following expression:
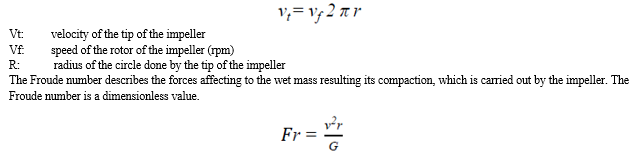
Fr: Froude number
V: speed of the impeller
r: radius of the circle done by the tip of the impeller
G: acceleration of gravity
At smaller, laboratory production scale the Froude number is usually higher, than in industrial scale. The higher number means bigger pellet size, more compact structure, which can result the cohesion of the particles to the pelletizing bowl [33].
- Determination Of End-Point Of High-Shear Pelletization
Critical point of the high-shear pelletization is the determination of the end-point of the production. The end-point is usually determined by the technologist according to the particle size, the particle size distribution, density, and the powder rheology. Instrumented high-shear machines have several parameters which are observed in order to forecast and determine the optimal end-point [34].
Most important methods Include:
- Measurement Of Power Consumption
One of the most commonly used methods is the measurement of the power consumption of the impeller. Scientific literature several times reported that the increase of the pellet size has close correlation to the increasing power consumption. This correlation is not linear, but it is characteristic and reproducible to the definite powder composition [35].
- Measurement Of The Impeller Torque
The second most commonly used method is the measurement of the impeller torque, which has good correlation to the previously mentioned method, since sometimes it is recommended to use the more complex measurement of the impeller torque due to insensitivities in the power consumption profile in case of definite weight of mass or other specific parameters. The profile of the impeller torque divides the pellet formation process into five phases. In the first phase the powder is moistened without forming liquid bridges. In the second phase liquid bridges are formed between the particles and first granules are formed. Formation of coarse agglomerates could be observed in the third phase and in the fourth phase large areas are filled with liquid. In the fifth phase when the liquid saturation equals 100%, the system becomes a suspension [35]. The end-point of the pelletization is usually during the fourth phase.
- Other Methods Based On The Measurement Of Impeller Torque
- Torque Rheometer Method: the third most commonly used method, which also provides information on rheological properties [38].
- Torque Reaction Force Method: applies Newton’s third law of action-reaction. During the measurement the rotor tries to move against the rotational direction which develops forces inside the engine, which can be detected and used for the end-point determination [35].
- Alternative Measurement Methods
There are several other measurement methods supplementing the lacks of the methods mentioned above:
- Application of NIR sensor – this method is based on the moisture changes of the particles. Its disadvantage is that the sensor can only detect the moisture on the surface of the wet mass [39]].
- Focused Beam Reflectance Measurement (FBMR) – the method is based on the particle size determination. This method cannot be used for end-point determination alone, but it can be a perfect supplement of other methods [40].
- Acoustic Sensor Measurement – one of the oldest methods of the end-point determination which can be used alone and as a supplementing tool as well. Development of the technology in recent years and modern analysis softwares provide more potential to this method [31].
D. Biopharmaceutical Background
Gastrointestinal candidiasis (GIC) often occurs in patients suffering from immunosuppression (e.g. related to HIV infection), but many researchers found GIC also in non-immuno suppressed humans. GIC belongs to one of the most discussed diseases, since it is still undecided whether it can cause systemic symptoms or not. Researches, however, indicate GIC diseases may be more prevalent than it is supposed. Several antifungal pharmaceutical ingredients are considered to be effective in the therapy of GIC as listed in Table I [43].
Table I. BCS classification of antifungal agents used in candidiasis
|
BCS Class |
Antifungal agent |
Biopharmaceutical properties |
|
1 |
--- |
High Solubility, High Permeability |
|
2 |
Clotrimazole, Itraconazole |
Low Solubility, High Permeability |
|
3 |
Fluconazole, Capsofungin |
High Solubility, Low Permeability |
|
4 |
Amphotericin B, Nystatin |
Low Solubility, Low Permeability |
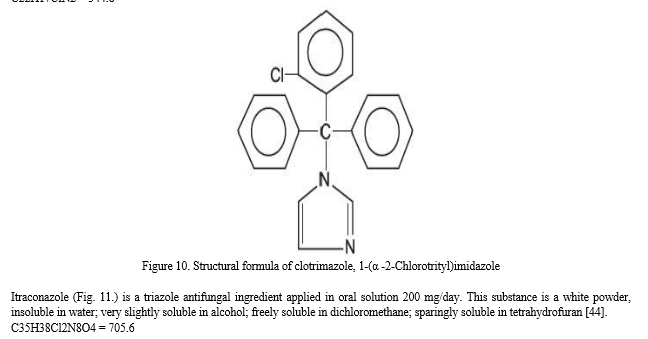
Clotrimazole (Fig. 10.) belongs to the group of imidazoles, it is applied in 10 mg troches 5 times a day in mucosal candidiasis. According to the Ph. Eur. 8. it is white or pale yellow crystalline powder. It is practically insoluble in water; soluble in alcohol, in acetone, in chloroform, and in methyl alcohol [44].
C22H17ClN2 = 344.8
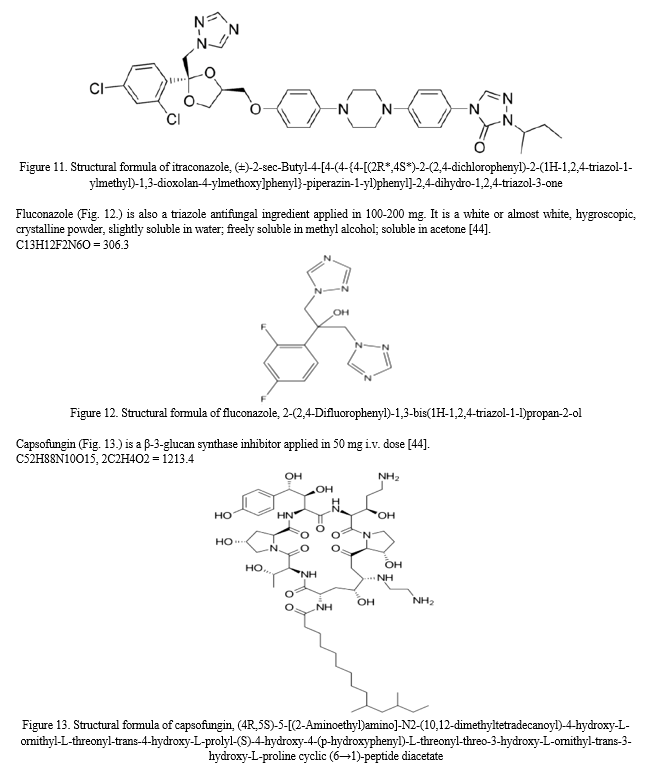
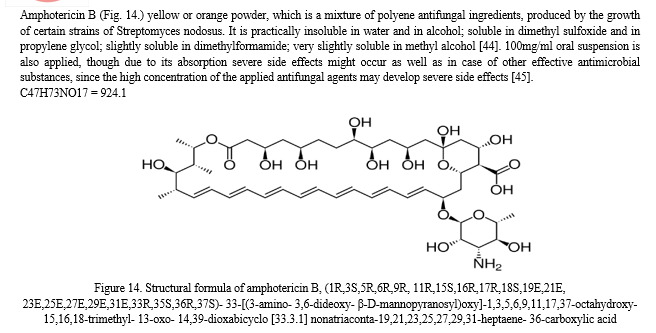
Pharmaceutical character of nystatin will be summarized later in chapter 4.1.1. Its effect is based on the topical action and the contact time with the mucous membrane, thus the formulation should adhere to the affected area as long as possible, however available dosage forms are unable to achieve an optimized effect, lacking the specialities of the adequate topical treatment. To reach this aim, mucoadhesive materials are needed to be added. Application of mucoadhesive polymers in the formulation, such as the anionic carbomer (CPL) and the non-ionic hydroxyethylcellulose (HEC) is a possible method for the targeted mucosal drug delivery which has been introduced to the pharmaceutics in the late 1960s [48].
In our study after preliminary examinations of high-shear granulation, nystatin was chosen as one of the most adequate API for the treatment of GIC due to its preferential properties [43]. Micropellets containing nystatin with increased specific surface were produced according to an experimental design containing bioadhesive excipients, which can prolong the contact time with the mucous membranes. The pellet size and the proportion of bioadhesive substances (carbopol®/hydroxyethylcellulose) were taken into consideration as variables affecting the therapy [51].
During the evaluation, in vitro spectrophotometrical, microbiological dissolution, ex vivo bioadhesion and swelling tests were carried out.
Effectiveness of the produced medicament samples was evaluated by the determination of the drug release [52]. Developing possibilities of nystatin determination methods are continuing intensively, since classic UV analytical methods in the pharmaceutical practice are not suitable to follow exact changes of antifungal activity of nystatin [53]. UV spectrum of nystatin demonstrates three main local maxima at 290, 306 and 318 nm which are only reliable at the same molecular state. Micellar structure of the polyene nystatin can lead to differences in the UV spectrum [54]. Numerous colour reactions were also tested, but there was no confirmed correlation with the biological activity [55]. A generalized assumption was later published regarding the lack of relationship for all polyene antibiotics between the UV spectrophotometric assay and the biological activity, which eventuated to include an agar diffusion method for the quantitative assay of nystatin in the Ph. Eur. 5. However, the agar diffusion method is sometimes time, cost and power consuming especially in case of large quantity of samples. Application of thin layer chromatography (TLC) based direct bioautography during the drug dissolution tests, microbiologically detected dissolution (MDD) can be carried out [56]. The MDD does not need strict sterile conditions and it is almost 100 times more sensitive than the agar diffusion method. Novel approach to enhance the UV spectrophotometric determination and to achieve better correlation is the application of the third-derivative spectrophotometric evaluation, which was applied in our experiments. The objective of the present study is to offer an optimized drug formulation by revealing the relationship between average pellet size, CPL and HEC content and optimized drug release to achieve a site-specific antifungal therapy.
Conclusion
Pelletization technology is gaining very much attention in present times as it has provided a competent pathway for the oral drug delivery system manufacturing. Pellets offers many biopharmaceuticals as well as technological recompenses over the conventional dosage forms. Pellets offer required strength for dose which can be blended for the delivery of incompatible bioactive agents and helps in providing different release profiles. In this review we will be discussing the extensively used techniques of pelletization, extrusion and spheronization in detail with their methods and applications in the field of pharmacy as a competent pathway for the novel drug delivery systems. An even more exciting area of application is the use of micropellets for subcutaneous and intramuscular administration and has a tremendous potential as depot formulations using biodegradable polymers. The pelletisation technology can be used to manufacture a broad range of pellet sizes and very small pellets down to 50?m containing drug loads of <95% achievable. This technology delivers almost perfectly spherical particles exhibiting a very narrow particle size distribution and excellent flow properties. In order to design and develop a suitable dosage form, the mechanisms and effective parameters need to be understood and clarified. Therefore, we review the importance and rationale of pharmaceutical development and production of drug pellets, possible formulation variables and aids during the pellets formulation, technologies involved in the fabrication of pellets, various mechanisms involved in the formation of pellets, characterization and application of the pellets and also discussed the results of recent researches on the pharmaceutical drug pellets.
References
[1] Nikhil Saxena , Rakesh Kumar , Narendra Kumar, Amit Mittal, Pellets: Formulation Skilss, Its Assessment Considerations For Multiparticulate Remedy Conveyance Coordination And Their Applicability In Herbals, European Journal of Molecular & Clinical Medicine, Volume 7, Issue 07, 2020, 2514-2524. [2] Kishore Manoharan, Navya Ajitkumar Bhaskaran, Lalit Kumar, Pellets and Techniques of Pelletization, Research Journal of Pharmacy & Technology, Vol 12, 2019. [3] Mansi Paradkar, Jinal Amin, Formulation development and evaluation of colon targeted delayed release methotrexate pellets for the treatment of colonic carcinoma, Brazilian Journal of Pharmaceutical Sciences, 2018;54(4):e17222. [4] K. Srinivasarao, K. Sai Lakshmi Jyothirmai and N. Rama Rao, Pellets and Pelletization Techniques: A Review, IJRPC 2017, 7(2), 141-147. [5] Niti Yadav, Anurag Verma, Pharmaceutical Pellets: A Versatile Carrier for Oral Controlled Delivery of Drugs, Indian Journal of Pharmaceutical Education and Research, Vol 50, Issue 3, ul-Sep (Suppl.), 2016. [6] V. R. Sirisha K. 1 , K. Vijaya Sri 2 , K. Suresh 3 , G. Kamalakar Reddy 3 , N. Devanna 4, A Review Of Pellets And Pelletization Process - A Multiparticulate Drug Delivery System, IJPSR, 2013; Vol. 4(6): 2145-2158. [7] P. Costa, J. Manuel, S. Lobo: Modelling and comparison of dissolution profiles. Eur. J. Pharm. Sci. 13: 123–133.2001. [8] F.O. Costa, J.J.S. Sousa, A.A.C.C. Pais, S.J. Formosinho: Comparison of dissolution profiles of Ibuprofen pellets. J. Control. Release 89: 199–212.2003. [9] M. Siewert J. Dressman, C. Brown, V. Shah: FIP/AAPS guidelines for dissolution/in vitro release testing of novel/special dosage forms. Dissolution Technologies 6–15.2003. [10] G. L. Amidon, H. Lennernas, V. P. Shah, J. R. Crison: A theoretical basis for a biopharmaceutic drug classification: the correlation of in vitro drug product dissolution and in vivo bioavailability. Pharmaceut. Res. 12: 413–420.1995. [11] Dévay, I. Antal: A gyógyszeres terápia biofarmáciai alapjai. Medicina Könyvkiadó, Budapest, 2009. [12] R. C. Schellekens, F. E. Stuurman, F. H. van der Weert, J. G. Kosterink, H. W. Frijlink: A novel dissolution method relevant to intestinal release behaviour and its application in the evaluation of modified release mesalazine products. Eur. J.Pharm. Sci. 30: 15–20.2007. [13] J. J. Sousa, A. Sousa, F. Podczeck, J. M. Newton: Influence of process conditions on drug release from pellets. Int. J. Pharm. 144: 159–169.1996. [14] R. E 2nd Cater: Chronic intestinal candidiasis as a possible etiological factor in the chronic fatigue syndrome. Med. Hypotheses 44:507–515.1995. [15] Järvinen, S. Nykänen, L. Paasiniemi, T. Hirjärvi-Lahti, J. Mattila: Enteric coating reduces upper gastrointestinal adverse reactions to doxycycline. Clin. Drug Investig. 10: 323–7.1995. [16] P. Macheras, A. Dokoumetzidis: On the heterogeneity of drug dissolution and release. Pharm. Res. 17: 108–12.2000. [17] R. Chopra, G. Alderborn, F. Podczeck, J. M. Newton: The influence of pellet shape and surface properties on the drug release from uncoated and coated pellets. Int. J. Pharm. 239: 171–8.2002. [18] R. Chatlapalli, B. D. Rohera: Physical characterization of HPMC and HEC and investigation of their use as pelletization aids. Int. J. Pharm. 161: 179–93.1998. [19] Ghebre-Sellassie: Pharmaceutical Pelletization Technology: Drugs and pharmaceutical sciences. Marcel Dekker, New York, 1–13.1989. [20] Faure, P. York, R. C. Rowe: Process control and scale-up of pharmaceutical wet granulation processes: a review. Eur. J. Pharm. Biopharm. 52: 269–77.2001. [21] C. Jorgensen, P. Luukkonen, J. Rantanen, R. Schaefer, A. M. Juppo, J. Vliruusi: Comparison of torque measurements and near-infrared spectroscopy in characterization of a wet granulation process. J. Pharm. Sci. 93: 2232–43.2004. [22] H. A. Rashid, J. Heinamaki, O. Antikainen, J. Yliruusi: Influence of the centrifugal granulating process on the properties of layered pellets, Eur. J. Pharm. Biopharm. 51: 227–234.2001. [23] P. G. Paterakis, E. S. Korakianiti, P. P. Dallas, D. M. Rekkas: Evaluation and simultaneous optimization of some pellets characteristics using a 33 factorial design and the desirability function, Int. J. Pharm. 248: 51–60.2002. [24] J.B. Schwartz, R. E. O\'Connor: Optimization techniques in pharmaceutical formulation and processing. Editors: Banker GS, Rhodes CT. Modern Pharmaceutics. 3rd ed. Marcel Dekker, New York, 727–52.1997. [25] D. Voinovich, M. Moneghini, B. Perissutti, E. Franceschinis: Melt pelletization in high shear mixer using a hydrophobic melt binder: influence of some apparatus and process variables. Eur. J. Pharm. Biopharm. 52: 305–13.2001. [26] F. Zhou, C. Vervaet, J. P. Remon: Influence of processing ont he characteristics of matrix pellets based on microcrystalline waxes and starch derivatives, Int. J. Pharm. 147: 23–30.1997. [27] Dévay: The Theory and Practice of Pharmaceutical Technology. University of Pécs, Pécs, 2013. [28] P. Holm: Theory of granulation. Editor: Parikh D.M., Handbook of Pharmaceutical Granulation Technology, Marcel Dekker, New York, 7–23.1997. [29] H. Rumpf: The strength of granules and agglomerates, In: Agglomeration, ed. K. W.A., Interscience, New York, 379–418.1962. [30] [24] D. M. Newitt, J.M. Conway-Jones: A contribution to the theory and practice of granulation, Trans. Instn. Chem. Engrs. 36: 422–442.1958. [31] N. Ouchiyama, T. Tanaka: The propability of coalescence in granulation kinetics, Ind. Eng. Chem. Process Des. Dev. 14.3: 286–289.1975. [32] H. G. Kristensen, P. Holm, T. Schæfer: Mechanical properties of moist agglomerates in relation to granulation mechanisms. Part I: Deformability of moist, densified agglomerates, Powder. Technol. 44: 227–237.1985. [33] H. G. Kristensen, P. Holm, T. Schæfer: Mechanical properties of moist agglomerates in relation to granulation mechanisms. Part II: Effects of particle size distribution, Powder. Technol. 44: 239–247.1985. [34] J. Ennis, J. Li, G. Tardos, R. Pfeffer: The influence of viscosity on the strength of an axially strained pendular liquid bridge, Chem. Eng. Sci. 45.10: 3071–3088.1990. [35] J. Ennis, G. Tardos, R. Pfeffer: A microlevel-based characterization of granulation phenomena, Powder. Technol. 65: 257–272.1991. [36] Vervaet, L. Baert, J. P. Remon: Extrusion-spheronisation. A literature review, Int. J. Pharm. 116: 131–146.1995. [37] L. Briens, D. Daniher, A. Tallevi: Monitoring high-shear granulation using sound and vibration measurements, Int. J. Pharm. 331: 54–60.2006. [38] Vojnovic, M. Moneghini, F. Rubessa: Optimalization of granulates in a high shear mixer by mixture design, Drug Dev. Ind. Pharm. 20.6: 1035–1047.1994. [39] P. Holm: Effect of impeller and chopper design on granulating in a high speed mixer, Drug Dev. Ind. Pharm. 13.9-11: 1675–1701.1987. [40] J. Kristensen, T. Schaefer, P. Kleinebudde: Direct pelletization in a rotary processor controlled by torque measurements. II: effects of changes in the content of microcrystalline cellulose. AAPS Pharmsci 2 .3: 24.2000. [41] Encyclopedia of Pharmaceutical Technology, 3rd Edition, Editor: J. Swarbrick: Informa Healthcare USA Inc., New York, 2007. [42] [36] Handbook of Pharmaceutical Excipients, 5th Edition, Editor: R. C. Rowe, P. J. Sheskey, S. C. Owen: Pharmaceutical Press, London, 2006. [43] S. R. Ghanta, R. Srinivas, C. T. Rhodes: Use of mixer torque measurements as an aid to optimizing wet granulation process. Drug Dev. Ind. Pharm. 10: 305–311.1984. [44] R. C. Rowe: Mixer torque rheometry - further advances, Pharm. Tech. Eur. 8.8: 38–48.1996. [45] [39] S. Watano: Direct control of wet granulation processes by image processing system, Powder Technol. 117: 163–172.2001. [46] J. Rantanen, S. Lehtola, P. Rämet, J. P. Mannermaa, J. Yliruusi: On-line monitoring of moisture content in an instrumented fluidized bed granulator with a multi-channel NIR moisture sensor, Powder Technol. 99: 163–170.2000. [47] S. Ganguly, J. Z. Gao: Application of on-line Focused Beam Reflectance Measurement Technology in high shear wet granulation, The AAPS Journal 7(S2): Abstract No. T3216.2005. [48] M. Whitaker, G. R. Baker, J. Westrup, P. A. Goulding, D. R. Rudd, R. M. Belchamber, M. P. Collins: Application of acoustic emission to the monitoring and end point determination of a high shear granulation process, Int. J. Pharm. 204: 79–91.2000. [49] M. Knoke: Clinical pictures of orointestinal candidiasis. Fiction or reality? Mycoses 39 Suppl. 1: 40–43.1996. [50] Martindale – The Complete Drug Reference, Editor: S. C. Sweetman: Pharmaceutical Press 36th Edition, London, 2009. [51] N. Ouchiyama, T. Tanaka: The propability of coalescence in granulation kinetics, Ind. Eng. Chem. Process Des. Dev. 14.3: 286–289.1975. [52] H. G. Kristensen, P. Holm, T. Schæfer: Mechanical properties of moist agglomerates in relation to granulation mechanisms. Part I: Deformability of moist, densified agglomerates, Powder. Technol. 44: 227–237.1985. [53] H. G. Kristensen, P. Holm, T. Schæfer: Mechanical properties of moist agglomerates in relation to granulation mechanisms. Part II: Effects of particle size distribution, Powder. Technol. 44: 239–247.1985. [54] J. Ennis, J. Li, G. Tardos, R. Pfeffer: The influence of viscosity on the strength of an axially strained pendular liquid bridge, Chem. Eng. Sci. 45.10: 3071–3088.1990. [55] J. Ennis, G. Tardos, R. Pfeffer: A microlevel-based characterization of granulation phenomena, Powder. Technol. 65: 257–272.1991. [56] Vervaet, L. Baert, J. P. Remon: Extrusion-spheronisation. A literature review, Int. J. Pharm. 116: 131–146.1995.
Copyright
Copyright © 2023 Ankit Agrawal, Sourabh D Jain, Ashutosh K Das, Arun K Gupta. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.

Download Paper
Paper Id : IJRASET49427
Publish Date : 2023-03-07
ISSN : 2321-9653
Publisher Name : IJRASET
DOI Link : Click Here
 Submit Paper Online
Submit Paper Online

